Abstract
Backgrounds/Aims
Post-hepatectomy liver failure (PHLF) is a major concern for patients with hepatocellular carcinoma (HCC) who have undergone liver resection. The albumin-bilirubin (ALBI) score is a novel model for assessing liver function. We aimed to investigate the effectiveness of the ALBI score as a predictor of PHLF in HCC patients who have undergone hepatectomy in South Korea.
Methods
Between January 2014 and November 2018, HCC patients who underwent hepatectomy and indocyanine retention rate at 15 min (ICG-R15) test were enrolled in this study.
Results
A total of 101 patients diagnosed with HCC underwent hepatectomy. Thirty-two patients (31.7%) experienced PHLF. The ALBI score (OR 2.83; 95% CI 1.22-6.55; p=0.015), ICG-R15 (OR 1.07; 95% CI 1.02-1.12; p=0.007) and ALBI grade (OR 2,86; 95% CI 1.08-7.58; p=0.035) were identified as independent predictors of PHLF by multivariable analysis. The area under the receiver operating characteristic curve of the ALBI score and ICG-R15 were 0.676 (95% CI 0.566-0.785) and 0.632 (95% CI 0.513-0.752), respectively. The optimal cutoff value of the ALBI score in predicting PHLF was -2.62, with a sensitivity of 75.0% and a specificity of 56.5%.
Approximately 800,000 people worldwide are diagnosed with hepatocellular carcinoma (HCC) annually, at an incidence rate of 4.7%. With a mortality rate of 8.2%, HCC was the fourth leading cause of cancer death in 2018.1 Surgical resection is the most effective treatment for patients at stage 0 or A with HCC, as per the Barcelona Clinic Liver Cancer classification.2-4 However, most HCC patients have chronic liver disease, including alcoholic liver disease and hepatitis B or C, which may lead to liver fibrosis and cirrhosis.5 Patients with underlying liver fibrosis and cirrhosis have a high risk of post-hepatectomy liver failure (PHLF).6-8 Despite improvements in surgical techniques and perioperative management, PHLF remains the main cause of postoperative mortality in up to 30% of cases.9-12 In a retrospective study of 488 patients diagnosed with HCC who underwent hepatectomy, overall survival and disease-free survival were significantly shorter in the PHLF group than in the non-PHLF group.13 Therefore, accurate preoperative assessment of liver function and prediction of PHLF are essential for improving the prognosis of HCC patients.14,15
Child-Pugh (CP) grade is the most widely used grading system for evaluating background liver status and determining the surgical management approach. However, CP grade is limited in accurately assessing liver function owing to the inclusion of subjective values such as grading of ascites and encephalopathy.16-18 An indocyanine green retention rate at 15 min (ICG-R15) test is generally performed in Asia and several European institutions prior to hepatectomy because it accurately represents the background liver status.16,19 However, the ICG-R15 test also has several limitations in providing accurate results, such as the need for bed rest before the test, long processing time, and multiple blood sampling.20
The albumin-bilirubin (ALBI) score was first found to predict the overall survival of HCC patients after hepatectomy in 2015 in Japan.20 It can be measured conveniently with a single blood sample and objectively by eliminating subjective parameters because it is based on serum albumin and bilirubin levels. The ALBI score has already been identified as an independent prognostic factor of primary biliary cirrhosis21 and as a predictor of overall survival after liver transplantation.22,23 In addition, recent studies have shown that the ALBI score could be a reliable predictor of PHLF.24-30 However, no study has been conducted to examine the role of the ALBI score in predicting PHLF in Koreans. In this study, we evaluated the effectiveness of the ALBI score in predicting PHLF among HCC patients who underwent hepatectomy in South Korea.
We reviewed the medical records of 146 HCC patients who underwent hepatectomy and the ICG-R15 test at Kosin University Gospel Hospital from January 2014 to November 2018. The exclusion criteria were as follows: 1) patients who received other treatments for HCC prior to hepatectomy, such as transcatheter arterial chemoembolization, 2) history of liver transplantation, 3) coexisting malignancies, and 4) insufficient medical records. Therefore, 101 patients were retrospectively enrolled in this study (Fig. 1). The requirement for written informed consent was waived for this retrospective research. This research was approved by the Ethics Committee of Kosin University Gospel Hospital (IRB No. 2020-06-003).
