Abstract
Background
Tacrolimus (TAC) is a main therapy for liver transplantation (LT) patients, but it has side effects such as chronic nephrotoxicity that progressively aggravate renal function. The purpose of this study was to retrospectively compare the renal function between a TAC group and a combination of everolimus and reduced TAC (EVR-TAC) group after deceased donor liver transplantation (DDLT).
Methods
The study comprised 131 patients who underwent DDLT between January 2013 and April 2018 at our institution. They received TAC or EVR-TAC after DDLT. Everolimus (EVR) was introduced between 1 and 6 months after DDLT.
Results
Thirty-six of 131 patients (27.5%) received EVR-TAC. The incidence of chronic kidney disease (CKD; estimated glomerular filtration rate, <60 mL/1.73 m2) in the EVR-TAC group was higher than in the TAC group (25% vs. 8.4%; P=0.019). Increasing serum creatinine (n=23, 63.9%) was the most common cause for adding EVR to treatment of the posttransplant patients. There were no statistical differences in acute rejection and CKD between the two groups. The TAC trough level was significantly lower in the EVR-TAC group than in the TAC group, and the renal function of the EVR-TAC group was worse than that of the TAC group until 1 year after DDLT. However, the renal function of the EVR-TAC group improved and became similar to that of TAC group at 3 years posttransplant.
Go to : 
Go to : 
The goal of immunosuppression (IS) in liver transplantation (LT) is to maintain graft function and to balance low acute rejection and IS drug-related adverse events. Tacrolimus (TAC) has been the most common therapy to prevent acute rejection after LT, but dose-related side effects were associated with acute and chronic renal dysfunction, neurotoxicity, increased malignancies, cardiovascular disease, and metabolic disorders [1].
Improved surgical techniques and advanced perioperative care have contributed to improved patient survival, which has led in turn to increased prevalence of late complications after LT such as chronic kidney disease (CKD). The cumulative incidence of chronic renal failure was about 20% at 3 years after transplantation and this was associated with a 4-fold increase in the risk of death [2]. One major contributor to renal dysfunction in LT recipients was calcineurin inhibitors (CNIs) [1,3].
Everolimus (EVR; Certican, Novartis, Basel, Switzerland) with a reduced-dose of TAC was found to have sufficient efficacy regarding rejection episodes and graft failure [4-7]. Additionally, the use of EVR facilitated safe reduction or withdrawal of CNIs to preserve renal function over the long-term after transplantation [3-8]. Since EVR coverage was introduced to Korean social health insurance in early 2016, the use of EVR in Korea has resulted in notable changes to IS regimens [9]. Korean social health insurance policy forcefully suggests administration of TAC and EVR concurrently.
In our center, we have used different immunosuppressive regimens, including a TAC-based regimen and EVR-TAC combination. This study compared efficacy and renal function in liver recipients who received EVR between 1 and 6 months after deceased donor liver transplantation (DDLT) and in those who did not use EVR.
Go to : 
