Abstract
Background
Considering that pulmonary embolism (PE) is one of the leading causes of mortality among pregnant women and that the D-dimer level in pregnancy can be highly fluctuating, a new and reliable D-dimer reference value is essential to identifying PE in this group of patients. Hence, the present study aimed to evaluate the diagnostic effect of D-dimer testing in pregnant women with suspected PE.
Methods
This study recruited 100 women with confirmed pregnancy or six weeks after delivery or abortion with suspected PE symptoms. Wells criteria, D-dimer values, and pregnancy trimesters were recorded. Definitive PE results were obtained using multidetector computed tomography (MDCT) or pulmonary ventilation/perfusion scans.
Results
D-dimer cut-off point in PE diagnosis was higher than 1,447 mg/L [sensitivity, 87.5%; specificity, 63.04%; area under the curve (AUC)=0.735; P=0.003]. In addition, the combination of Wells criteria with the D-dimer test indicated that the cut-off points of D-dimer in PE likely and unlikely women were 1,962 and 1,447 mg/L, respectively, and had acceptable and significant diagnostic value in PE detection. In addition, the diagnostic value of D-dimer in pregnancy trimesters was not found to be significant (P>0.05).
Conclusion
The new cut-off points of 1,447 and 1,962 mg/L were determined for D-dimer in pregnant women with likely and unlikely PE, respectively. Moreover, the new cut-off points in the first and second trimesters of pregnancy were 1,701 mg/L and 1,451 mg/L, respectively, which indicated no statistically acceptable diagnostic value.
Pregnancy is a hypercoagulable state due to increased coagulation factors, especially fibrinogen and factor VII [1]. An increase in these factors counteracts severe bleeding during delivery. However, a hypercoagulable state can lead to thromboembolism, which accounts for 20% of all deaths during pregnancy. The mortality rate of thromboembolism is higher than that of bleeding and hypertension. The chance of developing pulmonary embolism (PE) in pregnant women is 1 per 1,000 cases, which is 2–4 times higher than the reported chance for other groups of women. In addition, the possibility of developing PE in a cesarean section is more than that in normal delivery. Multiple pregnancies, age >35 years, absolute rest, obesity, family history of PE, previous embolism, and hypertension increase the risk of embolism [2]. In the prenatal period, the risk of developing deep vein thrombosis (DVT) and PE was higher in the first and third trimesters than in the second trimester. Moreover, blood clot formation in the postnatal period is higher than that before delivery [3, 4].
In cases other than pregnancy, VTE and PE diagnostic pathways are based on a combination of the clinical scoring system (Wells criteria), measurement of D-dimer level, blood testing and ultrasound imaging, ventilation/perfusion, or computed tomography (CT) scans [5].
A screening test with appropriate and noninvasive sensitivity and specificity is of great significance in diagnosing PE during pregnancy. In this regard, imaging techniques expose the fetus to radiation. Moreover, the risk of developing breast cancer compared to the typical underlying breast cancer risk in women aged <35 years increased by 16% using computed tomography. Therefore, the presentation of a system for classifying pregnant women into low-risk groups for PE, thus avoiding unnecessary imaging, would be highly valued. However, unfortunately, there is no valid scoring system during pregnancy, and clinical manifestations can be confused with healthy pregnancy characteristics [6].
