Definition of COVID-19 infection, clinical status, and other clinical variables
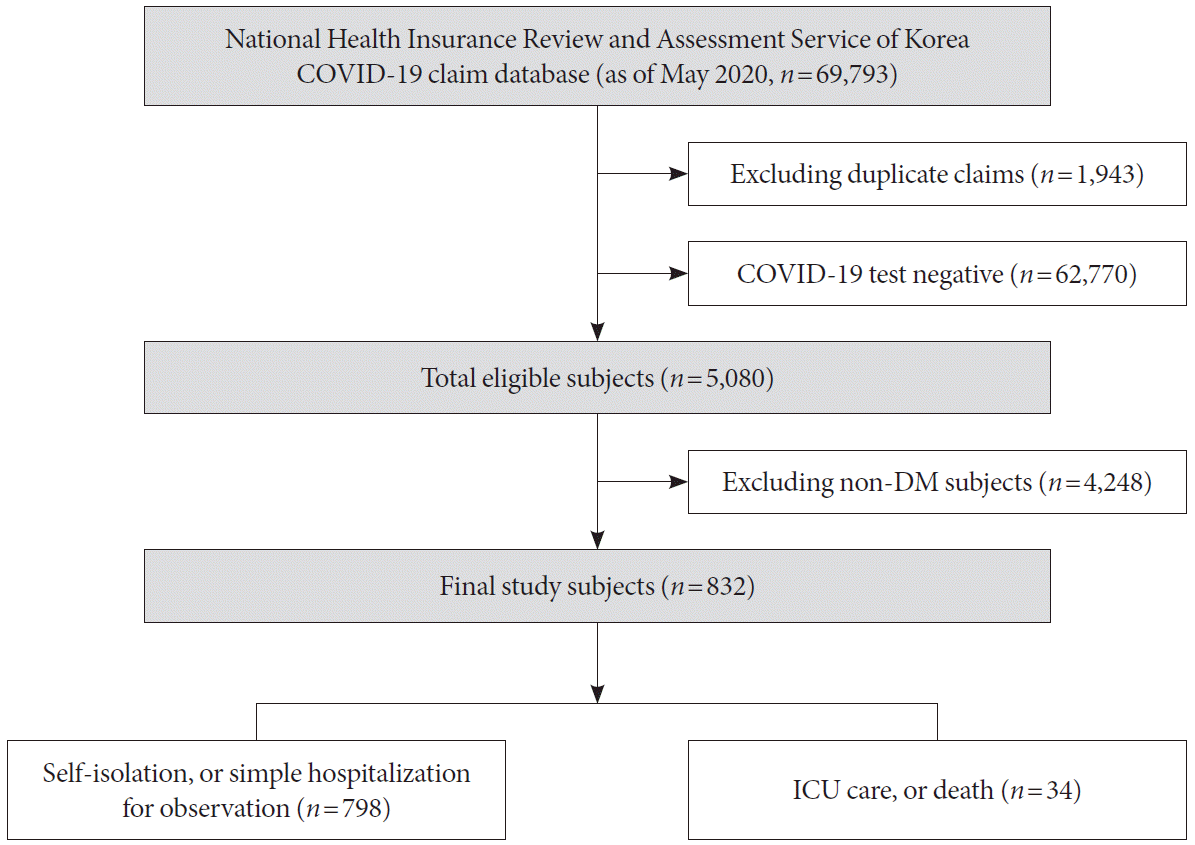
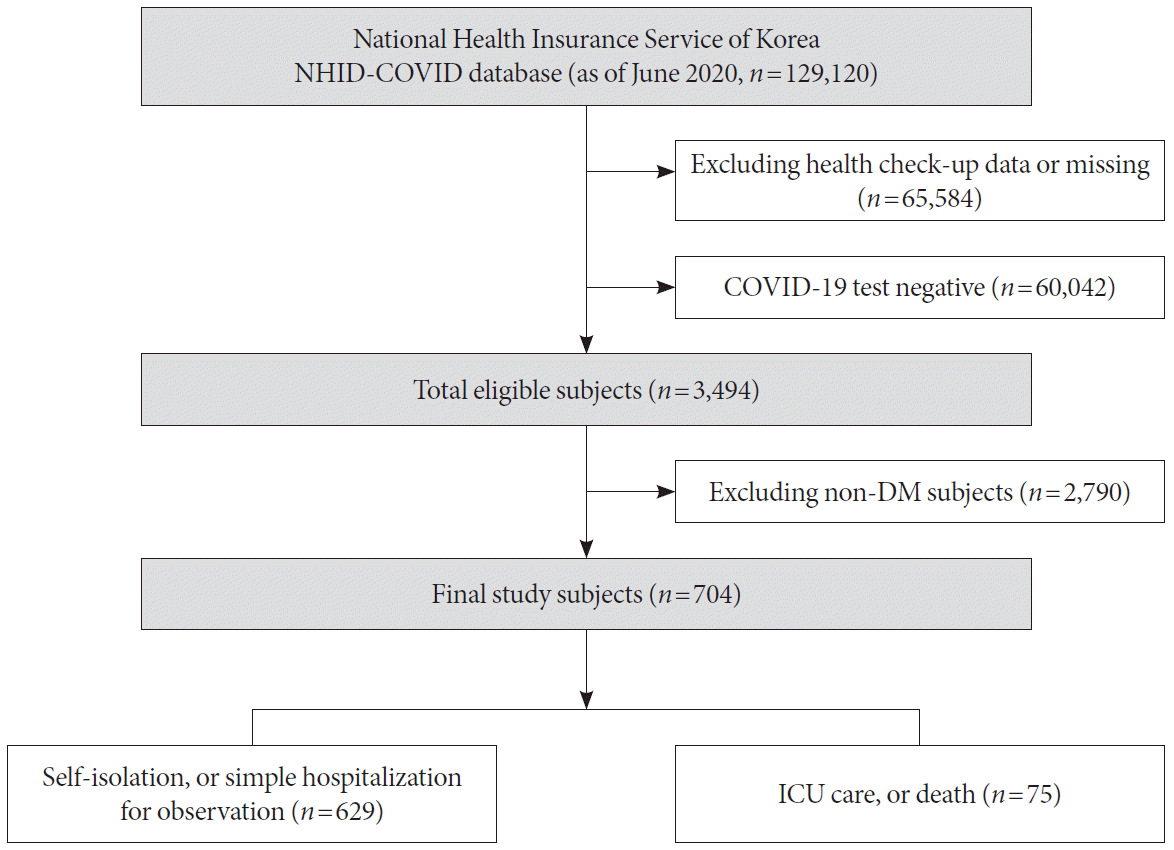
The clinical variables that can be identified in Korea’s COVID-19 claim database are the subject’s age, sex, diagnostic code, medication prescription, outpatient care, hospitalization, critical care, and death. COVID-19 diagnosis was defined as ICD10 diagnosis codes B34.2, B97.2, U18, U18.1, or U07.x. Intensive care was defined as a procedure code for endotracheal intubation or mechanical ventilation, or the charge of an intensive care unit management fee. We analyzed subjects according to a mild case of self-isolation or simple hospitalization, or a severe/lethal case of death or intensive care.
The subject’s comorbidity was defined as a diagnostic code. DM was defined as E10.x, E11.x, E12.x, E13.x, or E14.x, hypertension as I10.x, I11.x, I12.x, I13.x, or I15.x, dyslipidemia as E78.x, cardiovascular disease as I20.x, I21.x, I22.x, I23.x, I24.x, or I25.x, and cerebrovascular disease as I61.x, I62.x, I63.x, or I64.x. Chronic kidney disease was defined as