This retrospective observational study was approved by the Samsung Medical Center Institutional Review Board (IRB No. SMC-2020-05-050). The consent of the participants was exempted by the IRB. The IRB waived the requirement for patient consent because this was a retrospective study of patient medical records. The accessed patient data were maintained with confidentiality in the present study.
We reviewed a prospectively collected database of the records of all adults who underwent DDLT between January 2013 and April 2018 at our institution; during that time, 278 adult DDLTs were performed at our institution.
Patients were excluded according to the following criteria: living donor LT, retransplantation, use of cyclosporin and/or antimetabolite monotherapy, discontinuation of EVR, history of continuous renal replacement therapy on the waiting list, history of proteinuria or hyperlipidemia, synchronous multiple organ transplants, pediatric LT (age <18 years), receiving a split graft, receiving a liver graft after donor cardiac death, and incomplete medical records. Patients who died from transplantation until May 2020 or received hemodialysis or kidney transplantation because of renal failure until May 2020, were also excluded. One hundred thirty-one patients were included in our study.
Patients underwent DDLT via a piggyback technique and side-to-side inferior vena cava reconstruction. IS strategy in our center was as described previously [10]. Steroids were used only up to 3 months after DDLT. In the combination of everolimus and reduced tacrolimus treatment (EVR-TAC) group, TAC was administered on the first 5 postoperative days, and EVR was administered from one to 6 months of the posttransplant period, with a target level of 4–8 ng/mL of TAC. Patients taking EVR did not received mycophenolate mofetil (MMF). In the TAC group, TAC was administered twice a day, starting from the fifth postoperative day, with a target level of 6–10 ng/mL. In the latter group, the combination with MMF was introduced in cases of renal failure to reduce the target levels to 4–8 ng/mL.
The endpoint of the study was the incidence of acute rejection and chronic renal failure. The EVR-TAC group was composed of recipients who were treated with EVR between 1 month and 6 months after DDLT. Use of EVR was chosen in recipients with renal dysfunction greater than 2.0 mg/dL, treated by one surgeon. Other surgeons did not use EVR in any recipients.
Renal function was evaluated by estimated glomerular filtration rate (eGFR) and creatinine level at 1, 3, 6, 12, 24, and 36 months. Graft rejection was diagnosed by liver biopsy according to the Banff criteria [11]. CKD was defined eGFR <60 mL/1.73 m2. The overall survival rate was calculated from the date of transplantation to the date of the last follow-up visit or until graft loss (defined as need for relisting or patient death).
