1. de Franchis R. Baveno V Faculty. Revising consensus in portal hypertension: report of the Baveno V consensus workshop on methodology of diagnosis and therapy in portal hypertension. J Hepatol. 2010; 53(4):762–768. PMID:
20638742.

2. Lin DY, Sheen IS, Chiu CT, Lin SM, Kuo YC, Liaw YF. Ultrasonographic changes of early liver cirrhosis in chronic hepatitis B: a longitudinal study. J Clin Ultrasound. 1993; 21(5):303–308. PMID:
8514896.

3. de Franchis R. Baveno VI Faculty. Expanding consensus in portal hypertension: report of the Baveno VI Consensus Workshop: stratifying risk and individualizing care for portal hypertension. J Hepatol. 2015; 63(3):743–752. PMID:
26047908.
4. Augustin S, Millán L, González A, Martell M, Gelabert A, Segarra A, et al. Detection of early portal hypertension with routine data and liver stiffness in patients with asymptomatic liver disease: a prospective study. J Hepatol. 2014; 60(3):561–569. PMID:
24211744.

5. Giannini E, Botta F, Borro P, Risso D, Romagnoli P, Fasoli A, et al. Platelet count/spleen diameter ratio: proposal and validation of a non-invasive parameter to predict the presence of oesophageal varices in patients with liver cirrhosis. Gut. 2003; 52(8):1200–1205. PMID:
12865282.

6. Kim G, Kim MY, Baik SK. Transient elastography versus hepatic venous pressure gradient for diagnosing portal hypertension: a systematic review and meta-analysis. Clin Mol Hepatol. 2017; 23(1):34–41. PMID:
28263953.

7. Hong WK, Kim MY, Baik SK, Shin SY, Kim JM, Kang YS, et al. The usefulness of non-invasive liver stiffness measurements in predicting clinically significant portal hypertension in cirrhotic patients: Korean data. Clin Mol Hepatol. 2013; 19(4):370–375. PMID:
24459641.

8. Maurice JB, Brodkin E, Arnold F, Navaratnam A, Paine H, Khawar S, et al. Validation of the Baveno VI criteria to identify low risk cirrhotic patients not requiring endoscopic surveillance for varices. J Hepatol. 2016; 65(5):899–905. PMID:
27388923.

9. Perazzo H, Fernandes FF, Castro Filho EC, Perez RM. Points to be considered when using transient elastography for diagnosis of portal hypertension according to the Baveno's VI consensus. J Hepatol. 2015; 63(4):1048–1049. PMID:
26206072.

10. Augustin S, Pons M, Genesca J. Validating the Baveno VI recommendations for screening varices. J Hepatol. 2017; 66(2):459–460. PMID:
27826055.

11. Marot A, Trépo E, Doerig C, Schoepfer A, Moreno C, Deltenre P. Liver stiffness and platelet count for identifying patients with compensated liver disease at low risk of variceal bleeding. Liver Int. 2017; 37(5):707–716. PMID:
27862856.

12. Bae J, Sinn DH, Kang W, Gwak GY, Choi MS, Paik YH, et al. Validation of the Baveno VI and the expanded Baveno VI criteria to identify patients who could avoid screening endoscopy. Liver Int. 2018; 38(8):1442–1448. PMID:
29495113.

13. Jangouk P, Turco L, De Oliveira A, Schepis F, Villa E, Garcia-Tsao G. Validating, deconstructing and refining Baveno criteria for ruling out high-risk varices in patients with compensated cirrhosis. Liver Int. 2017; 37(8):1177–1183. PMID:
28160373.

14. Silva MJ, Bernardes C, Pinto J, Loureiro R, Duarte P, Mendes M, et al. Baveno VI recommendation on avoidance of screening endoscopy in cirrhotic patients: Are we there yet? GE Port J Gastroenterol. 2017; 24(2):79–83. PMID:
28848787.

15. Sousa M, Fernandes S, Proença L, Silva AP, Leite S, Silva J, et al. The Baveno VI criteria for predicting esophageal varices: validation in real life practice. Rev Esp Enferm Dig. 2017; 109(10):704–707. PMID:
28776387.

16. Castéra L, Foucher J, Bernard PH, Carvalho F, Allaix D, Merrouche W, et al. Pitfalls of liver stiffness measurement: a 5-year prospective study of 13,369 examinations. Hepatology. 2010; 51(3):828–835. PMID:
20063276.

17. Muller M, Gennisson JL, Deffieux T, Tanter M, Fink M. Quantitative viscoelasticity mapping of human liver using supersonic shear imaging: preliminary in vivo feasibility study. Ultrasound Med Biol. 2009; 35(2):219–229. PMID:
19081665.
18. Kim TY, Kim TY, Kim Y, Lim S, Jeong WK, Sohn JH. Diagnostic performance of shear wave elastography for predicting esophageal varices in patients with compensated liver cirrhosis. J Ultrasound Med. 2016; 35(7):1373–1381. PMID:
27208198.

