1. Lee CH, Lin LJ, Cheng CL, Kao Yang YH, Chen JY, Tsai LM. 2010; Incidence and cumulative recurrence rates of venous thrombo-embolism in the Taiwanese population. J Thromb Haemost. 8:1515–23. DOI:
10.1111/j.1538-7836.2010.03873.x. PMID:
20345707.

2. Jang MJ, Bang SM, Oh D. 2011; Incidence of venous thromboembolism in Korea: from the Health Insurance Review and Assessment Service database. J Thromb Haemost. 9:85–91. DOI:
10.1111/j.1538-7836.2010.04108.x. PMID:
20942850.
6. Yhim HY, Choi WI, Kim SH, et al. 2019; Long-term rivaroxaban for the treatment of acute venous thromboembolism in patients with active cancer in a prospective multicenter trial. Korean J Intern Med. 34:1125–35. DOI:
10.3904/kjim.2018.097. PMID:
29788694. PMCID:
PMC6718768.

7. Boon GJAM, Van Dam LF, Klok FA, Huisman MV. 2018; Management and treatment of deep vein thrombosis in special populations. Expert Rev Hematol. 11:685–95. DOI:
10.1080/17474086.2018.1502082. PMID:
30016119.

8. EINSTEIN-PE Investigators. 2012; Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med. 366:1287–97. DOI:
10.1056/NEJMoa1113572. PMID:
22449293.
9. Agnelli G, Buller HR, Cohen A, et al. 2013; Oral apixaban for the treatment of acute venous thromboembolism. N Engl J Med. 369:799–808. DOI:
10.1056/NEJMoa1302507. PMID:
23808982.

10. Hokusai-VTE Investigators. 2013; Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism. N Engl J Med. 369:1406–15. DOI:
10.1056/NEJMoa1306638. PMID:
23991658.

11. Schulman S, Kearon C, Kakkar AK, et al. 2009; Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 361:2342–52. DOI:
10.1056/NEJMoa0906598. PMID:
19966341.

12. Hakeam HA, Al-Sanea N. 2017; Effect of major gastrointestinal tract surgery on the absorption and efficacy of direct acting oral anticoagulants (DOACs). J Thromb Thrombolysis. 43:343–51. DOI:
10.1007/s11239-016-1465-x. PMID:
28050755.
13. Raskob GE, van Es N, Verhamme P, et al. 2018; Edoxaban for the treatment of cancer-associated venous thromboembolism. N Engl J Med. 378:615–24. DOI:
10.1056/NEJMoa1711948. PMID:
29231094.

14. Lee AY, Levine MN, Baker RI, et al. 2003; Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med. 349:146–53. DOI:
10.1056/NEJMoa025313. PMID:
12853587.

15. Galanaud JP, Laroche JP, Righini M. 2013; The history and historical treatments of deep vein thrombosis. J Thromb Haemost. 11:402–11. DOI:
10.1111/jth.12127. PMID:
23297815.

16. Kearon C, Akl EA, Ornelas J, et al. 2016; Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest. 149:315–52. DOI:
10.1016/j.chest.2015.11.026. PMID:
26867832.

17. Horner D, Hogg K, Body R, Nash MJ, Baglin T, Mackway-Jones K. 2014; The anticoagulation of calf thrombosis (ACT) project: results from the randomized controlled external pilot trial. Chest. 146:1468–77. DOI:
10.1378/chest.14-0235. PMID:
25010443.

18. Lagerstedt CI, Olsson CG, Fagher BO, Oqvist BW, Albrechtsson U. 1985; Need for long-term anticoagulant treatment in symptomatic calf-vein thrombosis. Lancet. 2:515–8. DOI:
10.1016/S0140-6736(85)90459-3. PMID:
2863541.

19. Pinede L, Ninet J, Duhaut P, et al. 2001; Comparison of 3 and 6 months of oral anticoagulant therapy after a first episode of proximal deep vein thrombosis or pulmonary embolism and comparison of 6 and 12 weeks of therapy after isolated calf deep vein thrombosis. Circulation. 103:2453–60. DOI:
10.1161/01.CIR.103.20.2453. PMID:
11369685.

20. Schwarz T, Buschmann L, Beyer J, Halbritter K, Rastan A, Schellong S. 2010; Therapy of isolated calf muscle vein thrombosis: a randomized, controlled study. J Vasc Surg. 52:1246–50. DOI:
10.1016/j.jvs.2010.05.094. PMID:
20630682.