Continuous variables were analyzed using the Mann-Whitney U-test, with results expressed as means±standard deviations or medians and ranges. Categorical variables were expressed as numbers and percentages of patients. Chi-square tests or Fisher’s exact tests were conducted to evaluate differences in frequencies of categorical variables between groups. Cumulative risk rates were estimated using the Kaplan-Meier method, and survival curves were compared with the log-rank test. A mixed model was used in repeated measures of eGFR, serum creatinine, and TAC trough level. All tests were two-sided, and statistical significance was defined as P<0.05. Analyses were carried out using the IBM SPSS ver. 24 (IBM Corp., Armonk, NY, USA).
Go to : 
Ninety-five patients (72.5%) received TAC-based IS and 36 patients (27.5%) received EVR-TAC. There were no statistically significant differences in age, body mass index, hypertension, diabetes, etiology for DDLT, coexistence of hepatocellular carcinoma (HCC), model for end-stage liver disease (MELD) score, Child-Pugh class, eGFR, or serum creatinine level between the two groups (Table 1). However, the incidence of CKD in the EVR-TAC group was higher than that in the TAC group (25% vs. 8.4%; P=0.019).
Baseline recipient characteristics
The EVR-TAC group showed high proportion of CKD and high serum creatinine level before LT compared with the TAC group. Accordingly, increasing serum creatinine (n=23, 63.9%) was the most common reason for adding EVR treatment. Other causes were increased liver function (n=6, 16.7%), de novo malignancy (n=1, 2.8%), adverse effects of MMF (n=2, 5.6%), and adverse effects of TAC (n=4, 11.1%). The median time from DDLT to add EVR treatment was 3.3 months (range, 1.1–6.0 months).
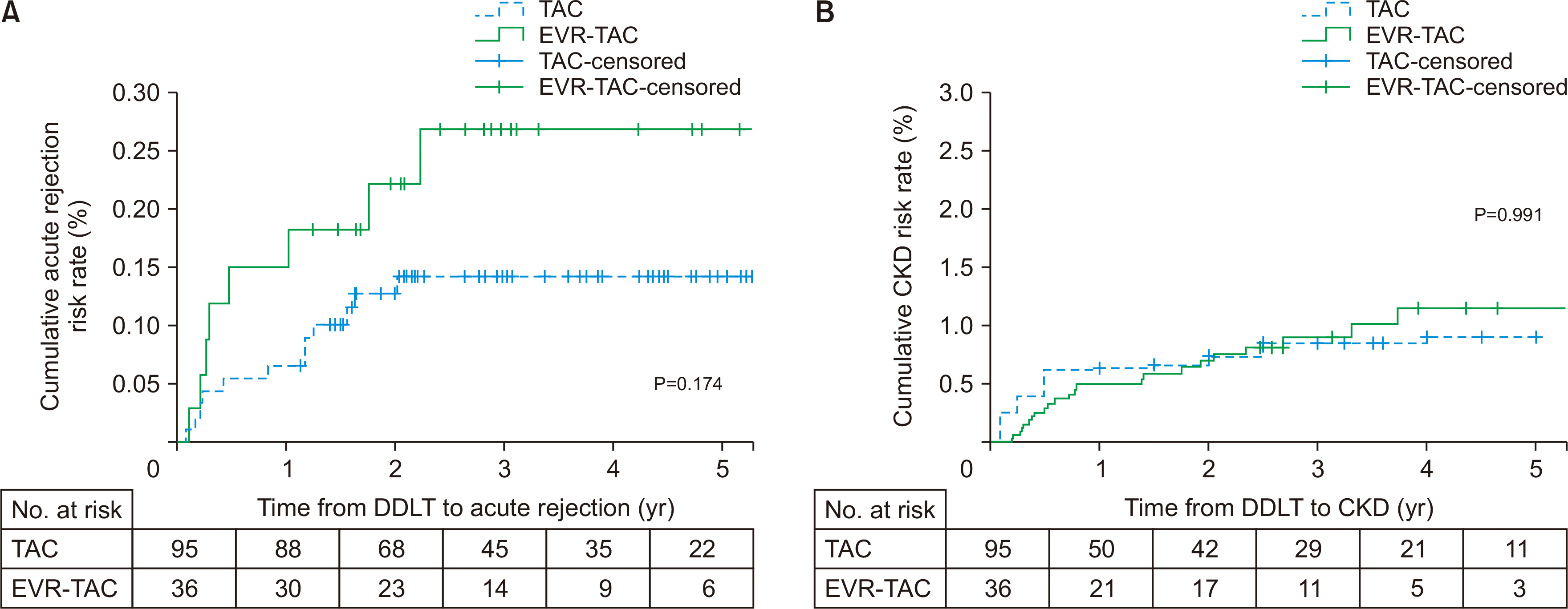
The median follow-up duration was 46.6 months (range, 36.0–77.8 months) in the TAC group and 46.2 months (range, 36.0–79.2 months) in the EVR-TAC group. The incidence of acute rejection was 12.6% (n=12) in the TAC group and 22.2% (n=8) in the EVR-TAC group. The cumulative acute rejection risk rate in the EVR-TAC group was higher than that in the TAC group (P=0.174) (Fig. 1A). However, there was no statistical difference in acute rejection between the two groups. The second and third acute cellular rejection (ACR) occurred in the EVR-TAC group. However, there were no more than two ACRs in the TAC group. A total of six patients stopped using EVR. Two patients did not want to take EVR. In addition, EVR discontinued due to hyperlipidemia (n=2) and infection (n=2).
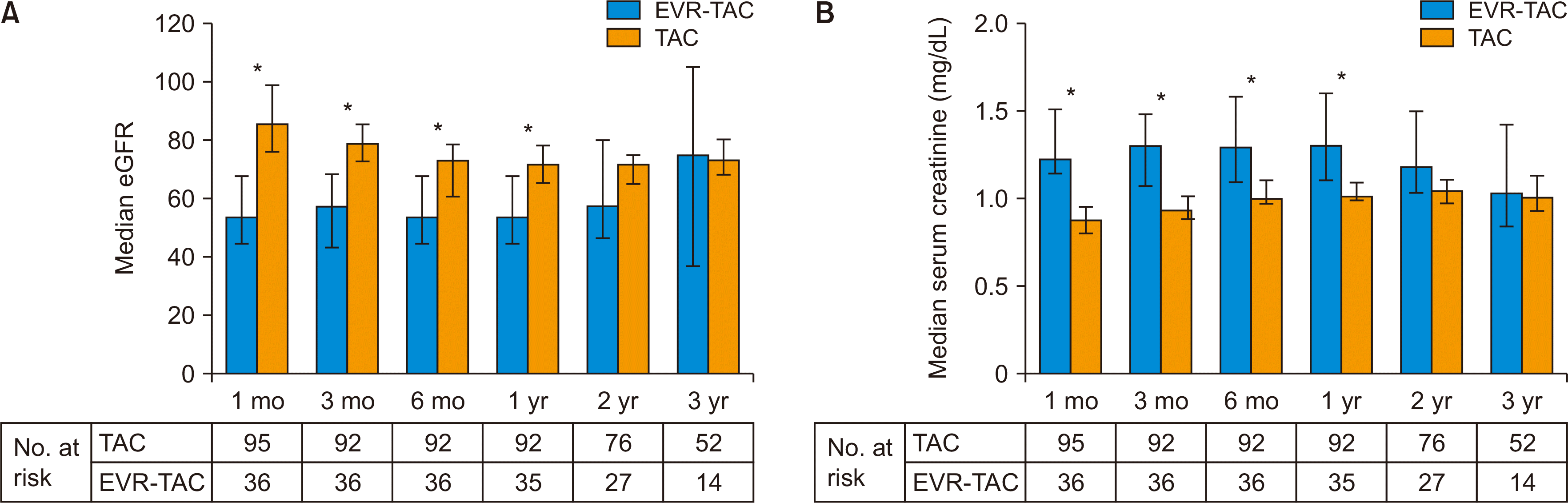
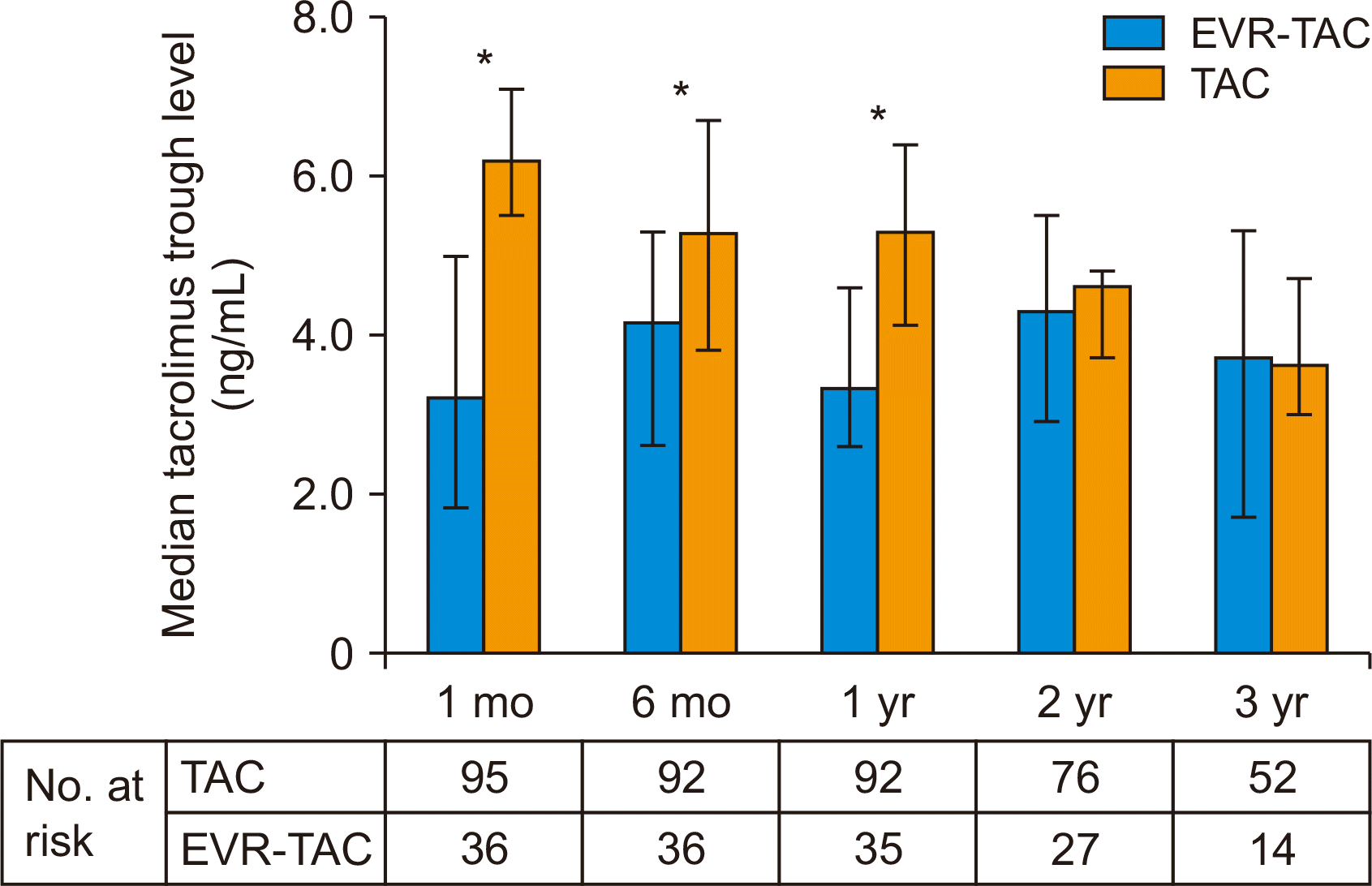
The incidence of CKD was 56.8% (n=54) in the TAC group and 75.0% (n=27) in the EVR-TAC group (P=0.070) and there was no statistically significant difference in cumulative CKD rate between the two groups (Fig. 1B). The serially measured median TAC trough level in the TAC group was higher than that in the EVR-TAC group within 1 year after DDLT, but the median TAC trough level after 2 years in the TAC group did not differ from that in the EVR-TAC group (Fig. 2). Serially measured median eGFR within 1 year of DDLT in the TAC group was significantly higher than that in the EVR-TAC group, and the median serum creatinine level within 1 year in the TAC group was significantly lower than that in the EVR-TAC group. The median eGFR and serum creatinine level at 3 years in the TAC group were similar to those in the EVR-TAC group (Fig. 3). The proteinuria caused by EVR was not severe and was observed. The incidence of cytomegalovirus (CMV) infection in the TAC group did not differ from that of the EVR-TAC group (63.2% vs. 61.1%; P=0.842). The incidences of proteinuria and hyperlipidemia in the EVR-TAC group were higher than those in the TAC group (19.4% vs. 2.1%, P=0.002 and 30.6% vs. 11.6%, P=0.017, respectively).