HCC and liver cirrhosis were diagnosed by CT and MRI prior to hepatectomy and confirmed by histopathological examination of surgical samples. The stage of HCC was classified according to the Barcelona Clinic Liver Cancer classification. PHLF was defined as serum total bilirubin >50 μmol/L (2.9 mg/dL) and prothrombin time index <50% (INR >1.7) on or after postoperative day 5, which is an available definition suggested by International Study Group of Liver Surgery (ISGLS).10,31 No adjustment of clinical status was required for grade A PHLF patients; noninvasive treatments involving the use of albumin, diuretics, and fresh-frozen plasma were provided to grade B PHLF patients. Invasive interventions such as hemodialysis, intubation and mechanical ventilation, circulatory support, or extracorporeal liver support were provided to patients with grade C PHLF.10 The extent of liver resection was based on Couinaud liver classification. Major hepatectomy was defined as resection of three or more Couinaud segments of the liver, while minor resection was defined as resection of fewer than three segments.32 Clinically significant portal hypertension was defined as a relatively low platelet count (<100×109/L) with splenomegaly (major diameter >12 cm) or the presence of esophageal/gastric varices.33,34 Postoperative mortality was determined by death within 90 days of surgery.
Classification of the CP grade was performed using previously published methods.35 The ALBI score was calculated using the following equation: ALBI score=-0.085×(albumin [g/L]+0.66×log10 (bilirubin [μmol/L]) and was classified as grade 1 (≤-2.60), grade 2 (>-2.60 to ≤-1.39), and grade 3 (> -1.39).18,19 The model for end-stage liver disease (MELD) score was calculated using the following formula: 9.57×ln (creatinine [mg/dL])+11.2×ln (INR)+3.78×ln (bilirubin [mg/dL])+ 6.43.36 The ICG retention test was performed by injecting a dose of 0.5 mg/kg of ICG rapidly via the peripheral vein of the forearm, and the ICG-R15 was calculated. The ICG-R15 grades were categorized as follows: grade A (<15), grade B (15-40), and grade C (>40).37 All indicators were collected 1 week prior to hepatectomy, and albumin or fresh-frozen plasma administration was not performed prior to the tests.
Continuous variables were expressed as medians (range) and analyzed using the Mann–Whitney U test. Categorical parameters were presented as frequencies and compared using the χ2 test or Fisher’s exact test. Univariable and multivariable logistic regression analyses were performed to determine independent risk factors associated with PHLF. Age, gender, liver cirrhosis, major hepatectomy, sodium, AST, ALT, maximum tumor size, intraoperative hypotension, ALBI score, ICG-R15 score and ALBI grade were included as independent variables in a backward stepwise multivariate regression for PHLF. Three different multivariate logistic regression analysis models for ALBI score, ICG-R15, and ALBI grade were performed to avoid collinearity. Variance inflation factors were obtained to identify the degree of collinearity between independent variables. The discriminative power of the ALBI score and ICG-R15 score in predicting PHLF was evaluated by receiver operating characteristic (ROC) curve analysis and expressed as area under the ROC curve (AUC). The cutoff values for PHLF were also determined by ROC curve analysis. P values <0.05 were considered statistically significant. Statistical analysis was conducted using SPSS version 23.0 (SPSS Inc., Chicago, IL, USA).
The clinical characteristics and laboratory data of the 101 patients were summarized in Table 1. The present study included 85 men (84.2%) and 16 women (15.8%), with a median age of 62.0 years. Cirrhosis occurred in 75 patients (74.3%). The majority of the patients had liver disease of CP grade A (96/101, 95.0%), and the remaining five had grade B (5.0%). A total of 23 patients (22.8%) underwent major hepatectomy, while 78 (77.4%) underwent minor hepatectomy. Three patients died because of PHLF within 90 days after surgery, with a postoperative mortality rate of 3.0%. The mean ALBI score of the total study population was -2.60 (range -2.90 to -2.10). According to the ALBI score, 50 patients (49.5%) were classified as grade 1, 49 (48.5%) as grade 2, and two (2.0%) as grade 3. The mean ICG-R15 value of the total study population was 10.8 (6.9-17.1); 68 patients (67.3%) were classified as grade A, 30 (29.7%) as grade B, and 3 (3.0%) as grade C. Of the 101 patients, PHLF occurred in 32 patients (31.7%); four patients (12.5%) had grade A, 24 (75.0%) had grade B, and four (12.5%) had grade C PHLF. The mean value of ICG-R15 with PHLF was 14.1 (range 8.2-22.5), significantly higher than that of ICG-R15 (9.9) without PHLF (p=0.041) (Table 1). The mean ALBI score of the PHLF group was -2.30 (range -2.65 to -1.86), which was significantly higher than that of the non-PHLF group (-2.66) (p=0.008).
In univariable analysis, AST, ALT, CP grade, MELD score, ICG-R15 score, ALBI score, and ALBI grade were significantly associated with PHLF (Table 2). For multivariable analysis in the total cohort, ALBI score, ICG-R15 and ALBI grade 2/3 were identified as significantly independent risk factors for PHLF (ALBI score: adjusted OR [aOR] 2.83; 95% CI 1.22-6.55; ICG-R15: aOR 1.07; 95% CI 1.02-1.12; ALBI grade 2/3: aOR 2.86; 95% CI 1.08-7.58) (Table 2). In order to eliminate the effect of small residual liver volume on PHLF, a subgroup analysis was conducted based on the extent of liver resection. Multivariable analysis in the minor hepatectomy group showed that ALBI score and ALBI grade 2/3 were significant risk factors for PHLF, but ICG-R15 was not significantly associated with PHLF (Table 3).