The use of fibrin-linked markers during pregnancy will help rule out the possibility of embolism and reduce the need for imaging. D-dimers are specific fibrin-derived cross-links that are produced by plasmin degradation of fibrin; therefore, their concentrations increase during thrombolysis. Many studies have concluded that using the D-dimer test and clinical evaluation in non-pregnant individuals is acceptable and reduces the need for hospital admissions [6, 7].
However, diagnostic methods used for pregnant women with suspected PE have not been extensively examined, and no definitive conclusion has been reached regarding the role of the D-dimer test in this respect [8]. The use of conventional D-dimer threshold in pregnant women has some limitations as D-dimer levels increase during pregnancy due to continued coagulation and fibrinolysis during normal placental development and consequently increases the number of false-positive results [9]. Given this point, some studies have suggested higher thresholds for D-dimers during pregnancy [10, 11]. A study conducted in 2015 revealed that healthy pregnant women had a moderate increase in D-dimer levels during pregnancy [12]. Another study indicated that the modified Wells score (MWS) along with negative D-dimer had a higher negative predictive value (>99%) for predicting PE in non-pregnant individuals, although this finding has not been confirmed in the pregnant population [13]. Therefore, the present study aimed to determine the reference value for the D-dimer range in pregnancy trimesters so that this parameter can be used as a predictor of PE during pregnancy.
We conducted a prospective cross-sectional study on pregnant or postpartum women clinically suspected of PE and admitted at Al-Zahra Hospital, the largest center of obstetrics and pulmonary disease in Isfahan, Iran, between March 2017 and February 2019. Women with confirmed pregnancy or at six-week following delivery or abortion who had signs and symptoms suspicious for PE were included. Clinically suspected PE was defined as the acute onset of new or worsening hypoxia, tachycardia, or unexplained dyspnea. Patients who had D-dimer and lower extremity color Doppler ultrasonography underwent multidetector computed tomography (MDCT) with the PE protocol. The selection of patients for MDCT was based on clinical suspicion and physician discretion. Therefore, the patients underwent MDCT regardless of their D-dimer level or other known valid criteria for non-pregnant patients. Patients with inherited or acquired thrombophilia based on documented medical records and those with a history of anticoagulant therapy were excluded from the study.
This study, which met the Declaration of Helsinki criteria, was approved by the Ethics Committee of Isfahan University of Medical Sciences. Written informed consent was obtained from eligible women who voluntarily agreed to participate in the study.
Patients’ demographic and clinical characteristics, including age, gestational age, trimester, number of fetuses (singleton vs. twin or multiple pregnancies), hormone therapy, fever, and chest pain, were collected. The Wells criteria were also determined and recorded [14]. The D-dimer level was measured using the VIDAS assay (bioMérieux), a highly sensitive quantitative enzyme-linked immunoassay (ELISA) to determine fibrin degradation products in human plasma.
Women with and without PE were identified using MDCT. The MDCT interpretation, as reported by the radiologists at our center, was used for the definitive diagnosis of PE. Accordingly, the score of the Wells criteria and the frequency of each symptom based on these criteria have been compared between PE and non-PE patients (Table 1). Clinical and demographic characteristics were compared between women with and without PE.