Go to : 
Acute kidney injury after LT is associated with poor long-term outcomes, including worse survival and higher incidence of CKD [12]. Chronic renal failure occurs in LT recipients at a rate of 8% after 1 year and 18% by 5 years. When eGFR is below 30 mL/min/1.73 m2, the risk of death increases dramatically [13]. Strategies of CNI reduction and withdrawal have been introduced in clinical practice in view of preventing CNIs-related toxicities. EVR-based regimens have been introduced to prevent CNIs-related toxicities and improve long-term renal function after LT [8,14].
In our study, the most frequent indication for introducing EVR in our population was renal dysfunction. The main endpoint was the change in renal function after introduction of EVR. We observed stable renal function, as assessed by eGFR and serum creatinine, in this population, which was followed for a minimum of 36 months. The EVR-TAC group had more CKD patients before DDLT than the TAC group. Thus, the median serum creatinine level in the EVR-TAC group was higher and the median eGFR in the EVR-TAC group was lower than those in the TAC group after DDLT. However, kidney function gradually recovered in patients who added EVR treatment between 1 and 6 months after DDLT; thus, median creatinine level and median eGFR were similar to those of the TAC group at 3 years after DDLT.
There is a consensus that TAC is the main immunosuppressant after LT, but the blood standard target dose is not well-defined [15]. Patients with early mean TAC level of 7–10 ng/mL after LT showed similar ACR rates and better graft survival compared to patients with TAC level greater than 10 ng/mL [16]. Thus, a low-dosage TAC level below 6 ng/mL could be considered sufficient to prevent most ACR [17], but median TAC levels was 3.6–6.2 ng/mL in the TAC group within 3 years posttransplant and 3.2–4.3 ng/mL in the EVR-TAC group. Our study showed that the acute rejection rate of the EVR-TAC group did not differ from that in the TAC group. Additional drugs such as EVR or MMF may be used to increase immune potency.
The timing of the introduction of EVR seems to play a decisive role in the balance between graft protection, renal protection, and adverse effects. EVR introduction with reduced exposure to TAC after 1 month posttransplant provides a significant benefit to renal function after 2 years of follow-up [4]. A meta-analysis reported that EVR combined with reduced TAC significantly improved creatinine clearance at 1 year, and decreased biopsy-proven acute rejection [18]. In our center, EVR is currently introduced at 1 to 6 months post-LT in combination with TAC. The median TAC trough levels from 1 month to 1 year in the EVR-TAC group were lower than those of the TAC group or the recommended TAC trough level. Experimental studies suggested that EVR can significantly improve glomerular hypertrophy in diabetic mice, and that EVR improves renal function [18].