21. Franco L, Giustozzi M, Agnelli G, Becattini C. 2017; Anticoagulation in patients with isolated distal deep vein thrombosis: a meta-analysis. J Thromb Haemost. 15:1142–54. DOI:
10.1111/jth.13677. PMID:
28316124.

22. Righini M, Galanaud JP, Guenneguez H, et al. 2016; Anticoagulant therapy for symptomatic calf deep vein thrombosis (CACTUS): a randomised, double-blind, placebo-controlled trial. Lancet Haematol. 3:e556–62. DOI:
10.1016/S2352-3026(16)30131-4. PMID:
27836513.
23. Ho P, Lim HY, Chua CC, et al. 2016; Retrospective review on isolated distal deep vein thrombosis (IDDVT) - a benign entity or not? Thromb Res. 142:11–6. DOI:
10.1016/j.thromres.2016.04.003. PMID:
27093232.

24. Chatterjee S, Chakraborty A, Weinberg I, et al. 2014; Thrombolysis for pulmonary embolism and risk of all-cause mortality, major bleeding, and intracranial hemorrhage: a meta-analysis. JAMA. 311:2414–21. DOI:
10.1001/jama.2014.5990. PMID:
24938564.

25. Schulman S, Kakkar AK, Goldhaber SZ, et al. 2014; Treatment of acute venous thromboembolism with dabigatran or warfarin and pooled analysis. Circulation. 129:764–72. DOI:
10.1161/CIRCULATIONAHA.113.004450. PMID:
24344086.

27. Goy J, Lee J, Levine O, Chaudhry S, Crowther M. 2015; Sub-segmental pulmonary embolism in three academic teaching hospitals: a review of management and outcomes. J Thromb Haemost. 13:214–8. DOI:
10.1111/jth.12803. PMID:
25442511.

29. den Exter PL, van Es J, Klok FA, et al. 2013; Risk profile and clinical outcome of symptomatic subsegmental acute pulmonary embolism. Blood. 122:1144–9. quiz 1329DOI:
10.1182/blood-2013-04-497545. PMID:
23736701.

30. O'Connell C. 2015; Incidentally found pulmonary embolism: what's the clinician to do? Hematology Am Soc Hematol Educ Program. 2015:197–201. DOI:
10.1182/asheducation-2015.1.197. PMID:
26637721.

31. Lee AYY, Kamphuisen PW, Meyer G, et al. 2015; Tinzaparin vs warfarin for treatment of acute venous thromboembolism in patients with active cancer: a randomized clinical trial. JAMA. 314:677–86. DOI:
10.1001/jama.2015.9243. PMID:
26284719.

32. Vedovati MC, Germini F, Agnelli G, Becattini C. 2015; Direct oral anticoagulants in patients with VTE and cancer: a systematic review and meta-analysis. Chest. 147:475–83. DOI:
10.1378/chest.14-0402. PMID:
25211264.

33. Young AM, Marshall A, Thirlwall J, et al. 2018; Comparison of an oral factor Xa inhibitor with low molecular weight heparin in patients with cancer with venous thromboembolism: results of a randomized trial (SELECT-D). J Clin Oncol. 36:2017–23. DOI:
10.1200/JCO.2018.78.8034. PMID:
29746227.

34. Key NS, Khorana AA, Kuderer NM, et al. 2020; Venous thrombo-embolism prophylaxis and treatment in patients with cancer: ASCO clinical practice guideline update. J Clin Oncol. 38:496–520. DOI:
10.1200/JCO.19.01461. PMID:
31381464.

35. Kim SA, Yhim HY, Bang SM. 2019; Current management of cancer-associated venous thromboembolism: focus on direct oral anticoagulants. J Korean Med Sci. 34:e52. DOI:
10.3346/jkms.2019.34.e52. PMID:
30787683. PMCID:
PMC6374546.
36. Scott G, Mahdi AJ, Alikhan R. 2015; Superficial vein thrombosis: a current approach to management. Br J Haematol. 168:639–45. DOI:
10.1111/bjh.13255. PMID:
25521017.

37. Di Nisio M, Wichers I, Middeldorp S. 2018; Treatment of lower extremity superficial thrombophlebitis. JAMA. 320:2367–8. DOI:
10.1001/jama.2018.16623. PMID:
30383173.

