INTRODUCTION
The World Health Organization estimates that 257 million people are affected by chronic HBV infections.
1 If chronic hepatitis B (CHB) is left untreated, approximately 15-40% of patients are likely to develop progressive liver diseases, such as liver cirrhosis or hepatocellular carcinoma, which are the common causes of HBV-related deaths.
2 Therefore, it is vital to treat and monitor patients with CHB infection.
Currently, several nucleos(t)ide analogs (NAs), such as lamivudine, adefovir dipivoxil, entecavir (ETV), telbivudine, and tenofovir disoproxil fumarate (TDF), have been approved for the treatment of chronic HBV infections.
3 All these NAs assist in targeting the inhibition of HBV DNA polymerase, inclusion in viral DNA, and chain termination. Based on the development of novel drugs and the weakness of conventional drugs, the first preferred drugs are constantly changing. ETV and TDF are currently recommended as the first-line treatment options based on the good tolerability, antiviral potency, and a high genetic barrier to resistance.
3-5
Several methods can be used to monitor HBV treatments. The serum HBV DNA quantitative test is the current method used in clinical trials, but while the serum HBV DNA does not reflect intrahepatic HBV cccDNA, a liver biopsy does. On the other hand, a liver biopsy is an invasive test and is unsuitable for monitoring the treatment of CHB patients. Several studies have reported that serum hepatitis B surface antigen (HBsAg) quantification tests, which indirectly reflect the intra-hepatic HBV cccDNA, may be useful.
6-10 Despite this, most studies comparing the effectiveness of drugs were based on serum HBV DNA quantitative tests, and only a few studies were based on the HBsAg quantification tests.
Previous studies analyzed the HBsAg quantification levels in CHB patients treated with ETV. On the other hand, it had a short 24-month follow-up period and only provided insight into the predictive ability of seroconversion in the hepatitis B e antigen (HBeAg) positive patients. This study did not show significant differences in quantification levels.