EVR allows a dose reduction of CNIs in LT. Recently, several reports have shown that de novo use of EVR reduces the incidence of renal impairment 1–2 years post-LT compared to CNIs therapy [3,4]. Many studies have demonstrated the efficacy of EVR-based regimens in renal protection compared to CNIs therapy [3,4,19]. The randomized H2304 study demonstrated that EVR with reduced TAC was associated with significantly better renal function and comparable efficacy over the first 3 years after LT compared with standard TAC therapy [4,5,20]. However, several studies have demonstrated that late conversion to EVR after LT, once CNIs-related kidney damage has already occurred, is not effective in restoring renal function [19,21-23]. Perhaps, long exposure to CNIs contributes to interstitial fibrosis and glomerular obliteration due to prolonged vasoconstriction of renal microcirculation [24].
EVR has several adverse events. EVR increases the risk of leukopenia likely due to inhibition of cell proliferation by blocking the cells from the G1 to S phase [18]. EVR also increases the risk of bacterial infection [6,19], perhaps due to deterioration of immune function associated with leukopenia. However, our study did not demonstrate an increase in CMV infection in the EVR-TAC group compared with the TAC group. In addition, EVR increases low-density lipoprotein cholesterol or proteinuria. The possible mechanism is that EVR inhibits secretion of vascular endothelial growth factor and blocks its signaling pathway [6,19]. In addition, the peripheral edema caused by EVR may be explained by decreased plasma osmolality because of proteinuria, which causes interstitial fluid reflux disorder and edema.
This study has several limitations. First, it was a small single-center retrospective study without protocol liver biopsies. Second, we did not separately present the profiles of patients after HCC recurrence or development of de novo malignancies alive at the time of data collection, as these data will be presented in future reports of ongoing studies.
In conclusion, DDLT recipients treated with EVR-TAC maintained stable renal function, whereas kidney function worsened in the TAC group. The present study suggests that EVR should be early introduced as soon as possible after DDLT to reduce exposure to high doses of TAC to preserve renal function. It may therefore be considered a valid option after DDLT in selected recipients at high risk for renal impairment or TAC-related comorbidities.
Go to : 
ACKNOWLEDGMENTS
Conflict of Interest
Jong Man Kim is an editorial board member of the journal but did not involve in the peer reviewer selection, evaluation, or decision process of this article. No other potential conflicts of interest relevant to this article were reported.
Author Contributions
Conceptualization: JMK. Data curation: SL, JMK, SK, JR, GSC. Formal analysis: SL, JMK. Investigation: JMK. Methodology: SL, JMK. Project administration: JWJ. Visualization: JMK. Writing–original draft: SL, JMK. Writing–review & editing: JMK.
Go to : 
REFERENCES
1. Brunet M, van Gelder T, Åsberg A, Haufroid V, Hesselink DA, Langman L, et al. 2019; Therapeutic drug monitoring of tacrolimus-personalized therapy: second consensus report. Ther Drug Monit. 41:261–307. DOI: 10.1097/FTD.0000000000000640. PMID: 31045868.