39. Beyer-Westendorf J, Schellong SM, Gerlach H, et al. 2017; Prevention of thromboembolic complications in patients with superficial-vein thrombosis given rivaroxaban or fondaparinux: the open-label, randomised, non-inferiority SURPRISE phase 3b trial. Lancet Haematol. 4:e105–13. DOI:
10.1016/S2352-3026(17)30014-5. PMID:
28219692.
40. Debourdeau P, Farge D, Beckers M, et al. 2013; International clinical practice guidelines for the treatment and prophylaxis of thrombosis associated with central venous catheters in patients with cancer. J Thromb Haemost. 11:71–80. DOI:
10.1111/jth.12071. PMID:
23217208.

41. Barco S, Atema JJ, Coppens M, Serlie MJ, Middeldorp S. 2017; Anticoagulants for the prevention and treatment of catheter-related thrombosis in adults and children on parenteral nutrition: a systematic review and critical appraisal. Blood Transfus. 15:369–77. DOI:
10.2450/2016.0031-16. PMID:
27483479. PMCID:
PMC5490734.


42. Baumann Kreuziger L, Jaffray J, Carrier M. 2017; Epidemiology, diagnosis, prevention and treatment of catheter-related thrombosis in children and adults. Thromb Res. 157:64–71. DOI:
10.1016/j.thromres.2017.07.002. PMID:
28710972.

44. Kahn SR, Lim W, Dunn AS, et al. 2012; Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 141(2 Suppl):e195S–e226S. DOI:
10.1378/chest.141.5.1369b. PMID:
22315261. PMCID:
PMC3278052.
45. De Stefano V, Martinelli I. 2010; Splanchnic vein thrombosis: clinical presentation, risk factors and treatment. Intern Emerg Med. 5:487–94. DOI:
10.1007/s11739-010-0413-6. PMID:
20532730.

46. Riva N, Ageno W. 2017; Approach to thrombosis at unusual sites: splanchnic and cerebral vein thrombosis. Vasc Med. 22:529–40. DOI:
10.1177/1358863X17734057. PMID:
29202678.

47. Smalberg JH, Arends LR, Valla DC, Kiladjian JJ, Janssen HL, Leebeek FW. 2012; Myeloproliferative neoplasms in Budd-Chiari syndrome and portal vein thrombosis: a meta-analysis. Blood. 120:4921–8. DOI:
10.1182/blood-2011-09-376517. PMID:
23043069.

48. Kearon C, Akl EA, Comerota AJ, et al. 2012; Antithrombotic therapy for VTE disease: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 141(2 Suppl):e419S–e96S. DOI:
10.1378/chest.141.5.1369b. PMID:
22315268. PMCID:
PMC3278049.
49. DeLeve LD, Valla DC, Garcia-Tsao G. American Association for the Study Liver Diseases. 2009; Vascular disorders of the liver. Hepatology. 49:1729–64. DOI:
10.1002/hep.22772. PMID:
19399912. PMCID:
PMC6697263.


50. Ageno W, Riva N, Schulman S, et al. 2015; Long-term clinical outcomes of splanchnic vein thrombosis: results of an international registry. JAMA Intern Med. 175:1474–80. DOI:
10.1001/jamainternmed.2015.3184. PMID:
26168152.

51. Ageno W, Beyer-Westendorf J, Garcia DA, Lazo-Langner A, McBane RD, Paciaroni M. 2016; Guidance for the management of venous thrombosis in unusual sites. J Thromb Thrombolysis. 41:129–43. DOI:
10.1007/s11239-015-1308-1. PMID:
26780742. PMCID:
PMC4715841.


52. Kourlaba G, Relakis J, Kontodimas S, Holm MV, Maniadakis N. 2016; A systematic review and meta-analysis of the epidemiology and burden of venous thromboembolism among pregnant women. Int J Gynaecol Obstet. 132:4–10. DOI:
10.1016/j.ijgo.2015.06.054. PMID:
26489486.

53. Bates SM, Rajasekhar A, Middeldorp S, et al. 2018; American Society of Hematology 2018 guidelines for management of venous thromboembolism: venous thromboembolism in the context of pregnancy. Blood Adv. 2:3317–59. DOI:
10.1182/bloodadvances.2018024802. PMID:
30482767. PMCID:
PMC6258928.


