2. Tanaka A, Takikawa H, Miwa H, Shimosegawa T, Mochida S, Koike K. 2016; Changing nomenclature for PBC from "primary biliary cirrhosis" to "primary biliary cholangitis". Hepatol Res. 46:725–726. DOI:
10.1111/hepr.12729. PMID:
27101399.

3. Lindor KD, Jorgensen RA, Therneau TM, Malinchoc M, Dickson ER. 1997; Ursodeoxycholic acid delays the onset of esophageal varices in primary biliary cirrhosis. Mayo Clin Proc. 72:1137–1140. DOI:
10.4065/72.12.1137. PMID:
9413293.

4. Poupon RE, Lindor KD, Cauch-Dudek K, Dickson ER, Poupon R, Heathcote EJ. 1997; Combined analysis of randomized controlled trials of ursodeoxycholic acid in primary biliary cirrhosis. Gastroenterology. 113:884–890. DOI:
10.1016/S0016-5085(97)70183-5.

5. Floreani A, Caroli D, Variola A, et al. 2011; A 35-year follow-up of a large cohort of patients with primary biliary cirrhosis seen at a single centre. Liver Int. 31:361–368. DOI:
10.1111/j.1478-3231.2010.02366.x. PMID:
21059170.

6. Cuperus FJ, Halilbasic E, Trauner M. 2014; Fibrate treatment for primary biliary cirrhosis. Curr Opin Gastroenterol. 30:279–286. DOI:
10.1097/MOG.0000000000000056. PMID:
24625898.

7. Corpechot C, Carrat F, Poupon R, Poupon RE. 2002; Primary biliary cirrhosis: incidence and predictive factors of cirrhosis development in ursodiol-treated patients. Gastroenterology. 122:652–658. DOI:
10.1053/gast.2002.31880. PMID:
11874998.

8. Parés A, Caballería L, Rodés J. 2006; Excellent long-term survival in patients with primary biliary cirrhosis and biochemical response to ursodeoxycholic Acid. Gastroenterology. 130:715–720. DOI:
10.1053/j.gastro.2005.12.029. PMID:
16530513.

9. Gebel T, Arand M, Oesch F. 1992; Induction of the peroxisome proliferator activated receptor by fenofibrate in rat liver. FEBS Lett. 309:37–40. DOI:
10.1016/0014-5793(92)80734-X.

10. Schoonjans K, Staels B, Auwerx J. 1996; Role of the peroxisome proliferator-activated receptor (PPAR) in mediating the effects of fibrates and fatty acids on gene expression. J Lipid Res. 37:907–925. DOI:
10.1016/S0022-2275(20)42003-6.

11. Honda A, Ikegami T, Nakamuta M, et al. 2013; Anticholestatic effects of bezafibrate in patients with primary biliary cirrhosis treated with ursodeoxycholic acid. Hepatology. 57:1931–1941. DOI:
10.1002/hep.26018. PMID:
22911624.

12. Willson TM, Brown PJ, Sternbach DD, Henke BR. 2000; The PPARs: from orphan receptors to drug discovery. J Med Chem. 43:527–550. DOI:
10.1021/jm990554g. PMID:
10691680.

13. Ghonem NS, Boyer JL. 2013; Fibrates as adjuvant therapy for chronic cholestatic liver disease: its time has come. Hepatology. 57:1691–1693. DOI:
10.1002/hep.26155. PMID:
23174993. PMCID:
PMC4048949.

15. Dohmen K, Mizuta T, Nakamuta M, Shimohashi N, Ishibashi H, Yamamoto K. 2004; Fenofibrate for patients with asymptomatic primary biliary cirrhosis. World J Gastroenterol. 10:894–898. DOI:
10.3748/wjg.v10.i6.894. PMID:
15040040. PMCID:
PMC4727018.

16. Walker LJ, Newton J, Jones DE, Bassendine MF. 2009; Comment on biochemical response to ursodeoxycholic acid and long-term prognosis in primary biliary cirrhosis. Hepatology. 49:337–338. DOI:
10.1002/hep.22670. PMID:
19035342.

17. Nakai S, Masaki T, Kurokohchi K, Deguchi A, Nishioka M. 2000; Combination therapy of bezafibrate and ursodeoxycholic acid in primary biliary cirrhosis: a preliminary study. Am J Gastroenterol. 95:326–327. DOI:
10.1111/j.1572-0241.2000.01667.x. PMID:
10638623.