2. Ojo AO, Held PJ, Port FK, Wolfe RA, Leichtman AB, Young EW, et al. 2003; Chronic renal failure after transplantation of a nonrenal organ. N Engl J Med. 349:931–40. DOI: 10.1056/NEJMoa021744. PMID: 12954741.


3. Saliba F, Fischer L, de Simone P, Bernhardt P, Bader G, Fung J. 2018; Association between renal dysfunction and major adverse cardiac events after liver transplantation: evidence from an international randomized trial of everolimus-based immunosuppression. Ann Transplant. 23:751–7. DOI: 10.12659/AOT.911030. PMID: 30361470. PMCID: PMC6248043.



4. Saliba F, De Simone P, Nevens F, De Carlis L, Metselaar HJ, Beckebaum S, et al. 2013; Renal function at two years in liver transplant patients receiving everolimus: results of a randomized, multicenter study. Am J Transplant. 13:1734–45. DOI: 10.1111/ajt.12280. PMID: 23714399.


5. Fischer L, Saliba F, Kaiser GM, De Carlis L, Metselaar HJ, De Simone P, et al. 2015; Three-year outcomes in de novo liver transplant patients receiving everolimus with reduced tacrolimus: follow-up results from a randomized, multicenter study. Transplantation. 99:1455–62. DOI: 10.1097/TP.0000000000000555. PMID: 26151607.