The AUC of the ALBI score and ICG-R15 in predicting PHLF were 0.676 (95% CI 0.566-0.785) and 0.632 (95% CI 0.513-0.752), respectively (Fig. 2). The optimal cutoff value of the ALBI score was -2.62, with a sensitivity of 75.0% and a specificity of 56.5%. The incidence of PHLF in patients with ALBI score >-2.62 was significantly higher than that in patients with an ALBI score ≤-2.62 (p=0.003) (Table 4). Among the patients with ALBI score >-2.62, four patients (16.7%) developed grade A PHLF, 16 (66.7%) had grade B, and four (16.7%) had grade C. Meanwhile, those with ALBI score ≤ -2.62, eight patients had grade B PHLF and none for grade A and C (Table 4). In the subgroup analysis, the discriminative power of ALBI score and ICG-R15 in predicting PHLF was also compared in the minor hepatectomy group. In the minor hepatectomy group, the AUC of the ALBI score and ICG-R15 were 0.657 (95% CI 0.525-0.789) and 0.639 (95% CI 0.497-0.782), respectively (Fig. 3).
In the present study, we first demonstrated the effectiveness of the ALBI score in predicting PHLF in patients with HCC who underwent hepatectomy in South Korea. Multivariate logistic regression analysis revealed that ALBI score and ALBI grade 2/3 had significant associations with PHLF. In addition, the discriminatory power of the ALBI score in predicting PHLF was comparable to that of ICG-R15. The optimal cutoff value of the ALBI score calculated by the ROC curve was -2.62.
We confirmed that the ALBI score and ALBI grade 2/3 had independent and significant associations with PHLF. This result was consistent with previous studies that reported the relationship between the ALBI score and PHLF. According to a retrospective study of 1,242 HCC patients in China, the ALBI score was an independent predictor of PHLF.27 Another retrospective study of 13,783 patients who underwent liver resection as per the National Surgical Quality Improvement Program in the United States of America, ALBI grades 2/3 were significant predictors of PHLF.30 In addition, Zhang et al.25 analyzed 338 HCC patients who underwent hepatectomy and verified that the ALBI grade was a significant prognostic factor of PHLF.
The AUC of the ALBI score in this study (0.676) was relatively lower than those in previous studies: 0.782 by Zhang et al.25, 0.723 by Wang et al.27, and 0.738 by Zou et al.28 This is probably because the study population in this study was smaller than that in previous studies. However, in contrast to previous studies, the ALBI score was compared to ICG-R15 in the present study. The ALBI score had a comparable predictive value for PHLF to ICG-R15. At times, the ICG-R15 test may not be available or accurate in clinical practice. Therefore, ALBI score could be used as an alternative tool for ICG-R15 in the prediction of PHLF as well as the decision on hepatectomy.
CP grade has been widely used for years as a criterion for estimating the risk of liver resection.38 In general, hepatectomy is considered appropriate for patients with CP grade A resectable HCC.39,40 However, in our study, some patients with CP grade A had liver dysfunction with increased serum bilirubin, decreased albumin, or existing ascites, and they developed PHLF. Therefore, background liver status may be different even in patients with CP grade A, so precise criteria are required to predict PHLF. Recent studies have reported significant differences in overall survival and postoperative morbidity depending on the ALBI scores of HCC patients with CP grade A.20,41 In this study, with the optimal cutoff value of the ALBI score of -2.62, we confirmed that the incidence of PHLF was significantly higher in patients with ALBI score >-2.62 than in patients with an ALBI score ≤-2.62. The ALBI score -2.62 is close to -2.60, which is the reference point for ALBI grades 1 and 2. Therefore, the results of our study were consistent with those of previous studies: the incidence of PHLF or postoperative morbidity was higher in patients with ALBI grade 2 than those with ALBI grade 1.26,28 In clinical practice, HCC patients with ALBI grade 2 should acknowledge the possibility of PHLF and prepare for or consider other treatments. Moreover, ALBI grade can be used to reclassify CP grade.
There are several limitations to the current study. First, the incidence of PHLF in the current study was relatively high. Many patients were thought to have impaired liver function, as the number of patients with ALBI grade 2 or 3 and ICG-R15 grade B or C was high in the study population. Second, since this study was a retrospective study and single-center data were used, selection bias may have occurred. Furthermore, due to the relatively small sample size, we did not include some variables such as clinically significant portal hypertension and PT (sec) that strongly reflect portal hypertension in the clinical practice. Third, the relationship between ALBI score and overall survival as well as disease-free survival was not confirmed because of the short follow-up period. A prospective study model with a large sample would be needed. In conclusion, our study confirmed that the ALBI score is an effective predictor of PHLF in patients with HCC, and its predictive ability is comparable to that of ICG-R15.
REFERENCES
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. 2018; Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 68:394–424. DOI: 10.3322/caac.21492. PMID: 30207593.