The administered Wells criteria included symptoms of DVT (3 points), PE was the most likely diagnosis (3 points), tachycardia >100 per minute (1.5 points), immobilization for 3 days/surgery within the previous 4 weeks (1.5 points), previous history of DVT/PE (1.5 points), hemoptysis (1 point), and active malignancy (1 point). Patients with ≤4 score were considered unlikely; otherwise, they were likely to have PE [15].
The SPSS software (version 24.0, SPSS Inc., IL, USA) was used for statistical analysis. Categorical variables were expressed as percentages and compared using the chi-squared test or Fisher's exact test. Continuous variables were compared using a t-test if normally distributed and a Mann- Whitney U test if non-normally distributed. To determine the cut-off value of the D-dimer level for diagnosing PE, we performed a receiver operating characteristic (ROC) analysis to determine the optimal sensitivity and specificity with respect to the D-dimer value. The area under the curve (AUC) was used to evaluate the cut-off value. The negative predictive value (NPV) and positive predictive value (PPV) were also evaluated. Statistical significance was set at P< 0.05.
In the present study, out of 100 pregnant women with suspected PE, 8% and 92% were with and without PE, respectively. Maternal and gestational ages in the sample were 30.38±6.67 years and 30.20±7.55 weeks, respectively. Of the eight women with PE, two (25%) and six (75%) patients were in the 1st and 3rd trimesters, respectively (P=0.015). The D-dimer level in women with PE was significantly higher than that in non-PE patients (median, 2,110 vs. 1,274; P=0.029). However, the levels of D-dimer in these patients in the three trimesters did not differ significantly. The mean score of Wells criteria in women with PE with the mean of 4.97±1.97 was significantly more than that of the non-PE women with a mean of 3.53±0.76 (P=0.008) (Table 1, Fig. 1). According to the Wells criteria, 36 women (36%) [5 with (62.5%) and 31 without (33.7%) confirmed PE] were likely to have PE (Wells score>4). The remaining patients (66.3%) were PE unlikely.
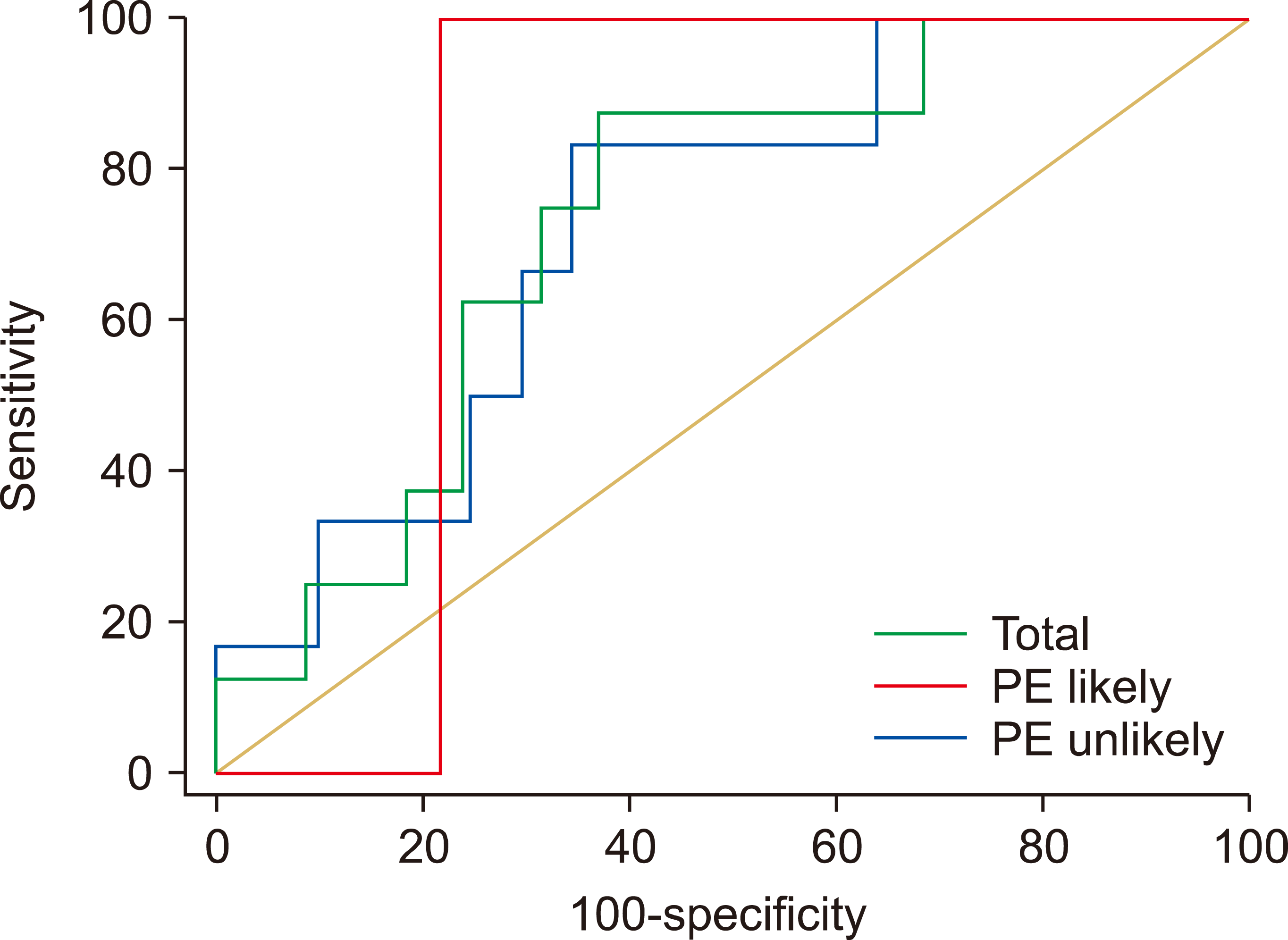
The diagnostic value of the Wells criteria in PE diagnosis was weak and insignificant (AUC=0.557, P=0.497). D-dimer level with a cut-off point greater than 1,447 mg/L and a sensitivity and specificity of 87.5% and 63.04%, respectively, indicated a significant diagnostic value for PE detection (AUC=0.735, P=0.003). Considering the risk of PE, it was also revealed that the mentioned diagnostic criteria had a significant and acceptable diagnostic power in PE detection in patients with probable PE (cut-off, 1,962 mg/L; P<0.001) and unlikely PE (cut-off, 1,447 mg/L; P=0.019) (Table 2, Fig. 2).
In addition, the results of D-dimer evaluation considering the pregnancy trimester revealed that although the levels of D-dimer in women in the second and third trimesters with a median of 1,348 and 1,327 mg/L, respectively, was more than the levels of D-dimer in women in the first trimester of pregnancy with a median of 592, this criteria did not reveal an appropriate PE diagnostic value (P>0.05) (Table 3).
The diagnosis of venous thromboembolism (VTE) during pregnancy remains a challenging issue in medicine. Physicians are searching for a precise noninvasive means to accurately diagnose VTE incidence in pregnant women [12, 16]. The significance of VTE-related mortality and morbidity led us to design the current study to establish a diagnostic reference range for D-dimer in pregnant women suspected of VTE.