6. Saliba F. 2017; Everolimus-based immunosuppression in liver transplant recipients. Am J Transplant. 17:2489. DOI: 10.1111/ajt.14388. PMID: 28654719.


7. Guan TW, Lin YJ, Ou MY, Chen KB. 2019; Efficacy and safety of everolimus treatment on liver transplant recipients: a meta-analysis. Eur J Clin Invest. 49:e13179. DOI: 10.1111/eci.13179. PMID: 31610022.

8. Saliba F, Duvoux C, Gugenheim J, Kamar N, Dharancy S, Salamé E, et al. 2017; Efficacy and safety of everolimus and mycophenolic acid with early tacrolimus withdrawal after liver transplantation: a multicenter randomized trial. Am J Transplant. 17:1843–52. DOI: 10.1111/ajt.14212. PMID: 28133906.


9. Kang SH, Hwang S, Ha TY, Song GW, Jung DH, Ahn CS, et al. 2019; Cross-sectional analysis of immunosuppressive regimens focused on everolimus after liver transplantation in a Korean high-volume transplantation center. Korean J Transplant. 33:98–105. DOI: 10.4285/jkstn.2019.33.4.98.

10. Kim JM, Lee KW, Song GW, Jung BH, Lee HW, Yi NJ, et al. 2016; Immunosuppression status of liver transplant recipients with hepatitis C affects biopsy-proven acute rejection. Clin Mol Hepatol. 22:366–71. DOI: 10.3350/cmh.2016.0022. PMID: 27729628. PMCID: PMC5066384.

11. Demetris AJ, Adeyi O, Bellamy CO, Clouston A, Charlotte F, et al. Banff Working Group. 2006; Liver biopsy interpretation for causes of late liver allograft dysfunction. Hepatology. 44:489–501. DOI: 10.1002/hep.21280. PMID: 16871565.

12. Trinh E, Alam A, Tchervenkov J, Cantarovich M. 2017; Impact of acute kidney injury following liver transplantation on long-term outcomes. Clin Transplant. 31:e12863. DOI: 10.1111/ctr.12863. PMID: 27801506.