9 A study of TDF-treated patients found that the HBsAg levels decreased more in HBeAg-positive patients than in negative patients, but the follow-up period was short.
11,12 Given the limitations of the short follow-up of previous studies, it is unclear if the HBsAg quantification levels at each treatment point differed according to the HBeAg status and the treatment drug in CHB patients. This study compared HBsAg quantification changes in CHB patients according to the HBeAg status and treatment drugs.
Go to :

SUBJECTS AND METHODS
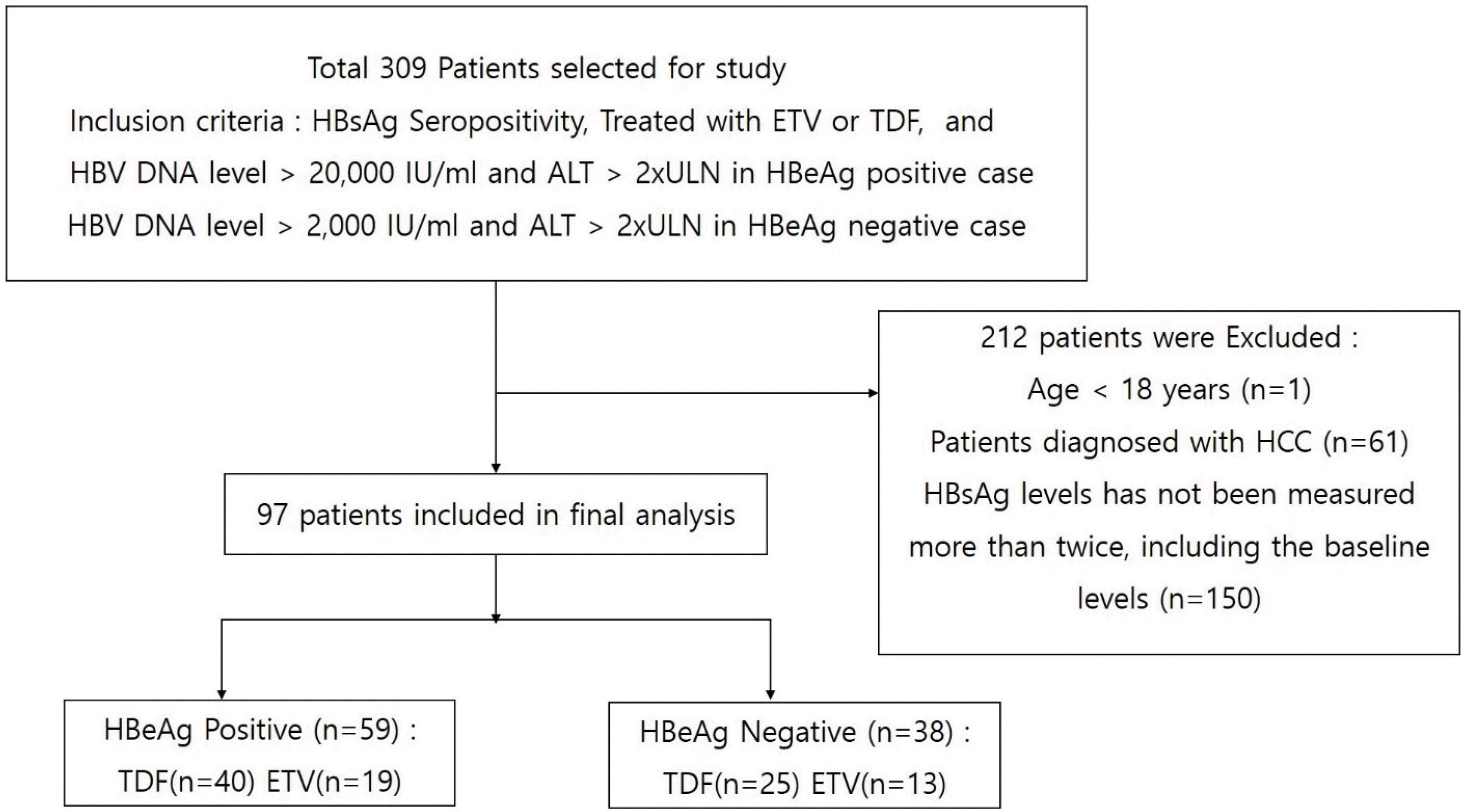
1. Patients
CHB patients who underwent at least 1 year of follow-up treatment with ETV or TDF at Konkuk University Medical Center were included in this retrospective study. Before the treatment, conventional liver and kidney function tests and serological hepatitis B markers were evaluated. The HBeAg, serum HBV DNA levels, serum HBsAg quantification, anti-hepatitis C virus antibody, and imaging tests to diagnose liver cirrhosis and hepatocellular carcinoma (HCC) were performed.
1) Inclusion criteria
(1) Presence of serum HBsAg for at least 6 months
(2) Presence of HBsAg quantification levels at the initiation of treatment
(3) HBV DNA level>20,000 IU/mL and ALT>2×upper limit of normal (ULN) in HBeAg-positive cases
HBV DNA level>2,000 IU/mL and ALT>2×ULN in HBeAg-negative cases
(4) Treatment duration of at least 1 year
(5) Patients treated with ETV or TDF
2) Exclusion criteria
(1) Patients younger than 18 years of age
(2) Patients diagnosed with HCC
(3) HBsAg level has not been measured more than twice
(4) Insufficient clinical data
The medical records of the participants were reviewed, and their HBsAg levels were measured annually for up to 6 years. The final follow-up visit was in April 2021. The data were censored if the patients changed drugs due to side effects, were lost to follow-up, died due to any other cause, or drug resistance was confirmed.
All patients provided written informed consent for the CHB treatment and the use of data. The Institutional Review Board of Konkuk University Medical Center approved this study (IRB No. KUH 1010689).