Various studies have represented a 2- to 6-fold increase in D-dimer levels during pregnancy [17]; however, this increase is not necessarily significant [8, 18, 19]. We observed considerable elevation in D-dimer levels during pregnancy, a trend that gradually continued from the first trimester to the third trimester.
Our results revealed significantly higher D-dimer levels among those with a definitive diagnosis of VTE using computed tomography and Wells scores than the healthy ones. The mere assessment of D-dimer as a stand-alone factor for the diagnosis of VTE during pregnancy at a cut-off level of 1,447 mg/L showed a statistically significant specificity, sensitivity, and NPV of 63.04%, 87.50%, and 98.3%, respectively. In further evaluations, we found no difference in assessing D-dimer levels in the risk assessment of PE based on pregnancy trimesters. The insignificant difference between the assessed groups may be attributed to our study's small sample population, which is a major drawback.
Kline et al. [11] conducted a follow-up study on 50 women from preconception to postpartum to achieve a cut-off point for D-dimer levels in different trimesters of pregnancy contributing to VTE. The cut-offs were 430 mg/L, 580 mg/L, 830 mg/L, and 1,200 mg/L for preconception, first trimester, second trimester, third trimester, and post-partum, respectively [11]. In another study, a cut-off point of 230 mg/L was considered for D-dimer and normal ranges were detected in 84%, 33%, and 1% of the females in the first, second, and third trimesters of pregnancy, respectively. Sensitivity and NPV of 100% were reported for D-dimer in this study; however, the diagnostic value of this biomarker decreased with increasing gestational age [20]. Wang et al. [18] conducted another investigation to set a threshold for D-dimer in each of the pregnancy trimesters and raised cut-offs, including 660 mg/L, 2,290 mg/L, and 3,120 mg/L. The thresholds mentioned in this study are surprisingly more than ours, which may be attributed to ethnicity, the wide age range of the studied population, or the analyzer used for the measurement of D-dimer [18]. A cut-off of 150 mg/L in another study to detect pulmonary embolism in pregnancy revealed outranges in 95.1%, 98.7%, and 100% of the cases in the first, second, and third trimesters of pregnancy, respectively. Therefore, by altering the cut-off to 500 mg/L, 4.8% and 23.8% of pregnant women in the second and third trimesters exceeded the cut-off values. In conclusion, they mentioned that D-dimer values for making the diagnosis of VTE would decrease with an increase in the pregnancy gestational age, while it is still a valuable means for ruling out the diagnosis of VTE [21]. Nevertheless, the significant difference between the mentioned studies and ours is their design, as we have conducted the current report on pregnant women with a definitive diagnosis of PTE, while the others have been conducted on cases suspected of VTE.
To the best of our knowledge, the current study is the first to assess D-dimer levels for the diagnosis of PTE. However, Chan et al. [22] performed a similar study on pregnant patients with a documented diagnosis of DVT. They assessed a wide range of D-dimer levels from 570 mg/L to 1,890 mg/L to assess D-dimer values for DVT diagnosis, in which the sensitivity and specificity ranged from 80% to 100% and 62% to 79%, respectively, with an increase in the proposed cut-off levels [22].