13. Allen AM, Kim WR, Therneau TM, Larson JJ, Heimbach JK, Rule AD. 2014; Chronic kidney disease and associated mortality after liver transplantation: a time-dependent analysis using measured glomerular filtration rate. J Hepatol. 61:286–92. DOI: 10.1016/j.jhep.2014.03.034. PMID: 24713190. PMCID: PMC4160310.


14. De Simone P, Saliba F, Dong G, Escrig C, Fischer L. 2016; Do patient characteristics influence efficacy and renal outcomes in liver transplant patients receiving everolimus? Clin Transplant. 30:279–88. DOI: 10.1111/ctr.12687. PMID: 26717035.


15. Rodríguez-Perálvarez M, Germani G, Darius T, Lerut J, Tsochatzis E, Burroughs AK. 2012; Tacrolimus trough levels, rejection and renal impairment in liver transplantation: a systematic review and meta-analysis. Am J Transplant. 12:2797–814. DOI: 10.1111/j.1600-6143.2012.04140.x. PMID: 22703529.


16. Rodríguez-Perálvarez M, Germani G, Papastergiou V, Tsochatzis E, Thalassinos E, Luong TV, et al. 2013; Early tacrolimus exposure after liver transplantation: relationship with moderate/severe acute rejection and long-term outcome. J Hepatol. 58:262–70. DOI: 10.1016/j.jhep.2012.09.019. PMID: 23023010.

17. Lerut J, Mathys J, Verbaandert C, Talpe S, Ciccarelli O, Lemaire J, et al. 2008; Tacrolimus monotherapy in liver transplantation: one-year results of a prospective, randomized, double-blind, placebo-controlled study. Ann Surg. 248:956–67. DOI: 10.1097/SLA.0b013e31819009c9. PMID: 19092340.

18. Tang CY, Shen A, Wei XF, Li QD, Liu R, Deng HJ, et al. 2015; Everolimus in de novo liver transplant recipients: a systematic review. Hepatobiliary Pancreat Dis Int. 14:461–9. DOI: 10.1016/S1499-3872(15)60419-2. PMID: 26459721.


19. Yee ML, Tan HH. 2017; Use of everolimus in liver transplantation. World J Hepatol. 9:990–1000. DOI: 10.4254/wjh.v9.i23.990. PMID: 28878864. PMCID: PMC5569278.



20. De Simone P, Nevens F, De Carlis L, Metselaar HJ, Beckebaum S, Saliba F, et al. 2012; Everolimus with reduced tacrolimus improves renal function in de novo liver transplant recipients: a randomized controlled trial. Am J Transplant. 12:3008–20. DOI: 10.1111/j.1600-6143.2012.04212.x. PMID: 22882750. PMCID: PMC3533764.


21. De Simone P, Metselaar HJ, Fischer L, Dumortier J, Boudjema K, Hardwigsen J, et al. 2009; Conversion from a calcineurin inhibitor to everolimus therapy in maintenance liver transplant recipients: a prospective, randomized, multicenter trial. Liver Transpl. 15:1262–9. DOI: 10.1002/lt.21827. PMID: 19790150.


22. Saliba F, Duvoux C, Dharancy S, Dumortier J, Calmus Y, Gugenheim J, et al. 2019; Early switch from tacrolimus to everolimus after liver transplantation: outcomes at 2 years. Liver Transpl. 25:1822–32. DOI: 10.1002/lt.25664. PMID: 31631501. PMCID: PMC7383505.



23. Saliba F, Dharancy S, Lorho R, Conti F, Radenne S, Neau-Cransac M, et al. 2011; Conversion to everolimus in maintenance liver transplant patients: a multicenter, retrospective analysis. Liver Transpl. 17:905–13. DOI: 10.1002/lt.22292. PMID: 21384525.


24. Holdaas H, Rostaing L, Serón D, Cole E, Chapman J, Fellstrøm B, et al. 2011; Conversion of long-term kidney transplant recipients from calcineurin inhibitor therapy to everolimus: a randomized, multicenter, 24-month study. Transplantation. 92:410–8. DOI: 10.1097/TP.0b013e318224c12d. PMID: 21697773.


Go to : 




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download