4. HBsAg levels measurement
HBsAg levels were measured at the baseline and annually after treatment, using a chemiluminescent microparticle immunoassay (Architect HBsAg QT, Abbott Diagnostics, Chicago, IL, USA; measurable range 0.05-250 IU/mL). The HBsAg level was quantified at a 1:500 dilution according to the manufacturer’s instructions. To bring HBsAg levels to within the measurable range, samples with values above and below this range required a lower and higher dilution, respectively.
5. Statistical analysis
The patients’ characteristics are presented as the mean±SD or number (%), as appropriate. A linear mixed model (LMM) was used to compare the HBsAg quantification levels during the follow-up period. Subsequently, an independent samples t-test was used to analyze the differences in the HBsAg quantification levels at each treatment point. Furthermore, the change in HBsAg level was compared using LMM analysis. For all statistical tests, a two-sided p-value <0.05 was considered significant. IBM Statistics SPSS 25 was used for statistical analysis.
Go to :

DISCUSSION
HBsAg loss and seroconversion in CHB patients are the ultimate goals of anti-HBV therapy.
1,3,13 On the other hand, HBsAg loss, and seroconversion are quite rare in CHB patients, and there is also a lack of long-term data on the changes in the HBsAg levels in patients treated with NAs, including TDF or ETV, which are currently used as first-choice drugs.
14
Nevertheless, the utility of the HBsAg quantification test is expanding gradually; it has been used to identify the risk factors for HCC, define the HBV disease phase, monitor patients treated with interferon and NAs, and predict HBsAg loss in the early stages of treatment.
7,15,16
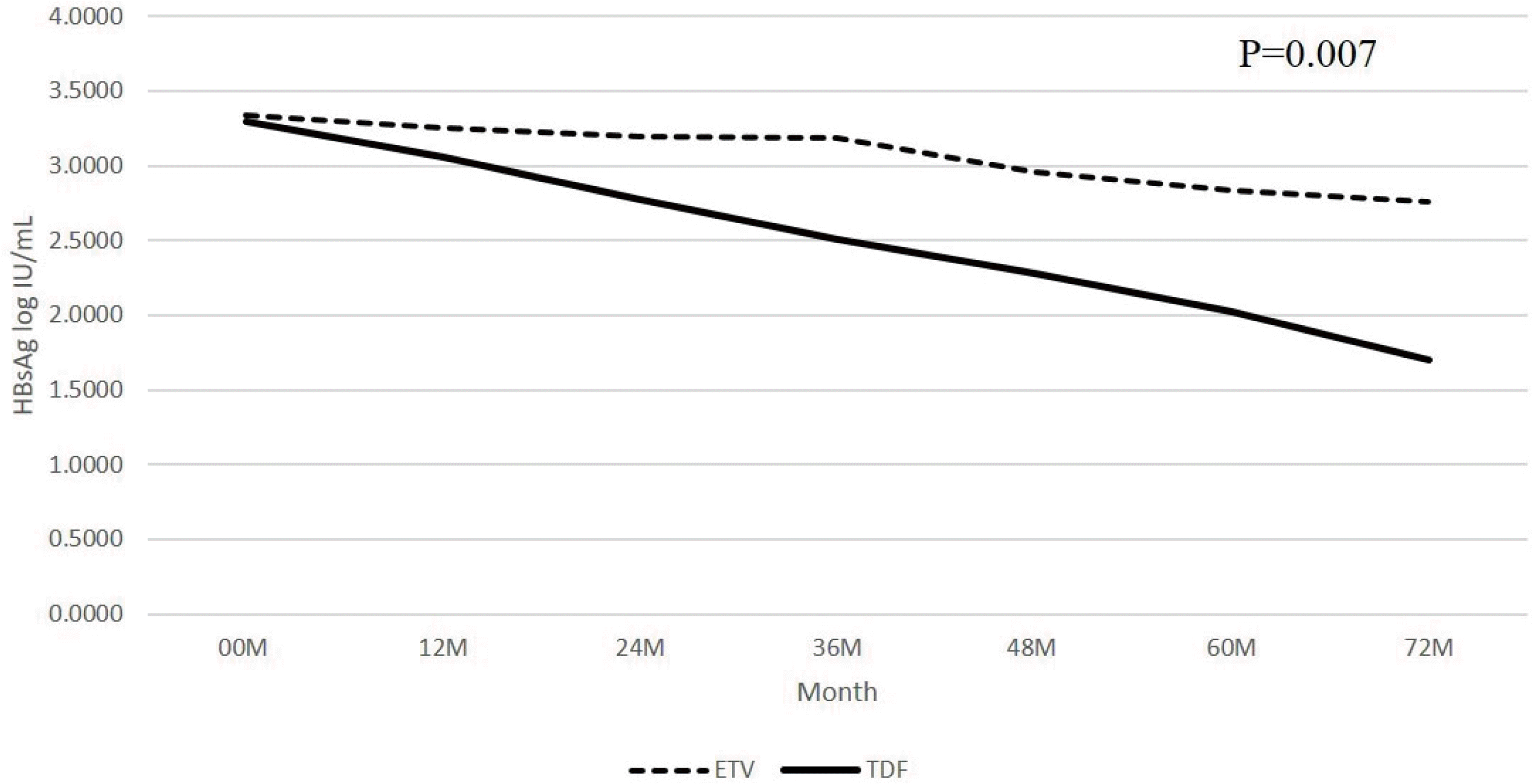
Based on these findings, this study compared the changes in the HBsAg level according to the HBeAg status in CHB patients treated with TDF or ETV over 6 years.
A previous study analyzed the changes in HBsAg levels in 529 CHB patients treated with ETV. Over 49.2 months of ETV treatment, the HBsAg levels decreased at a median annual rate of 0.12 and 0.09 log IU/mL in the HBeAg-positive and
-negative patients, respectively.
17 Unlike previous studies, this study showed that among the CHB patients treated with ETV, the HBeAg-positive and HBeAg-negative patients showed a 0.06 and 0.11 log IU/mL decrease per year, respectively.