18. Kanda T, Yokosuka O, Imazeki F, Saisho H. 2003; Bezafibrate treatment: a new medical approach for PBC patients? J Gastroenterol. 38:573–578. DOI:
10.1007/s00535-002-1102-7. PMID:
12825134.

19. Itakura J, Izumi N, Nishimura Y, et al. 2004; Prospective randomized crossover trial of combination therapy with bezafibrate and UDCA for primary biliary cirrhosis. Hepatol Res. 29:216–222. DOI:
10.1016/j.hepres.2004.04.001. PMID:
15288014.

20. Iwasaki S, Ohira H, Nishiguchi S, et al. 2008; The efficacy of ursodeoxycholic acid and bezafibrate combination therapy for primary biliary cirrhosis: a prospective, multicenter study. Hepatol Res. 38:557–564. DOI:
10.1111/j.1872-034X.2007.00305.x. PMID:
18452482.

21. Liberopoulos EN, Florentin M, Elisaf MS, Mikhailidis DP, Tsianos E. 2010; Fenofibrate in primary biliary cirrhosis: a pilot study. Open Cardiovasc Med J. 4:120–126. DOI:
10.2174/1874192401004010120. PMID:
20556204. PMCID:
PMC2885597.

22. Levy C, Peter JA, Nelson DR, et al. 2011; Pilot study: fenofibrate for patients with primary biliary cirrhosis and an incomplete response to ursodeoxycholic acid. Aliment Pharmacol Ther. 33:235–242. DOI:
10.1111/j.1365-2036.2010.04512.x. PMID:
21083674.

23. Han XF, Wang QX, Liu Y, et al. 2012; Efficacy of fenofibrate in Chinese patients with primary biliary cirrhosis partially responding to ursodeoxycholic acid therapy. J Dig Dis. 13:219–224. DOI:
10.1111/j.1751-2980.2012.00574.x. PMID:
22435507.

24. Dohmen K, Tanaka H, Haruno M. 2013; Effectiveness of fenofibrate in comparison to bezafibrate for patients with asymptomatic primary biliary cirrhosis. Fukuoka Igaku Zasshi. 104:350–361.
25. Cheung AC, Lapointe-Shaw L, Kowgier M, et al. 2016; Combined ursodeoxycholic acid (UDCA) and fenofibrate in primary biliary cholangitis patients with incomplete UDCA response may improve outcomes. Aliment Pharmacol Ther. 43:283–293. DOI:
10.1111/apt.13465. PMID:
26559762.

26. Hegade VS, Khanna A, Walker LJ, Wong LL, Dyson JK, Jones DEJ. 2016; Long-term fenofibrate treatment in primary biliary cholangitis improves biochemistry but not the UK-PBC risk score. Dig Dis Sci. 61:3037–3044. DOI:
10.1007/s10620-016-4250-y. PMID:
27435324.