N18.x, asthma as J45.x or J46.x, and chronic obstructive pulmonary disease as J44.x. Cancer was defined as C00.x, C01.x, C02.x, C03.x, C04. x, C05.x, C06.x, C07.x, C08.x, C09.x, C10.x, C11.x, C12.x, C13. x, C14.x, C15.x, C16.x, C17.x, C18.x, C19.x, C20.x, C21.x, C22. x, C23.x, C24.x, C25.x, C26.x, C30.x, C31.x, C32.x, C33.x, C34. x, C37.x, C38.x, C39.x, C40.x, C41.x, C43.x, C44.x, C45.x, C46. x, C47.x, C48.x, C49.x, C50.x, C51.x, C52.x, C53.x, C54.x, C55. x, C56.x, C57.x, C58.x, C60.x, C61.x, C62.x, C63.x, C64.x, C65. x, C66.x, C67.x, C68.x, C69.x, C70.x, C71.x, C72.x, C73.x, C74. x, C75.x, C76.x, C77.x, C78.x, C79.x, C80.x, C81.x, C82.x, C83. x, C84.x, C85.x, C86.x, C88.x, C90.x, C91.x, C92.x, C93.x, C94. x, C95.x, C96.x, and C97.x.
The subject’s medication use was assessed based on the initial date of COVID-19 diagnosis. If the prescription was confirmed within 180 days of diagnosis and was for at least 90 days, the subject was defined as a user of a specific drug. And the use of medication was considered both inpatient and outpatient, and both oral and injectable agents such as insulin were defined in the same method.