Another study conducted for 144 weeks in patients treated with TDF for more than 4 years on patients with CHB who were HBeAg-negative compared the changes between a TDF stop group and a TDF continue group. The median change in the HBsAg level was a -0.59 log IU/mL in patients who discontinued TDF therapy and remained off-therapy, -0.51 log IU/mL in those who restarted TDF therapy, and -0.21 log IU/mL in patients who continued TDF therapy.
18 The present study showed a change of -0.23 log IU/mL per year in HBeAg-negative patients treated with TDF, which is a relatively larger decrease than that in the TDF continuation group in the previous study. The present study period was relatively long (72 months), but the number of patients who participated in the analysis was relatively small. These factors may have caused the difference from the previous research results.
In the case of HBsAg seroconversion in the present study, two patients were identified: one in the HBeAg-positive group and one in the HBeAg-negative group. The annual incidence of HBsAg loss is estimated to be approximately 1%-2% in Asian and Western populations and approximately 0.13% for Asian patients only.
14,19,20 In a meta-analysis of 34 studies that included 42,588 CHB patients, HBsAg seroclearance was reported at approximately 1.02% (95% CI 0.79-1.27).
21 Similarly, the seroclearance of the HBsAg has been studied widely. On the other hand, studies on the seroconversion of HBsAg are rare, and the data vary widely from study to study. One study reported that the seroconversion of HBsAg occurred in approximately 37.3% of patients with HBsAg loss,
22 while another study reported that HBsAg seroconversion occurred in approximately 51.7% of the patients with HBsAg loss.
23 As such, the loss and seroconversion of HBsAg in patients treated with NAs are very rare and were observed to be rare in the 7-year follow-up period of the present study. A previous study predicted the timing of HBsAg loss with an annual HBsAg reduction rate in patients treated with NAs. The median time to HBsAg loss for HBeAg-positive and HBeAg-negative patients was 36 (9.6-98.3) years and 39 (1.3-80.5) years, respectively, after the start of therapy.