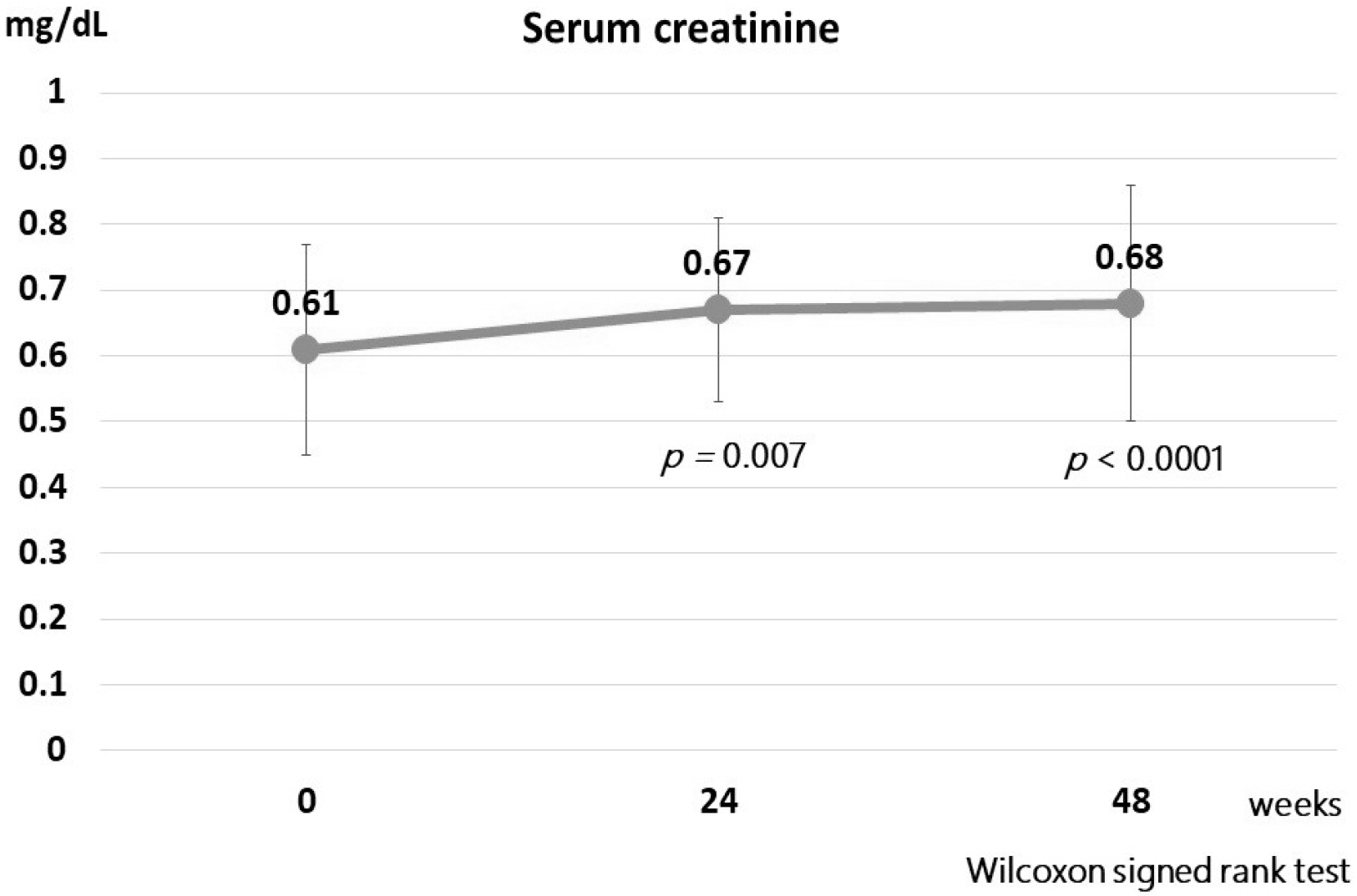
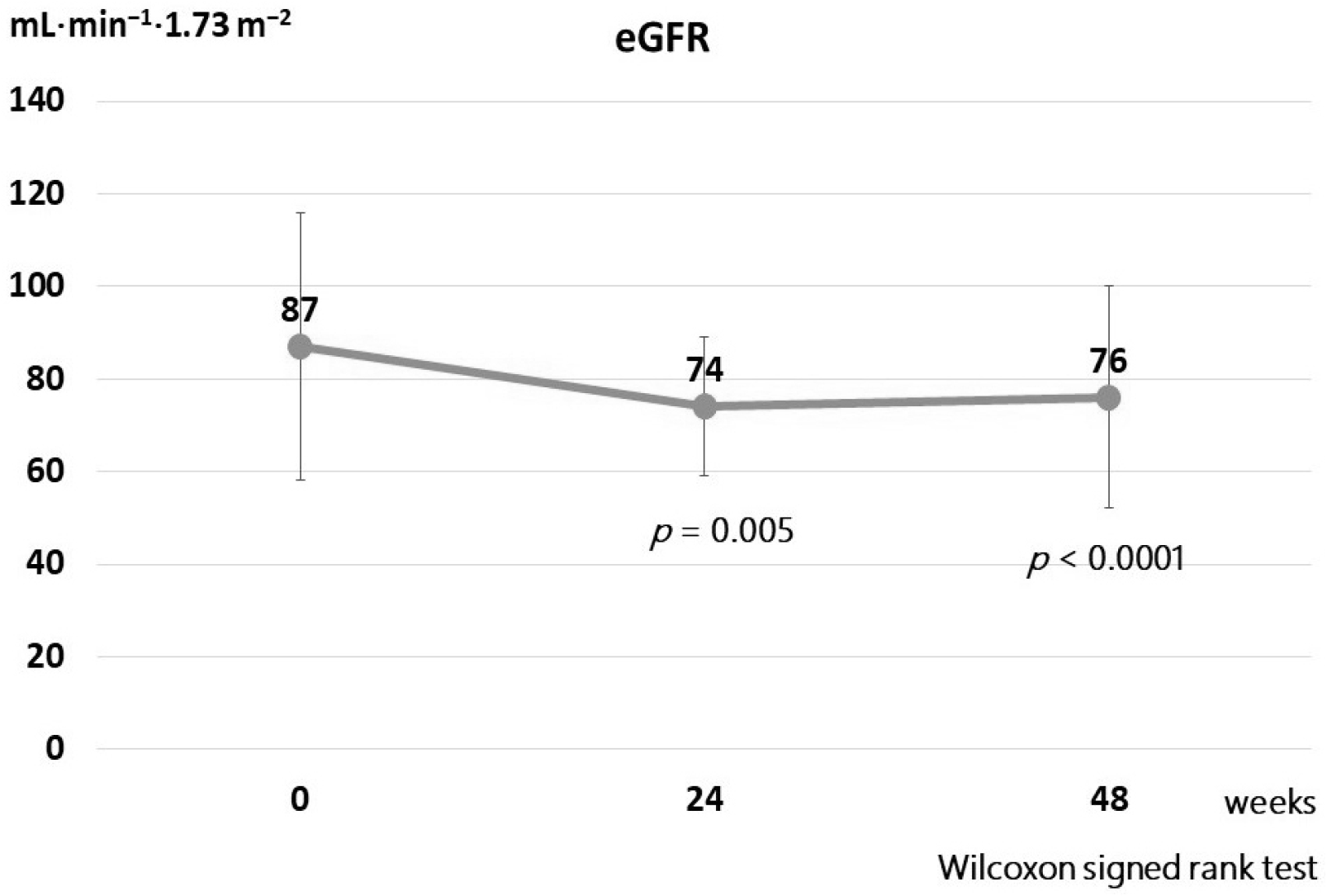
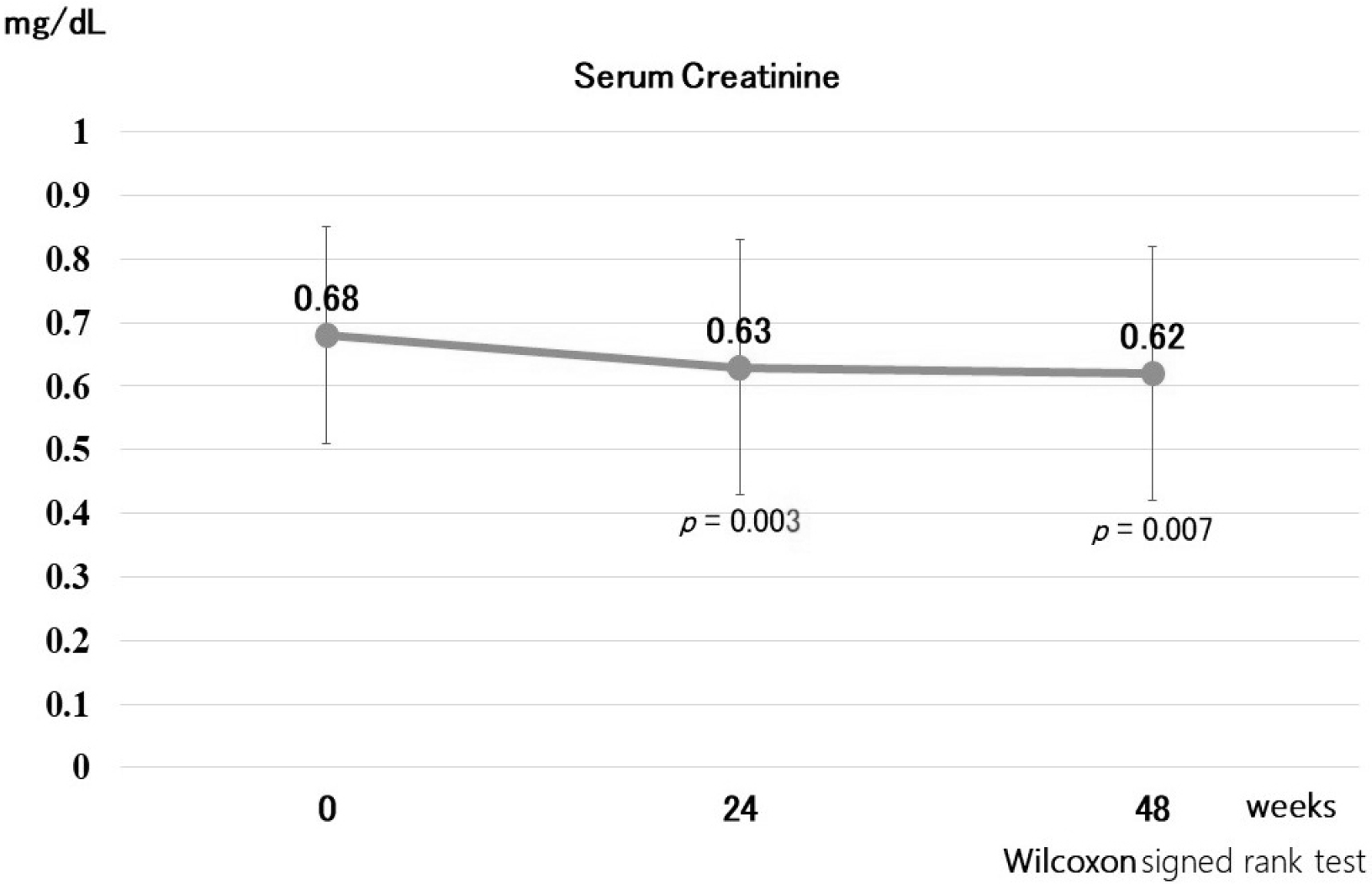
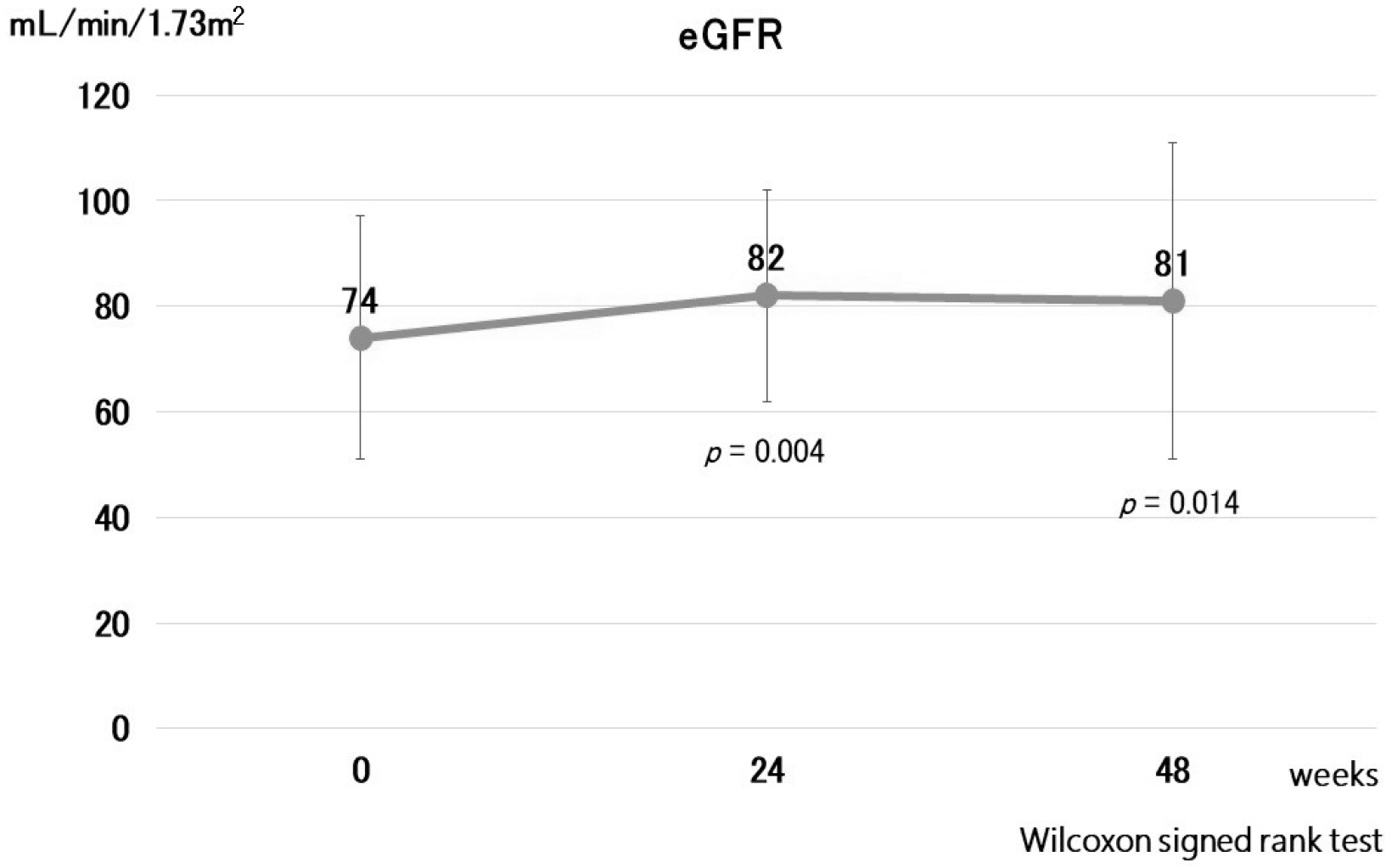
27. Lipscombe J, Lewis GF, Cattran D, Bargman JM. 2001; Deterioration in renal function associated with fibrate therapy. Clin Nephrol. 55:39–44.
30. Hottelart C, El Esper N, Rose F, Achard JM, Fournier A. 2002; Fenofibrate increases creatininemia by increasing metabolic production of creatinine. Nephron. 92:536–541. DOI:
10.1159/000064083. PMID:
12372935.