2. European Association for the Study of the Liver. 2019; Corrigendum to "EASL clinical practice guidelines: management of hepatocellular carcinoma" [J Hepatol 2018;69:182-236]. J Hepatol. 70:817. DOI: 10.1016/j.jhep.2019.01.020. PMID: 30739718.

3. Heimbach JK, Kulik LM, Finn RS, et al. 2018; AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 67:358–380. DOI: 10.1002/hep.29086. PMID: 28130846.


4. Omata M, Cheng AL, Kokudo N, et al. 2017; Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: a 2017 update. Hepatol Int. 11:317–370. DOI: 10.1007/s12072-017-9799-9. PMID: 28620797. PMCID: PMC5491694.



5. Hernaez R, Solà E, Moreau R, Ginès P. 2017; Acute-on-chronic liver failure: an update. Gut. 66:541–553. DOI: 10.1136/gutjnl-2016-312670. PMID: 28053053. PMCID: PMC5534763.



6. Mai RY, Wang YY, Bai T, et al. 2019; Combination of ALBI and APRI To predict post-hepatectomy liver failure after liver resection for HBV-related HCC patients. Cancer Manag Res. 11:8799–8806. DOI: 10.2147/CMAR.S213432. PMID: 31632139. PMCID: PMC6779592.


7. Farges O, Malassagne B, Flejou JF, Balzan S, Sauvanet A, Belghiti J. 1999; Risk of major liver resection in patients with underlying chronic liver disease: a reappraisal. Ann Surg. 229:210–215. DOI: 10.1097/00000658-199902000-00008. PMID: 10024102. PMCID: PMC1191633.


8. Belghiti J, Hiramatsu K, Benoist S, Massault P, Sauvanet A, Farges O. 2000; Seven hundred forty-seven hepatectomies in the 1990s: an update to evaluate the actual risk of liver resection. J Am Coll Surg. 191:38–46. DOI: 10.1016/S1072-7515(00)00261-1. PMID: 10898182.

9. Dimick JB, Wainess RM, Cowan JA, Upchurch GR Jr, Knol JA, Colletti LM. 2004; National trends in the use and outcomes of hepatic resection. J Am Coll Surg. 199:31–38. DOI: 10.1016/j.jamcollsurg.2004.03.005. PMID: 15217626.

10. Rahbari NN, Garden OJ, Padbury R, et al. 2011; Posthepatectomy liver failure: a definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery. 149:713–724. DOI: 10.1016/j.surg.2010.10.001. PMID: 21236455.


11. Schreckenbach T, Liese J, Bechstein WO, Moench C. 2012; Posthepatectomy liver failure. Dig Surg. 29:79–85. DOI: 10.1159/000335741. PMID: 22441624.


12. Kauffmann R, Fong Y. 2014; Post-hepatectomy liver failure. Hepatobiliary Surg Nutr. 3:238–246. DOI: 10.3978/j.issn.2304-3881.2014.09.01. PMID: 25392835. PMCID: PMC4207837.