24
In the present study, LMM analysis showed that TDF was more effective than ETV in reducing the HBsAg reduction over time. An additional independent samples t-test showed that the difference between these two drug treatment groups was only seen in the HBeAg-negative patient group and significant from the 36 months of treatment (
Tables 2-
4).
Because each patient had a different baseline HBsAg level, additional analysis was performed to compensate for this. The differences in HBsAg levels between the baseline and at each treatment time point were compared using LMM analysis for each drug. Numerically, the TDF treatment group showed a larger decrease in the HBsAg levels than in the ETV treatment group, but there was no significant difference (
Tables 5-
7). This result might be because the baseline HBsAg level of HBeAg-negative patients treated with TDF was relatively lower than the baseline HBsAg level of HBeAg-negative patients treated with ETV, even though there was no significant significance (
Table 4).
Table 7
Mean Difference in HBsAg Log10 IU/mL Level from the Baseline (00 Months) for Each Treatment Time Point According to the HBeAg Status and Drug
|
Months |
Total patients |
HBeAg (+) patients |
HBeAg (-) patients |
|
|
|
|
ETV |
TDF |
Significant |
ETV |
TDF |
Significant |
ETV |
TDF |
Significant |
|
12 |
-0.0844 |
-0.2854 |
0.067 |
-0.0876 |
-0.3128 |
0.194 |
-0.0798 |
-0.2421 |
0.082 |
|
24 |
-0.1787 |
-0.5017 |
0.012a
|
-0.2066 |
-0.5255 |
0.141 |
-0.1415 |
-0.4704 |
0.144 |
|
36 |
-0.1985 |
-0.6763 |
0.001a
|
-0.2527 |
-0.6807 |
0.051 |
-0.1389 |
-0.6705 |
0.037a
|
|
48 |
-0.3363 |
-0.8365 |
0.009a
|
-0.3187 |
-0.8310 |
0.170 |
-0.3539 |
-0.8429 |
0.112 |
|
60 |
-0.4630 |
-0.9837 |
0.046a
|
-0.4476 |
-0.8829 |
0.281 |
-0.4765 |
-1.1550 |
0.050a
|
|
72 |
-0.5418 |
-1.1265 |
0.073 |
-0.3961 |
-0.9037 |
0.324 |
-0.6633 |
-1.4235 |
0.066 |

This study had some limitations. First, there were a small number of patients who participated in the actual study. Moreover, and the follow-up period increased, fewer patients participated in the analysis at the time of treatment. Thus, the statistical confidence may be relatively low. Another limitation is that relatively older patients were involved in the HBeAg-negative patient group regarding baseline characteristics. On the other hand, owing to the natural course of the disease, it is believed that the age of HBeAg-negative patients can be relatively high. In addition, the possibility of selection bias is also a limitation because some of the total patients treated with CHB were confirmed with the HBsAg level.
This study was a long-term retrospective follow-up analysis evaluating the changes in the HBsAg levels in CHB patients receiving NAs in clinical practice. One of the strengths of this study was that it directly compared the effects of reducing the HBsAg level of ETV and TDF in Korean patients treated with CHB for the long term. On the other hand, during the 72-month follow-up observation, the actual number of HBsAg seroconversions was small.
Therefore, a large-scale and long-term study will be needed to determine if the decrease in HBsAg levels leads to HBsAg loss or seroconversion. Nevertheless, this study is meaningful because the efficiency of TDF and ETV on the HBsAg level was compared directly over the long term.
In HBeAg-negative patients, the HBsAg levels were lower after 36 months of treatment in patients treated with TDF than in those treated with ETV. Therefore, TDF may be superior to ETV regarding the HBsAg level reduction in patients with a HBeAg-negative status.
Go to :





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download