31. Ishibashi S, Yamashita S, Arai H, et al. 2016; Effects of K-877, a novel selective PPARα modulator (SPPARMα), in dyslipidaemic patients: a randomized, double blind, active- and placebo-controlled, phase 2 trial. Atherosclerosis. 249:36–43. DOI:
10.1016/j.atherosclerosis.2016.02.029. PMID:
27062408.

32. Ishibashi S, Arai H, Yokote K, et al. 2018; Efficacy and safety of pemafibrate (K-877), a selective peroxisome proliferator-activated receptor α modulator, in patients with dyslipidemia: results from a 24-week, randomized, double blind, active-controlled, phase 3 trial. J Clin Lipidol. 12:173–184. DOI:
10.1016/j.jacl.2017.10.006. PMID:
29203092.
33. Yamashita S, Masuda D, Matsuzawa Y. 2019; Clinical applications of a novel selective PPARα modulator, pemafibrate, in dyslipidemia and metabolic diseases. J Atheroscler Thromb. 26:389–402. DOI:
10.5551/jat.48918. PMID:
30930344. PMCID:
PMC6514171.

34. Joshita S, Umemura T, Yamashita Y, et al. 2019; Biochemical and plasma lipid responses to pemafibrate in patients with primary biliary cholangitis. Hepatol Res. 49:1236–1243. DOI:
10.1111/hepr.13361. PMID:
31077509.

35. Working Subgroup (English version) for Clinical Practice Guidelines for Primary Biliary Cirrhosis. Guidelines for the management of primary biliary cirrhosis: the Intractable Hepatobiliary Disease Study Group supported by the Ministry of Health, Labour and Welfare of Japan. Hepatol Res. 2014; 44 Suppl S1:71–90. DOI:
10.1111/hepr.12270. PMID:
24397841.
36. Lindor KD, Gershwin ME, Poupon R, et al. 2009; Primary biliary cirrhosis. Hepatology. 50:291–308. DOI:
10.1002/hep.22906. PMID:
19554543.

37. Zhang LN, Shi TY, Shi XH, et al. 2013; Early biochemical response to ursodeoxycholic acid and long-term prognosis of primary biliary cirrhosis: results of a 14-year cohort study. Hepatology. 58:264–272. DOI:
10.1002/hep.26322. PMID:
23408380.

38. Hazzan R, Tur-Kaspa R. 2010; Bezafibrate treatment of primary biliary cirrhosis following incomplete response to ursodeoxycholic acid. J Clin Gastroenterol. 44:371–373. DOI:
10.1097/MCG.0b013e3181c115b3. PMID:
19881358.