Anthropometric assessments of subjects were performed by health care professionals in the NHID-COVID dataset. Height, weight, waist circumference, and blood pressure were assessed. Information on the lifestyle was obtained through standardized self-reported questionnaires. Subjects were classified according to smoking status as nonsmoker, former smoker, or current smoker. Individuals who consumed 30 g of alcohol per day were classified as being heavy alcohol consumers [
22,
23]. Regular physical activity was defined as performance of strenuous exercise for at least once per week [
23].
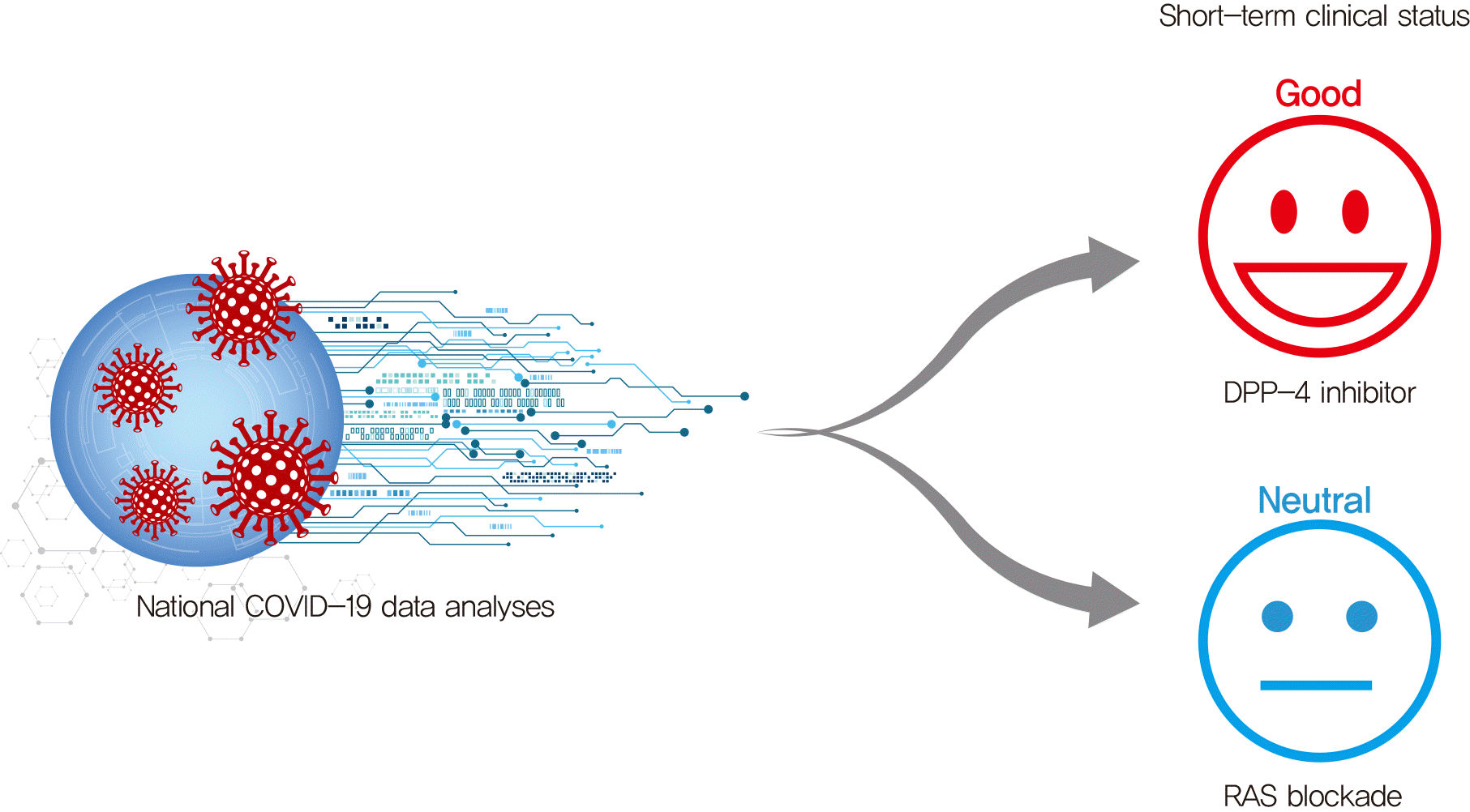
Thereafter, differences in characteristics and clinical status were compared according to whether the subject used DPP-4i and/or RAS blockade. We also investigated the possibility of synergy during the combined use of these two drugs.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download