1. Link MS. Clinical practice. Evaluation and initial treatment of supraventricular tachycardia. N Engl J Med. 2012; 367(15):1438–1448. PMID:
23050527.
2. Orejarena LA, Vidaillet H Jr, DeStefano F, Nordstrom DL, Vierkant RA, Smith PN, et al. Paroxysmal supraventricular tachycardia in the general population. J Am Coll Cardiol. 1998; 31(1):150–157. PMID:
9426034.

3. Benowitz NL. Drug therapy. Pharmacologic aspects of cigarette smoking and nicotine addiction. N Engl J Med. 1988; 319(20):1318–1330. PMID:
3054551.
4. Howard G, Wagenknecht LE, Burke GL, Diez-Roux A, Evans GW, McGovern P, et al. Cigarette smoking and progression of atherosclerosis: the Atherosclerosis Risk in Communities (ARIC) study. JAMA. 1998; 279(2):119–124. PMID:
9440661.
5. Goldenberg I, Moss AJ, McNitt S, Zareba W, Daubert JP, Hall WJ, et al. Cigarette smoking and the risk of supraventricular and ventricular tachyarrhythmias in high-risk cardiac patients with implantable cardioverter defibrillators. J Cardiovasc Electrophysiol. 2006; 17(9):931–936. PMID:
16759297.

6. Chamberlain AM, Agarwal SK, Folsom AR, Duval S, Soliman EZ, Ambrose M, et al. Smoking and incidence of atrial fibrillation: results from the Atherosclerosis Risk in Communities (ARIC) study. Heart Rhythm. 2011; 8(8):1160–1166. PMID:
21419237.

7. Heeringa J, Kors JA, Hofman A, van Rooij FJ, Witteman JC. Cigarette smoking and risk of atrial fibrillation: the Rotterdam Study. Am Heart J. 2008; 156(6):1163–1169. PMID:
19033014.

8. Suzuki S, Otsuka T, Sagara K, Kano H, Matsuno S, Takai H, et al. Association between smoking habits and the first-time appearance of atrial fibrillation in Japanese patients: evidence from the Shinken Database. J Cardiol. 2015; 66(1):73–79. PMID:
25458170.

9. Zuo H, Nygård O, Vollset SE, Ueland PM, Ulvik A, Midttun Ø, et al. Smoking, plasma cotinine and risk of atrial fibrillation: the Hordaland Health Study. J Intern Med. 2018; 283(1):73–82. PMID:
28940460.

10. Suzuki S, Sagara K, Otsuka T, Kano H, Matsuno S, Takai H, et al. Effects of smoking habit on the prevalence of atrial fibrillation in Japanese patients with special reference to sex differences. Circ J. 2013; 77(12):2948–2953. PMID:
24065034.

11. Connor Gorber S, Schofield-Hurwitz S, Hardt J, Levasseur G, Tremblay M. The accuracy of self-reported smoking: a systematic review of the relationship between self-reported and cotinine-assessed smoking status. Nicotine Tob Res. 2009; 11(1):12–24. PMID:
19246437.
12. Wagenknecht LE, Burke GL, Perkins LL, Haley NJ, Friedman GD. Misclassification of smoking status in the CARDIA study: a comparison of self-report with serum cotinine levels. Am J Public Health. 1992; 82(1):33–36. PMID:
1536331.

13. Benowitz NL. Cotinine as a biomarker of environmental tobacco smoke exposure. Epidemiol Rev. 1996; 18(2):188–204. PMID:
9021312.

14. Agaku IT, King BA. Validation of self-reported smokeless tobacco use by measurement of serum cotinine concentration among US adults. Am J Epidemiol. 2014; 180(7):749–754. PMID:
25125690.

15. Kim BJ, Seo DC, Kim BS, Kang JH. Relationship between cotinine-verified smoking status and incidence of hypertension in 74,743 Korean adults. Circ J. 2018; 82(6):1659–1665. PMID:
29491326.

16. SRNT Subcommittee on Biochemical Verification. Biochemical verification of tobacco use and cessation. Nicotine Tob Res. 2002; 4(2):149–159. PMID:
12028847.
17. Kim BJ, Han JM, Kang JG, Rhee EJ, Kim BS, Kang JH. Relationship of cotinine-verified and self-reported smoking status with metabolic syndrome in 116,094 Korean adults. J Clin Lipidol. 2017; 11(3):638–645.e2. PMID:
28431854.

18. Goette A, Lendeckel U, Kuchenbecker A, Bukowska A, Peters B, Klein HU, et al. Cigarette smoking induces atrial fibrosis in humans via nicotine. Heart. 2007; 93(9):1056–1063. PMID:
17395670.

19. Mehta MC, Jain AC, Mehta A, Billie M. Cardiac arrhythmias following intravenous nicotine: experimental study in dogs. J Cardiovasc Pharmacol Ther. 1997; 2(4):291–298. PMID:
10684470.

20. Miyauchi M, Qu Z, Miyauchi Y, Zhou SM, Pak H, Mandel WJ, et al. Chronic nicotine in hearts with healed ventricular myocardial infarction promotes atrial flutter that resembles typical human atrial flutter. Am J Physiol Heart Circ Physiol. 2005; 288(6):H2878–86. PMID:
15665050.

21. Kim BJ, Han JM, Kang JG, Kim BS, Kang JH. The association between self-reported versus nicotine metabolite-confirmed smoking status and coronary artery calcification. Coron Artery Dis. 2018; 29(3):254–261. PMID:
28984635.