39. Lens S, Leoz M, Nazal L, Bruguera M, Parés A. 2014; Bezafibrate normalizes alkaline phosphatase in primary biliary cirrhosis patients with incomplete response to ursodeoxycholic acid. Liver Int. 34:197–203. DOI:
10.1111/liv.12290. PMID:
23998489.

40. Corpechot C, Chazouillères O, Rousseau A, et al. 2018; A placebo-controlled trial of bezafibrate in primary biliary cholangitis. N Engl J Med. 378:2171–2181. DOI:
10.1056/NEJMoa1714519. PMID:
29874528.

41. Ghonem NS, Auclair AM, Hemme CL, et al. 2020; Fenofibrate improves liver function and reduces the toxicity of the bile acid pool in patients with primary biliary cholangitis and primary sclerosing cholangitis who are partial responders to ursodiol. Clin Pharmacol Ther. 108:1213–1223. DOI:
10.1002/cpt.1930. PMID:
32480421.

42. Dohmen K, Tanaka H, Haruno M, Aishima S. 2016; Biochemical and histological effectiveness of long-term use of fenofibrate for asymptomatic primary biliary cholangitis. J Gastroenterol Hepatol Res. 5:2125–2131. DOI:
10.17554/j.issn.2224-3992.2016.05.635.

43. Pineda Torra I, Claudel T, Duval C, Kosykh V, Fruchart JC, Staels B. 2003; Bile acids induce the expression of the human peroxisome proliferator-activated receptor alpha gene via activation of the farnesoid X receptor. Mol Endocrinol. 17:259–272. DOI:
10.1210/me.2002-0120. PMID:
12554753.
44. Smit JJ, Schinkel AH, Oude Elferink RP, et al. 1993; Homozygous disruption of the murine mdr2 P-glycoprotein gene leads to a complete absence of phospholipid from bile and to liver disease. Cell. 75:451–462. DOI:
10.1016/0092-8674(93)90380-9.

45. Chianale J, Vollrath V, Wielandt AM, et al. 1996; Fibrates induce mdr2 gene expression and biliary phospholipid secretion in the mouse. Biochem J. 314:781–786. DOI:
10.1042/bj3140781. PMID:
8615769. PMCID:
PMC1217124.

46. Matsumoto T, Miyazaki H, Nakahashi Y, et al. 2004; Multidrug resistance3 is in situ detected in the liver of patients with primary biliary cirrhosis, and induced in human hepatoma cells by bezafibrate. Hepatol Res. 30:125–136. DOI:
10.1016/j.hepres.2004.08.015. PMID:
15588777.

47. Unal A, Torun E, Sipahioglu MH, et al. 2008; Fenofibrate-induced acute renal failure due to massive rhabdomyolysis after coadministration of statin in two patients. Intern Med. 47:1017–1019. DOI:
10.2169/internalmedicine.47.0939. PMID:
18520113.

48. Forsblom C, Hiukka A, Leinonen ES, Sundvall J, Groop PH, Taskinen MR. 2010; Effects of long-term fenofibrate treatment on markers of renal function in type 2 diabetes: the FIELD Helsinki substudy. Diabetes Care. 33:215–220. DOI:
10.2337/dc09-0621. PMID:
19846798. PMCID:
PMC2809252.

49. Jun M, Zhu B, Tonelli M, et al. 2012; Effects of fibrates in kidney disease: a systematic review and meta-analysis. J Am Coll Cardiol. 60:2061–2071. DOI:
10.1016/j.jacc.2012.07.049. PMID:
23083786.
50. Arai H, Yamashita S, Yokote K, et al. 2018; Efficacy and safety of pemafibrate versus fenofibrate in patients with high triglyceride and low HDL cholesterol levels: a multicenter, placebo-controlled, double-blind, randomized trial. J Atheroscler Thromb. 25:521–538. DOI:
10.5551/jat.44412. PMID:
29628483. PMCID:
PMC6005227.

51. Duan W, Ou X, Wang X, et al. 2018; Efficacy and safety of fenofibrate add-on therapy for patients with primary biliary cholangitis and a suboptimal response to UDCA. Rev Esp Enferm Dig. 110:557–563. DOI:
10.17235/reed.2018.5533/2018. PMID:
29739227.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download