13. Iguchi K, Hatano E, Yamanaka K, Tanaka S, Taura K, Uemoto S. 2014; The impact of posthepatectomy liver failure on the recurrence of hepatocellular carcinoma. World J Surg. 38:150–158. DOI: 10.1007/s00268-013-2247-7. PMID: 24132820.


14. van Mierlo KM, Schaap FG, Dejong CH, Olde Damink SW. 2016; Liver resection for cancer: new developments in prediction, prevention and management of postresectional liver failure. J Hepatol. 65:1217–1231. DOI: 10.1016/j.jhep.2016.06.006. PMID: 27312944.


15. van den Broek MA, Olde Damink SW, Dejong CH, et al. 2008; Liver failure after partial hepatic resection: definition, pathophysiology, risk factors and treatment. Liver Int. 28:767–780. DOI: 10.1111/j.1478-3231.2008.01777.x. PMID: 18647141.


16. Imamura H, Sano K, Sugawara Y, Kokudo N, Makuuchi M. 2005; Assessment of hepatic reserve for indication of hepatic resection: decision tree incorporating indocyanine green test. J Hepatobiliary Pancreat Surg. 12:16–22. DOI: 10.1007/s00534-004-0965-9. PMID: 15754094.


17. Durand F, Valla D. 2008; Assessment of prognosis of cirrhosis. Semin Liver Dis. 28:110–122. DOI: 10.1055/s-2008-1040325. PMID: 18293281.


18. Johnson PJ, Berhane S, Kagebayashi C, et al. 2015; Assessment of liver function in patients with hepatocellular carcinoma: a new evidence-based approach-the ALBI grade. J Clin Oncol. 33:550–558. DOI: 10.1200/JCO.2014.57.9151. PMID: 25512453. PMCID: PMC4322258.


19. Kubota K, Makuuchi M, Kusaka K, et al. 1997; Measurement of liver volume and hepatic functional reserve as a guide to decision-making in resectional surgery for hepatic tumors. Hepatology. 26:1176–1181. DOI: 10.1053/jhep.1997.v26.pm0009362359. PMID: 9362359.

20. Sonohara F, Yamada S, Tanaka N, et al. 2018; Perioperative and prognostic implication of albumin-bilirubin-TNM score in Child-Pugh class A hepatocellular carcinoma. Ann Gastroenterol Surg. 3:65–74. DOI: 10.1002/ags3.12212. PMID: 30697612. PMCID: PMC6345730.



21. Chan AW, Chan RC, Wong GL, et al. 2015; New simple prognostic score for primary biliary cirrhosis: albumin-bilirubin score. J Gastroenterol Hepatol. 30:1391–1396. DOI: 10.1111/jgh.12938. PMID: 25753927.


22. Ma T, Li QS, Wang Y, et al. 2019; Value of pretransplant albumin-bilirubin score in predicting outcomes after liver transplantation. World J Gastroenterol. 25:1879–1889. DOI: 10.3748/wjg.v25.i15.1879. PMID: 31057301. PMCID: PMC6478615.



23. Zhang W, Liu C, Tan Y, et al. 2018; Albumin-bilirubin score for predicting post-transplant complications following adult-to-adult living donor liver transplantation. Ann Transplant. 23:639–646. DOI: 10.12659/AOT.910824. PMID: 30201946. PMCID: PMC6248303.



24. Li MX, Zhao H, Bi XY, et al. 2017; Prognostic value of the albumin-bilirubin grade in patients with hepatocellular carcinoma: validation in a Chinese cohort. Hepatol Res. 47:731–741. DOI: 10.1111/hepr.12796. PMID: 27558521.


25. Zhang ZQ, Xiong L, Zhou JJ, et al. 2018; Ability of the ALBI grade to predict posthepatectomy liver failure and long-term survival after liver resection for different BCLC stages of HCC. World J Surg Oncol. 16:208. DOI: 10.1186/s12957-018-1500-9. PMID: 30326907. PMCID: PMC6192221.



26. Andreatos N, Amini N, Gani F, et al. 2017; Albumin-bilirubin score: predicting short-term outcomes including bile leak and post-hepatectomy liver failure following hepatic resection. J Gastrointest Surg. 21:238–248. DOI: 10.1007/s11605-016-3246-4. PMID: 27619809.


27. Wang YY, Zhong JH, Su ZY, et al. 2016; Albumin-bilirubin versus Child-Pugh score as a predictor of outcome after liver resection for hepatocellular carcinoma. Br J Surg. 103:725–734. DOI: 10.1002/bjs.10095. PMID: 27005482.