On the contrary, we found insignificant results by the evaluation of Wells score values alone for the assessment of VTE; these findings are consistent with the presentations of the literature as the clinical manifestations of VTE in pregnancy are not reliable, whereas up to 70% of the pregnant women have the complaint of shortness of breath or edema in lower extremity and pain are the other common conditions that pregnant women may struggle with [20]; however, we found that the concurrent use of D-dimer with Wells score likely for PE showed a significant probability of VTE at a cut-off of 1,962 mg/L, with a sensitivity of 99%, specificity of 78.12%, and NPV of 99%. Concurrent use of Wells criteria and D-dimer in pregnancy for the first time has significantly improved the probability of PE diagnosis, while the studies conducted on the general population have confirmed our findings using the YEARS criteria [23, 24].
In summary, we found a cut-off of 1,447 mg/L with a specificity, sensitivity, and NPV of 63.04%, 87.50%, and 98.3%, respectively, for PE diagnosis in pregnant women regardless of their gestational age. With the addition of the Wells score contributing to the likelihood of pulmonary embolism, the threshold increased to 1,962 mg/L with a sensitivity of 99%, specificity of 78.12%, and NPV of 99%, regardless of the mentioned factors. The assessment of trimester-related risk of VTE development revealed insignificant associations. Variability in D-dimer thresholds in the different studies conducted all over the world may be attributed to diverse factors such as ethnicity and environmental factors, and to a greater extent, to the analyzers used for the measurement of D-dimer. Therefore, further evaluations in which the confounders are well-controlled are strongly recommended.
As mentioned above, failure to provide different cut-offs for D-dimer according to the pregnancy trimesters is the most significant limitation of our study. This inability is partly due to the small sample size of the study, which not only restricted us to divide the population into three trimesters of pregnancy but also limited the generalizability of the outcomes. Therefore, further multicenter studies with larger study populations are recommended. Failure to control for confounding variables affecting the study outcomes is another remarkable limitation of our study.
According to the results of the present study addressing pregnant women with suspected PE, the Wells criteria alone were insufficient to identify PE. However, the combination of these criteria with the D-dimer level indicated that the D-dimer value at 1,962 mg/L cut-off point for PE likely patients and D-dimer value at 1,447 mg/L cut-off point for PE unlikely patients were significant in diagnosing PE. In addition, although the level and distribution of D-dimer were significant with increasing gestational age, the reference value for D-dimer in different pregnancy trimesters was not significant. Therefore, further studies are required to confirm this hypothesis.
REFERENCES
1. Heit JA, Kobbervig CE, James AH, Petterson TM, Bailey KR, Melton LJ 3rd. 2005; Trends in the incidence of venous thrombo-embolism during pregnancy or postpartum: a 30-year population- based study. Ann Intern Med. 143:697–706. DOI: 10.7326/0003-4819-143-10-200511150-00006. PMID: 16287790.
2. Leung AN, Bull TM, Jaeschke R, et al. 2011; An official American Thoracic Society/Society of Thoracic Radiology clinical practice guideline: evaluation of suspected pulmonary embolism in pregnancy. Am J Respir Crit Care Med. 184:1200–8. DOI: 10.1164/rccm.201108-1575ST. PMID: 22086989.