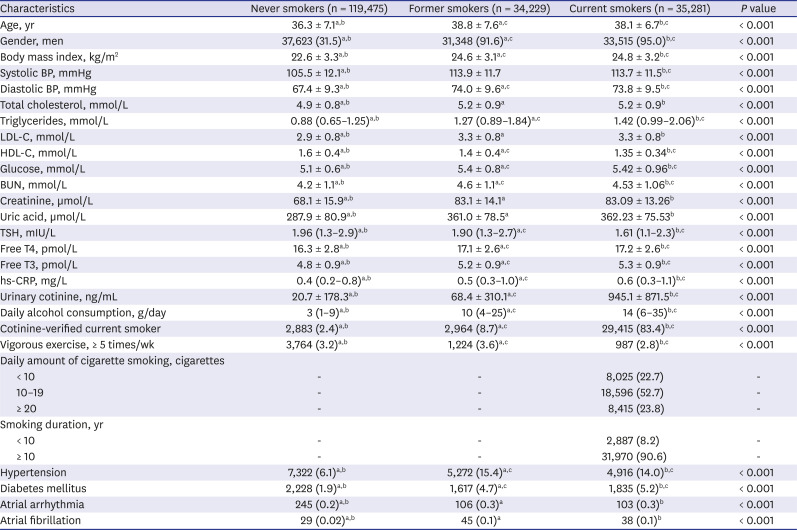
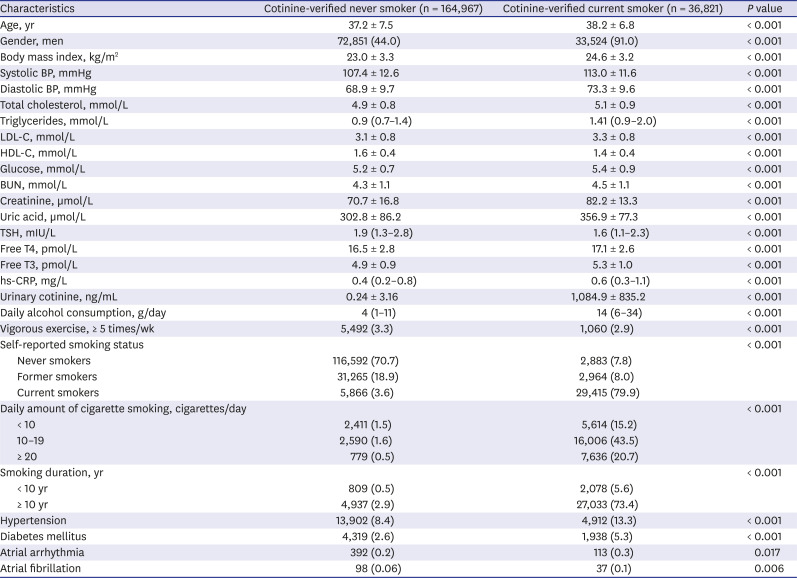
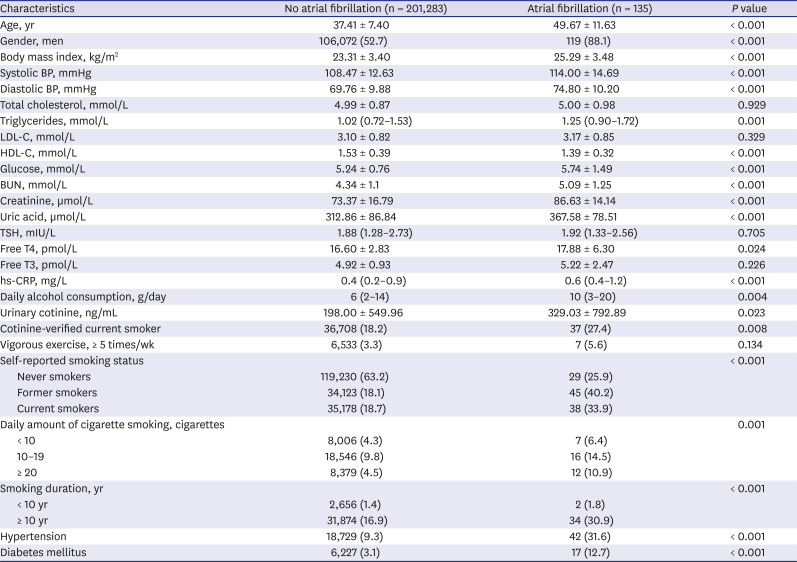
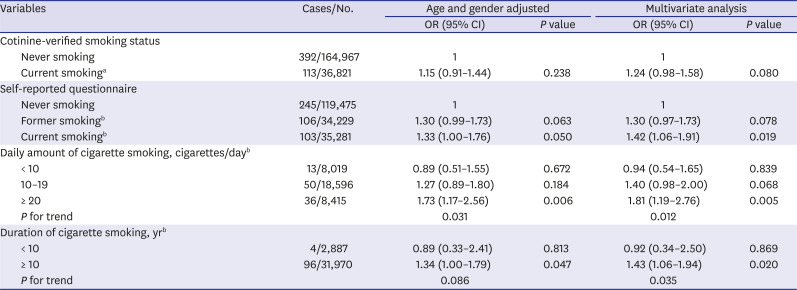




 PDF
PDF Citation
Citation Print
Print




 XML Download
XML Download