19. Alvarez MA, Cirera I, Solà R, Bargalló A, Morillas RM, Planas R. Long-term clinical course of decompensated alcoholic cirrhosis: a prospective study of 165 patients. J Clin Gastroenterol. 2011; 45(10):906–911. PMID:
21814145.
20. Bell H, Jahnsen J, Kittang E, Raknerud N, Sandvik L. Long-term prognosis of patients with alcoholic liver cirrhosis: a 15-year follow-up study of 100 Norwegian patients admitted to one unit. Scand J Gastroenterol. 2004; 39(9):858–863. PMID:
15513384.

21. Friedrich-Rust M, Ong MF, Martens S, Sarrazin C, Bojunga J, Zeuzem S, et al. Performance of transient elastography for the staging of liver fibrosis: a meta-analysis. Gastroenterology. 2008; 134(4):960–974.e8. PMID:
18395077.

22. Tsochatzis EA, Gurusamy KS, Ntaoula S, Cholongitas E, Davidson BR, Burroughs AK. Elastography for the diagnosis of severity of fibrosis in chronic liver disease: a meta-analysis of diagnostic accuracy. J Hepatol. 2011; 54(4):650–659. PMID:
21146892.

23. Nahon P, Kettaneh A, Tengher-Barna I, Ziol M, de Lédinghen V, Douvin C, et al. Assessment of liver fibrosis using transient elastography in patients with alcoholic liver disease. J Hepatol. 2008; 49(6):1062–1068. PMID:
18930329.

24. Nguyen-Khac E, Chatelain D, Tramier B, Decrombecque C, Robert B, Joly JP, et al. Assessment of asymptomatic liver fibrosis in alcoholic patients using Fibroscan: prospective comparison with seven non-invasive laboratory tests. Aliment Pharmacol Ther. 2008; 28(10):1188–1198. PMID:
18705692.

25. Janssens F, de Suray N, Piessevaux H, Horsmans Y, de Timary P, Stärkel P. Can transient elastography replace liver histology for determination of advanced fibrosis in alcoholic patients: a real-life study. J Clin Gastroenterol. 2010; 44(8):575–582. PMID:
20104185.
26. Michalak S, Rousselet MC, Bedossa P, Pilette C, Chappard D, Oberti F, et al. Respective roles of porto-septal fibrosis and centrilobular fibrosis in alcoholic liver disease. J Pathol. 2003; 201(1):55–62. PMID:
12950017.

27. Zhang X, Wong GL, Wong VW. Application of transient elastography in nonalcoholic fatty liver disease. Clin Mol Hepatol. 2020; 26(2):128–141. PMID:
31696690.

28. Sharma P, Kirnake V, Tyagi P, Bansal N, Singla V, Kumar A, et al. Spleen stiffness in patients with cirrhosis in predicting esophageal varices. Am J Gastroenterol. 2013; 108(7):1101–1107. PMID:
23629600.

29. Stefanescu H, Grigorescu M, Lupsor M, Procopet B, Maniu A, Badea R. Spleen stiffness measurement using Fibroscan for the noninvasive assessment of esophageal varices in liver cirrhosis patients. J Gastroenterol Hepatol. 2011; 26(1):164–170. PMID:
21175810.

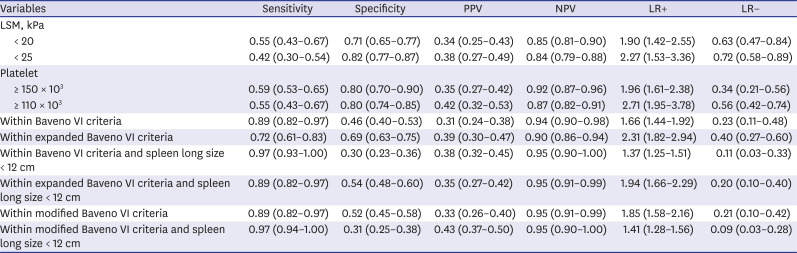
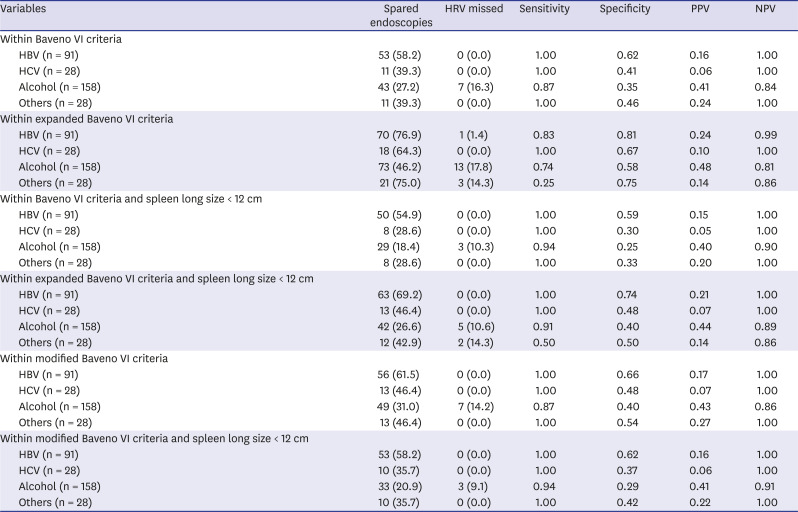




 PDF
PDF Citation
Citation Print
Print




 XML Download
XML Download