3. Marik PE, Plante LA. 2008; Venous thromboembolic disease and pregnancy. N Engl J Med. 359:2025–33. DOI: 10.1056/NEJMra0707993. PMID: 18987370.

4. Jacobsen AF, Skjeldestad FE, Sandset PM. 2008; Incidence and risk patterns of venous thromboembolism in pregnancy and puerperium--a register-based case-control study. Am J Obstet Gynecol. 198:233e1–7. DOI: 10.1016/j.ajog.2007.08.041. PMID: 17997389.

5. Arya R. 2011; How I manage venous thromboembolism in pregnancy. Br J Haematol. 153:698–708. DOI: 10.1111/j.1365-2141.2011.08684.x. PMID: 21501137.

6. Wells PS, Anderson DR, Rodger M, et al. 2003; Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl J Med. 349:1227–35. DOI: 10.1056/NEJMoa023153. PMID: 14507948.

7. Kearon C, Julian JA, Newman TE, Ginsberg JS. 1998; Noninvasive diagnosis of deep venous thrombosis. McMaster Diagnostic Imaging Practice Guidelines Initiative. Ann Intern Med. 128:663–77. DOI: 10.7326/0003-4819-128-8-199804150-00011. PMID: 9537941.
8. Hedengran KK, Andersen MR, Stender S, Szecsi PB. 2016; Large D-dimer fluctuation in normal pregnancy: a longitudinal cohort study of 4,117 samples from 714 healthy Danish women. Obstet Gynecol Int. 2016:3561675. DOI: 10.1155/2016/3561675. PMID: 27190521. PMCID: PMC4852125.

9. Szecsi PB, Jørgensen M, Klajnbard A, Andersen MR, Colov NP, Stender S. 2010; Haemostatic reference intervals in pregnancy. Thromb Haemost. 103:718–27. DOI: 10.1160/TH09-10-0704. PMID: 20174768.

10. Kawaguchi S, Yamada T, Takeda M, et al. 2013; Changes in d-dimer levels in pregnant women according to gestational week. Pregnancy Hypertens. 3:172–7. DOI: 10.1016/j.preghy.2013.03.003. PMID: 26106030.

11. Kline JA, Williams GW, Hernandez-Nino J. 2005; D-dimer concentrations in normal pregnancy: new diagnostic thresholds are needed. Clin Chem. 51:825–9. DOI: 10.1373/clinchem.2004.044883. PMID: 15764641.

12. Murphy N, Broadhurst DI, Khashan AS, Gilligan O, Kenny LC, O'Donoghue K. 2015; Gestation-specific D-dimer reference ranges: a cross-sectional study. BJOG. 122:395–400. DOI: 10.1111/1471-0528.12855. PMID: 24828148.

13. van Belle A, Büller HR, Huisman MV, et al. 2006; Effectiveness of managing suspected pulmonary embolism using an algorithm combining clinical probability, D-dimer testing, and computed tomography. JAMA. 295:172–9. DOI: 10.1001/jama.295.2.172. PMID: 16403929.

14. Goodacre S, Horspool K, Nelson-Piercy C, et al. 2019; The DiPEP study: an observational study of the diagnostic accuracy of clinical assessment, D-dimer and chest x-ray for suspected pulmonary embolism in pregnancy and postpartum. BJOG. 126:383–92. DOI: 10.1111/1471-0528.15286. PMID: 29782079. PMCID: PMC6519154.

15. Wolf SJ, McCubbin TR, Feldhaus KM, Faragher JP, Adcock DM. 2004; Prospective validation of Wells Criteria in the evaluation of patients with suspected pulmonary embolism. Ann Emerg Med. 44:503–10. DOI: 10.1016/j.annemergmed.2004.04.002. PMID: 15520710.

16. Lippi G, Cervellin G, Casagranda I, Morelli B, Testa S, Tripodi A. 2014; D-dimer testing for suspected venous thromboembolism in the emergency department. Consensus document of AcEMC, CISMEL, SIBioC, and SIMeL. Clin Chem Lab Med. 52:621–8. DOI: 10.1515/cclm-2013-0706. PMID: 24152904.

17. Grossman KB, Arya R, Peixoto AB, Akolekar R, Staboulidou I, Nicolaides KH. 2016; Maternal and pregnancy characteristics affect plasma fibrin monomer complexes and D-dimer reference ranges for venous thromboembolism in pregnancy. Am J Obstet Gynecol. 215:466e1–8. DOI: 10.1016/j.ajog.2016.05.013. PMID: 27179442.

18. Wang M, Lu S, Li S, Shen F. 2013; Reference intervals of D-dimer during the pregnancy and puerperium period on the STA-R evolution coagulation analyzer. Clin Chim Acta. 425:176–80. DOI: 10.1016/j.cca.2013.08.006. PMID: 23954836.

19. Kovac M, Mikovic Z, Rakicevic L, et al. 2010; The use of D-dimer with new cutoff can be useful in diagnosis of venous thromboembolism in pregnancy. Eur J Obstet Gynecol Reprod Biol. 148:27–30. DOI: 10.1016/j.ejogrb.2009.09.005. PMID: 19804940.

20. Bourjeily G, Paidas M, Khalil H, Rosene-Montella K, Rodger M. 2010; Pulmonary embolism in pregnancy. Lancet. 375:500–12. DOI: 10.1016/S0140-6736(09)60996-X. PMID: 19889451.