28. Zou H, Wen Y, Yuan K, Miao XY, Xiong L, Liu KJ. 2018; Combining albumin-bilirubin score with future liver remnant predicts post-hepatectomy liver failure in HBV-associated HCC patients. Liver Int. 38:494–502. DOI: 10.1111/liv.13514. PMID: 28685924.


29. Zou H, Yang X, Li QL, Zhou QX, Xiong L, Wen Y. 2018; A comparative study of albumin-bilirubin score with Child-Pugh score, model for end-stage liver disease score and indocyanine green R15 in predicting posthepatectomy liver failure for hepatocellular carcinoma patients. Dig Dis. 36:236–243. DOI: 10.1159/000486590. PMID: 29495004.


30. Fagenson AM, Gleeson EM, Pitt HA, Lau KN. 2020; Albumin-bilirubin score vs model for end-stage liver disease in predicting post-hepatectomy outcomes. J Am Coll Surg. 230:637–645. DOI: 10.1016/j.jamcollsurg.2019.12.007. PMID: 31954813.


31. Balzan S, Belghiti J, Farges O, et al. 2005; The "50-50 criteria" on postoperative day 5: an accurate predictor of liver failure and death after hepatectomy. Ann Surg. 242:824–829. DOI: 10.1097/01.sla.0000189131.90876.9e. PMID: 16327492. PMCID: PMC1409891.


32. Pol B, Campan P, Hardwigsen J, Botti G, Pons J, Le Treut YP. 1999; Morbidity of major hepatic resections: a 100-case prospective study. Eur J Surg. 165:446–453. DOI: 10.1080/110241599750006686. PMID: 10391161.

33. Chen X, Zhai J, Cai X, et al. 2012; Severity of portal hypertension and prediction of postoperative liver failure after liver resection in patients with Child-Pugh grade A cirrhosis. Br J Surg. 99:1701–1710. DOI: 10.1002/bjs.8951. PMID: 23132418.

34. Augustin S, Millán L, González A, et al. 2014; Detection of early portal hypertension with routine data and liver stiffness in patients with asymptomatic liver disease: a prospective study. J Hepatol. 60:561–569. DOI: 10.1016/j.jhep.2013.10.027. PMID: 24211744.


35. Pugh RN, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R. 1973; Transection of the oesophagus for bleeding oesophageal varices. Br J Surg. 60:646–649. DOI: 10.1002/bjs.1800600817. PMID: 4541913.


36. Butt AA, Ren Y, Lo Re V 3rd, Taddei TH, Kaplan DE. 2017; Comparing Child-Pugh, MELD, and FIB-4 to predict clinical outcomes in hepatitis C virus-infected persons: results from ERCHIVES. Clin Infect Dis. 65:64–72. DOI: 10.1093/cid/cix224. PMID: 28369305.


37. Omagari K, Ohba K, Kadokawa Y, et al. 2006; Comparison of the grade evaluated by "liver damage" of Liver Cancer Study Group of Japan and Child-Pugh classification in patients with hepatocellular carcinoma. Hepatol Res. 34:266–272. DOI: 10.1016/j.hepres.2005.05.010. PMID: 16460997.


38. Forner A, Llovet JM, Bruix J. 2012; Hepatocellular carcinoma. Lancet. 379:1245–1255. DOI: 10.1016/S0140-6736(11)61347-0. PMID: 29307467.


39. Bruix J, Reig M, Sherman M. 2016; Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma. Gastroenterology. 150:835–853. DOI: 10.1053/j.gastro.2015.12.041. PMID: 26795574.


40. Clavien PA, Petrowsky H, DeOliveira ML, Graf R. 2007; Strategies for safer liver surgery and partial liver transplantation. N Engl J Med. 356:1545–1559. DOI: 10.1056/NEJMra065156. PMID: 17429086.


41. Dong ZR, Zou J, Sun D, et al. 2017; Preoperative Albumin-bilirubin score for postoperative solitary hepatocellular carcinoma within the milan criteria and Child-Pugh A cirrhosis. J Cancer. 8:3862–3867. DOI: 10.7150/jca.21313. PMID: 29151974. PMCID: PMC5688940.