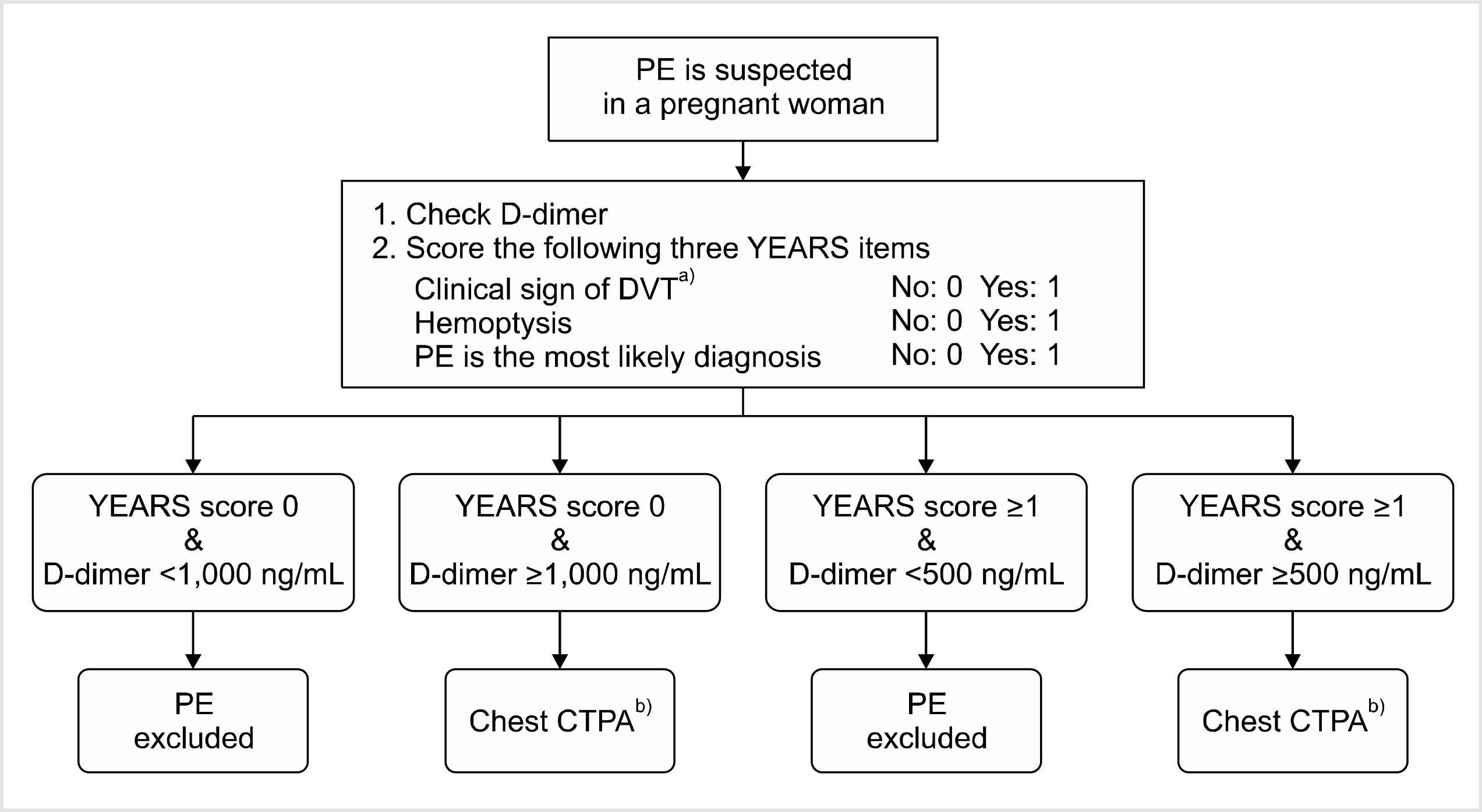
54. van der Pol LM, Tromeur C, Bistervels IM, et al. 2019; Pregnancy-adapted YEARS algorithm for diagnosis of suspected pulmonary embolism. N Engl J Med. 380:1139–49. DOI:
10.1056/NEJMoa1813865. PMID:
30893534.

55. Lameijer H, Aalberts JJJ, van Veldhuisen DJ, Meijer K, Pieper PG. 2018; Efficacy and safety of direct oral anticoagulants during pregnancy; a systematic literature review. Thromb Res. 169:123–7. DOI:
10.1016/j.thromres.2018.07.022. PMID:
30036784.

56. Knol HM, Schultinge L, Veeger NJ, Kluin-Nelemans HC, Erwich JJ, Meijer K. 2012; The risk of postpartum hemorrhage in women using high dose of low-molecular-weight heparins during pregnancy. Thromb Res. 130:334–8. DOI:
10.1016/j.thromres.2012.03.007. PMID:
22475315.