21. Ercan Ş, Özkan S, Yücel N, Orçun A. 2015; Establishing reference intervals for D-dimer to trimesters. J Matern Fetal Neonatal Med. 28:983–7. DOI: 10.3109/14767058.2014.940891. PMID: 25060670.

22. Chan WS, Lee A, Spencer FA, et al. 2010; D-dimer testing in pregnant patients: towards determining the next 'level' in the diagnosis of deep vein thrombosis. J Thromb Haemost. 8:1004–11. DOI: 10.1111/j.1538-7836.2010.03783.x.
23. Kabrhel C, Mark Courtney D, Camargo CA Jr, et al. 2009; Potential impact of adjusting the threshold of the quantitative D‐dimer based on pretest probability of acute pulmonary embolism. Acad Emerg Med. 16:325–32. DOI: 10.1111/j.1553-2712.2009.00368.x. PMID: 19298619.

24. Kabrhel C, Van Hylckama Vlieg A, Muzikanski A, et al. 2018; Multicenter evaluation of the YEARS criteria in emergency department patients evaluated for pulmonary embolism. Acad Emerg Med. 25:987–94. DOI: 10.1111/acem.13417. PMID: 29603819.
Fig. 1
The box plot of D-dimer level presented according to the pregnancy trimesters for patients with and without PE.
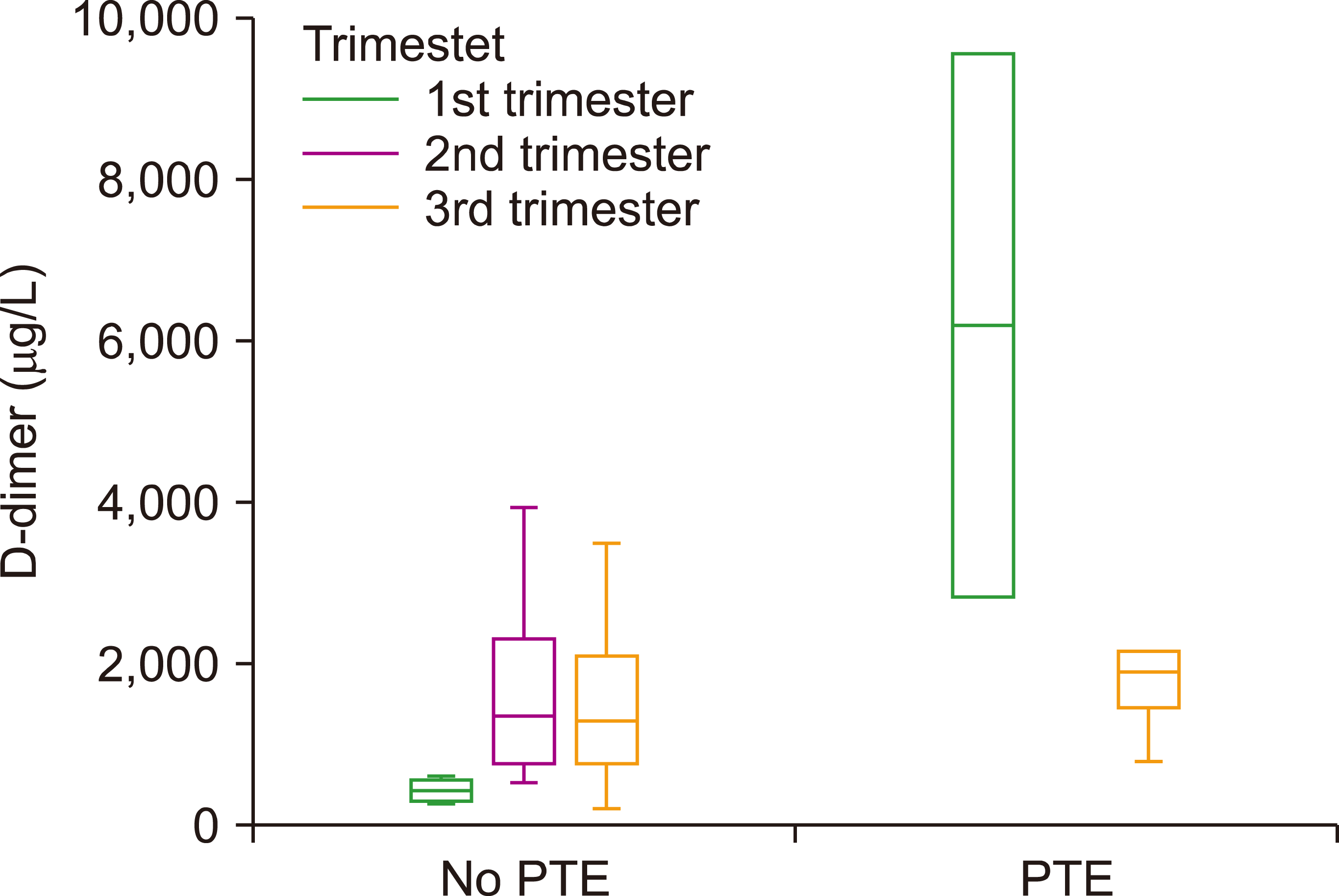
Table 1
Baseline characteristics and the score of Wells criteria.
| Characteristicsa) | Total (N=100) | PE (N=8) | Non-PE (N=92) | P |
|---|---|---|---|---|
| Age, year | 30.38±6.67 | 32.57±7.48 | 30.21±6.62 | 0.369 |
| Gestational age, week | 30.20±7.55 | 25.33±16.02 | 30.58±6.55 | 0.461 |
| 1st trimester | 5 (5%) | 2 (25%) | 3 (3.3%) | 0.015 |
| 2nd trimester | 16 (16%) | 0 (0%) | 16 (17.4%) | |
| 3rd trimester | 79 (79%) | 6 (75%) | 73 (79.3%) | |
| Twin infants | 1 (1%) | 1 (12.5%) | 0 (0%) | 0.080 |