Fig. 1
Flowchart of study population. HCC, hepatocellular carcinoma; TACE, transcatheter arterial chemoembolization.
Fig. 2
ROC curve analyses of ALBI score and ICG-R15 in predicting PHLF in the total cohort. ROC, receiver operation characteristic; PHLF, post-hepatectomy liver failure; ALBI, albumin-bilirubin; ICG-R15, indocyanine green retention rate at 15 minutes.
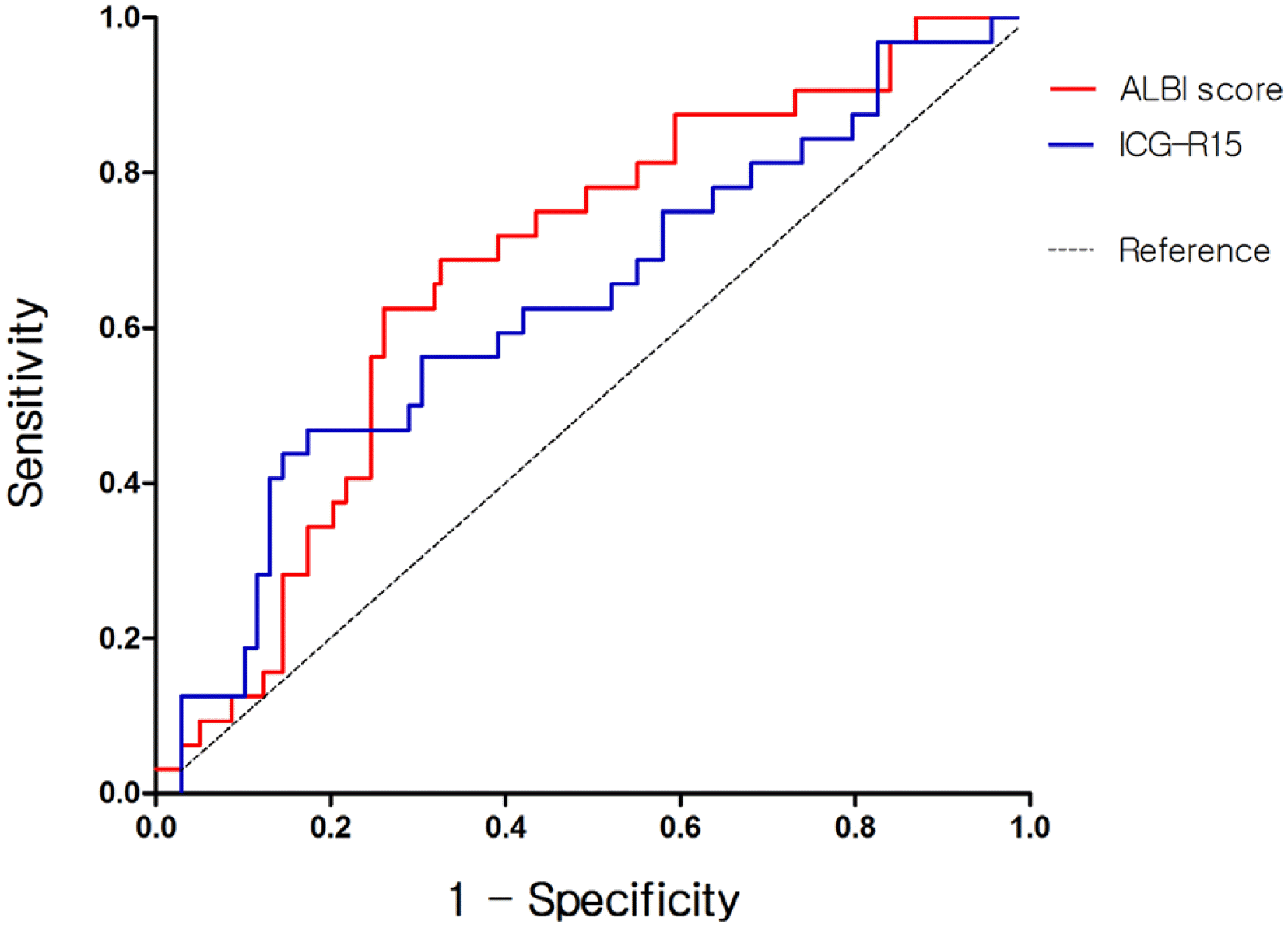
Fig. 3
ROC curve analyses of ALBI score and ICG-R15 in predicting PHLF in the minor hepatectomy group. ROC, receiver operation characteristic; PHLF, post-hepatectomy liver failure; ALBI, albumin-bilirubin; ICG-R15, indocyanine green retention rate at 15 minutes.
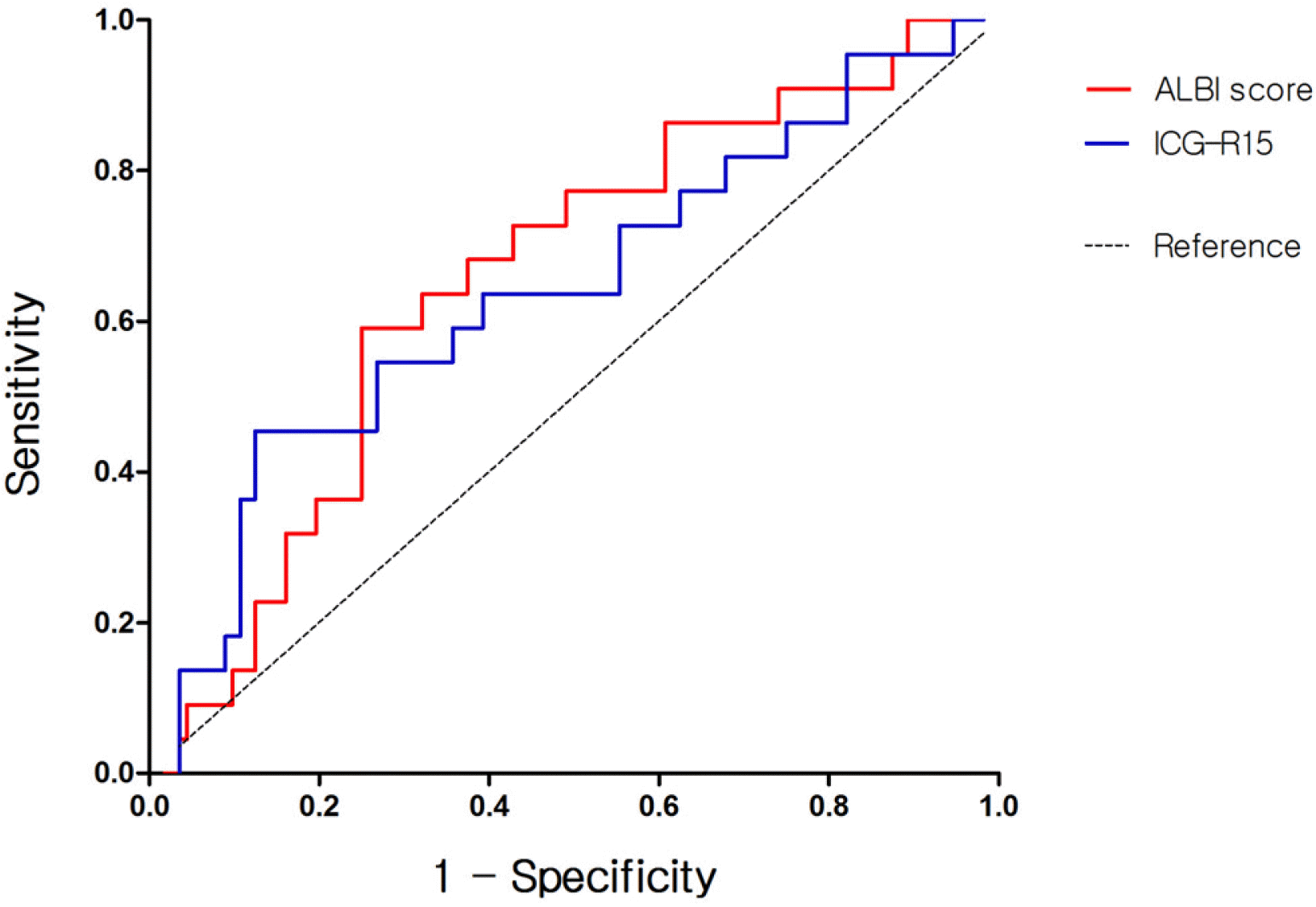
Table 1
Baseline Characteristics of Study Population
PHLF, post-hepatectomy liver failure; HBsAg, hepatitis B surface antigen; HCV, hepatitis C virus; AST, aspartate aminotransferase; ALT, alanine aminotransferase; PT, prothrombin time; AFP, alpha-fetoprotein; CSPH, clinically significant portal hypertension; MELD, model for end-stage liver disease; ICG-R15, indocyanine green retention rate at 15 minutes; ALBI, albumin-bilirubin; BCLC, Barcelona Clinic Liver Cancer.
Table 2
Univariable and Multivariable Logistic Regression Analysis for Post-hepatectomy Liver Failure in the Total Cohort
Table 3
Multivariable Logistic Regression Analysis for Post-hepatectomy Liver Failure in the Subgroup of Minor Hepatectomy




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download