57. Klaassen IL, van Ommen CH, Middeldorp S. 2015; Manifestations and clinical impact of pediatric inherited thrombophilia. Blood. 125:1073–7. DOI:
10.1182/blood-2014-05-536060. PMID:
25564402.

58. Nowak-Göttl U, von Kries R, Göbel U. 1997; Neonatal symptomatic thromboembolism in Germany: two year survey. Arch Dis Child Fetal Neonatal Ed. 76:F163–7. DOI:
10.1136/fn.76.3.F163. PMID:
9175945. PMCID:
PMC1720641.
59. van Ommen CH, Heijboer H, Büller HR, Hirasing RA, Heijmans HS, Peters M. 2001; Venous thromboembolism in childhood: a prospective two-year registry in The Netherlands. J Pediatr. 139:676–81. DOI:
10.1067/mpd.2001.118192. PMID:
11713446.

61. Andrew M, David M, Adams M, et al. 1994; Venous thromboembolic complications (VTE) in children: first analyses of the Canadian Registry of VTE. Blood. 83:1251–7. DOI:
10.1182/blood.V83.5.1251.1251. PMID:
8118029.
62. Miletich J, Sherman L, Broze G Jr. 1987; Absence of thrombosis in subjects with heterozygous protein C deficiency. N Engl J Med. 317:991–6. DOI:
10.1056/NEJM198710153171604. PMID:
3657866.

63. Tait RC, Walker ID, Reitsma PH, et al. 1995; Prevalence of protein C deficiency in the healthy population. Thromb Haemost. 73:87–93. DOI:
10.1055/s-0038-1653730. PMID:
7740502.

64. Mateo J, Oliver A, Borrell M, Sala N, Fontcuberta J. 1997; Laboratory evaluation and clinical characteristics of 2,132 consecutive unselected patients with venous thromboembolism--results of the Spanish Multicentric Study on Thrombophilia (EMET-Study). Thromb Haemost. 77:444–51. DOI:
10.1055/s-0038-1655986. PMID:
9065991.

65. Heijboer H, Brandjes DP, Büller HR, Sturk A, ten Cate JW. 1990; Deficiencies of coagulation-inhibiting and fibrinolytic proteins in outpatients with deep-vein thrombosis. N Engl J Med. 323:1512–6. DOI:
10.1056/NEJM199011293232202. PMID:
2146503.

66. Pintao MC, Ribeiro DD, Bezemer ID, et al. 2013; Protein S levels and the risk of venous thrombosis: results from the MEGA case-control study. Blood. 122:3210–9. DOI:
10.1182/blood-2013-04-499335. PMID:
24014240.

69. Andrew M, Paes B, Milner R, et al. 1987; Development of the human coagulation system in the full-term infant. Blood. 70:165–72. DOI:
10.1182/blood.V70.1.165.165. PMID:
3593964.

70. Appel IM, Grimminck B, Geerts J, Stigter R, Cnossen MH, Beishuizen A. 2012; Age dependency of coagulation parameters during childhood and puberty. J Thromb Haemost. 10:2254–63. DOI:
10.1111/j.1538-7836.2012.04905.x. PMID:
22909016.

71. Gavva C, Sarode R, Zia A. 2017; A clinical audit of thrombophilia testing in pediatric patients with acute thromboembolic events: impact on management. Blood Adv. 1:2386–91. DOI:
10.1182/bloodadvances.2017009514. PMID:
29296888. PMCID:
PMC5729621.


73. Manco-Johnson MJ, Grabowski EF, Hellgreen M, et al. 2002; Laboratory testing for thrombophilia in pediatric patients. On behalf of the Subcommittee for Perinatal and Pediatric Thrombosis of the Scientific and Standardization Committee of the International Society of Thrombosis and Haemostasis (ISTH). Thromb Haemost. 88:155–6. DOI:
10.1055/s-0037-1613169. PMID:
12152657.

74. Ageno W, Dentali F, Squizzato A, et al. 2010; Evidence and clinical judgment: treatment of cerebral vein thrombosis. Thromb Haemost. 103:1109–15. DOI:
10.1160/TH09-12-0827. PMID:
20390232.

75. De Stefano V, Rossi E. 2013; Testing for inherited thrombophilia and consequences for antithrombotic prophylaxis in patients with venous thromboembolism and their relatives. A review of the Guidelines from Scientific Societies and Working Groups. Thromb Haemost. 110:697–705. DOI:
10.1160/TH13-01-0011. PMID:
23846575.