| Preeclampsia | 4 (4%) | 0 (0%) | 4 (4.3%) | 0.547 |
| Hormone therapy | 3 (3%) | 0 (0%) | 3 (3.3%) | 0.604 |
| Fever | 11 (11%) | 3 (37.5%) | 8 (8.7%) | 0.041 |
| Chest pain | 32 (32%) | 2 (25%) | 30 (32.6%) | 0658 |
| D-dimer, mg/Lb) | 1,325.50 (186.0–9,547.0) | 2,110.0 (785–9,547) | 1,274.50 (186–7,977) | 0.029 |
| Wells score | 3.60±0.94 | 4.44±1.97 | 3.53±0.76 | 0.008 |
| PE likely according to Wells criteria | 36 (36%) | 5 (62.5%) | 31 (33.7) | 0.104 |
| The Wells criteria | ||||
| Symptoms of DVTc) | 2 (2%) | 2 (25%) | 0 (0%) | 0.006 |
| PE is the most likely diagnosis | 100 (100%) | 8 (100%) | 92 (100%) | - |
| Tachycardia | 32 (32%) | 3 (37.5%) | 29 (31.5%) | 0.708 |
| Immobilization/surgery | 2 (2%) | 0 (0%) | 2 (2.2%) | 0.674 |
| Previous DVT/PE | 0 (0%) | 0 (0%) | 0 (0%) | - |
| Hemoptysis | 3 (3%) | 1 (12.5%) | 2 (2.2%) | 0.223 |
| Active malignancy | 0 (0%) | 0 (0%) | 0 (0%) | - |
Table 2
Specification of the diagnostic value of the Wells criteria and D-Dimer level for PE detection.
| Variables | Clinical probability of PEa) | Cut-off | Sen. | Spec. | PPV | NPV | AUC | P |
|---|---|---|---|---|---|---|---|---|
| Wells score | Total | 3 | 75.00 | 35.87 | 9.2 | 94.3 | 0.557 | 0.497 |
| D-dimer, mg/L | Total | 1,447 | 87.50 | 63.04 | 17.1 | 98.3 | 0.735 | 0.003 |
| PE unlikely (N=67) | 1,447 | 83.33 | 65.57 | 19.2 | 97.6 | 0.730 | 0.019 | |
| PE likely (N=33) | 1,962 | 99.00 | 78.12 | 22.2 | 99.0 | 0.781 | <0.001 |
Table 3
Specification of the diagnostic value of D-dimer level for PE detection considering the pregnancy trimester.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download