76. Neshat-Vahid S, Pierce R, Hersey D, Raffini LJ, Faustino EV. 2016; Association of thrombophilia and catheter-associated thrombosis in children: a systematic review and meta-analysis. J Thromb Haemost. 14:1749–58. DOI:
10.1111/jth.13388. PMID:
27306795. PMCID:
PMC5035642.


77. Monagle P, Chan AKC, Goldenberg NA, et al. 2012; Antithrombotic therapy in neonates and children: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 141:e737S–e801S. DOI:
10.1378/chest.11-2308. PMID:
22315277. PMCID:
PMC3278066.
78. Monagle P, Cuello CA, Augustine C, et al. 2018; American Society of Hematology 2018 Guidelines for management of venous thromboembolism: treatment of pediatric venous thrombo-embolism. Blood Adv. 2:3292–316. DOI:
10.1182/bloodadvances.2018024786. PMID:
30482766. PMCID:
PMC6258911.


79. Raffini LJ, Scott JP. 2016; Thrombotic disorders in children. In: Kliegman RM, Stanton BF, St Geme JW, Schor N, eds. Nelson textbook of pediatrics. 20th ed. Philadelphia, PA:. Elsevier,. 2394–7.
83. Hirsh J, Bauer KA, Donati MB, Gould M, Samama MM, Weitz JI. 2008; Parenteral anticoagulants: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 133(6 Suppl):141S–59S. DOI:
10.1378/chest.08-0689. PMID:
18574264.
84. Pollack CV Jr, Reilly PA, van Ryn J, et al. 2017; Idarucizumab for dabigatran reversal - full cohort analysis. N Engl J Med. 377:431–41. DOI:
10.1056/NEJMoa1707278. PMID:
28693366.

85. Connolly SJ, Crowther M, Eikelboom JW, et al. 2019; Full study report of andexanet alfa for bleeding associated with factor Xa inhibitors. N Engl J Med. 380:1326–35. DOI:
10.1056/NEJMoa1814051. PMID:
30730782. PMCID:
PMC6699827.
86. Ansell JE, Bakhru SH, Laulicht BE, et al. 2017; Single-dose ciraparantag safely and completely reverses anticoagulant effects of edoxaban. Thromb Haemost. 117:238–45. DOI:
10.1160/TH16-03-0224. PMID:
27853809. PMCID:
PMC6260118.


88. van Ryn J, Stangier J, Haertter S, et al. 2010; Dabigatran etexilate--a novel, reversible, oral direct thrombin inhibitor: interpretation of coagulation assays and reversal of anticoagulant activity. Thromb Haemost. 103:1116–27. DOI:
10.1160/TH09-11-0758. PMID:
20352166.

89. Hawes EM, Deal AM, Funk-Adcock D, et al. 2013; Performance of coagulation tests in patients on therapeutic doses of dabigatran: a cross-sectional pharmacodynamic study based on peak and trough plasma levels. J Thromb Haemost. 11:1493–502. DOI:
10.1111/jth.12308. PMID:
23718677.

90. Hillarp A, Baghaei F, Fagerberg Blixter I, et al. 2011; Effects of the oral, direct factor Xa inhibitor rivaroxaban on commonly used coagulation assays. J Thromb Haemost. 9:133–9. DOI:
10.1111/j.1538-7836.2010.04098.x. PMID:
20946166.

91. Seiffge DJ, Traenka C, Polymeris AA, et al. 2017; Intravenous thrombolysis in patients with stroke taking rivaroxaban using drug specific plasma levels: experience with a standard operation procedure in clinical practice. J Stroke. 19:347–55. DOI:
10.5853/jos.2017.00395. PMID:
28877563. PMCID:
PMC5647628.


92. Schmitz EM, Boonen K, van den Heuvel DJ, et al. 2014; Determination of dabigatran, rivaroxaban and apixaban by ultra-performance liquid chromatography - tandem mass spectrometry (UPLC-MS/MS) and coagulation assays for therapy monitoring of novel direct oral anticoagulants. J Thromb Haemost. 12:1636–46. DOI:
10.1111/jth.12702. PMID:
25142183.

93. Forbes HL, Polasek TM. 2017; Potential drug-drug interactions with direct oral anticoagulants in elderly hospitalized patients. Ther Adv Drug Saf. 8:319–28. DOI:
10.1177/2042098617719815. PMID:
29593860. PMCID:
PMC5865464.






 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download