1. Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 2003; 26(Suppl 1):S5–S20. PMID:
12502614.
2. Kim YJ, Kim JG, Lee JY, Lee KS, Joe SG, Park JY, et al. Development and progression of diabetic retinopathy and associated risk factors in Korean patients with type 2 diabetes: the experience of a tertiary center. J Korean Med Sci. 2014; 29(12):1699–1705. PMID:
25469073.

3. Yau JW, Rogers SL, Kawasaki R, Lamoureux EL, Kowalski JW, Bek T, et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012; 35(3):556–564. PMID:
22301125.

4. Cheung N, Mitchell P, Wong TY. Diabetic retinopathy. Lancet. 2010; 376(9735):124–136. PMID:
20580421.

5. Alinia C, Mohammadi SF, Lashay A, Rashidian A. Impact of diabetic retinopathy on health-related quality of life in iranian diabetics. Iran J Public Health. 2017; 46(1):55–65. PMID:
28451530.
6. Cho S, Shin JY, Kim HJ, Eun SJ, Kang S, Jang WM, et al. Chasms in achievement of recommended diabetes care among geographic regions in Korea. J Korean Med Sci. 2019; 34(31):e190. PMID:
31392852.

7. Kussmann M, Morine MJ, Hager J, Sonderegger B, Kaput J. Perspective: a systems approach to diabetes research. Front Genet. 2013; 4:205. PMID:
24187547.

8. Jee D, Lee WK, Kang S. Prevalence and risk factors for diabetic retinopathy: the Korea National Health and Nutrition Examination Survey 2008–2011. Invest Ophthalmol Vis Sci. 2013; 54(10):6827–6833. PMID:
24065813.

9. Liu L, Wu J, Yue S, Geng J, Lian J, Teng W, et al. Incidence density and risk factors of diabetic retinopathy within type 2 diabetes: a five-year cohort study in China (report 1). Int J Environ Res Public Health. 2015; 12(7):7899–7909. PMID:
26184262.

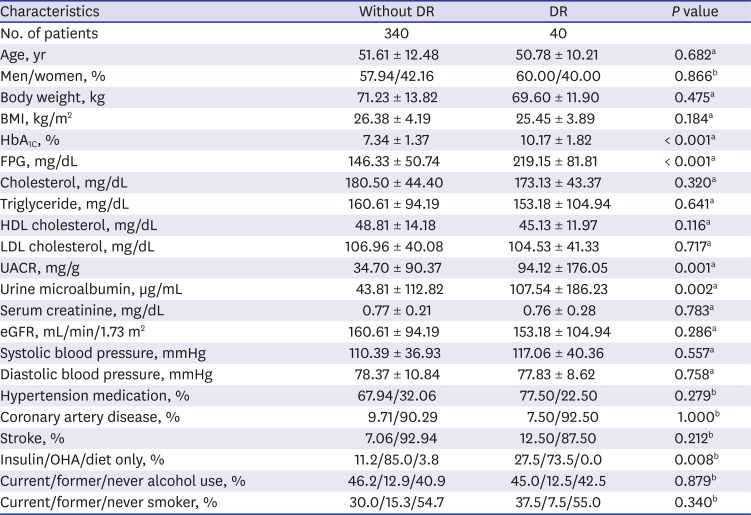
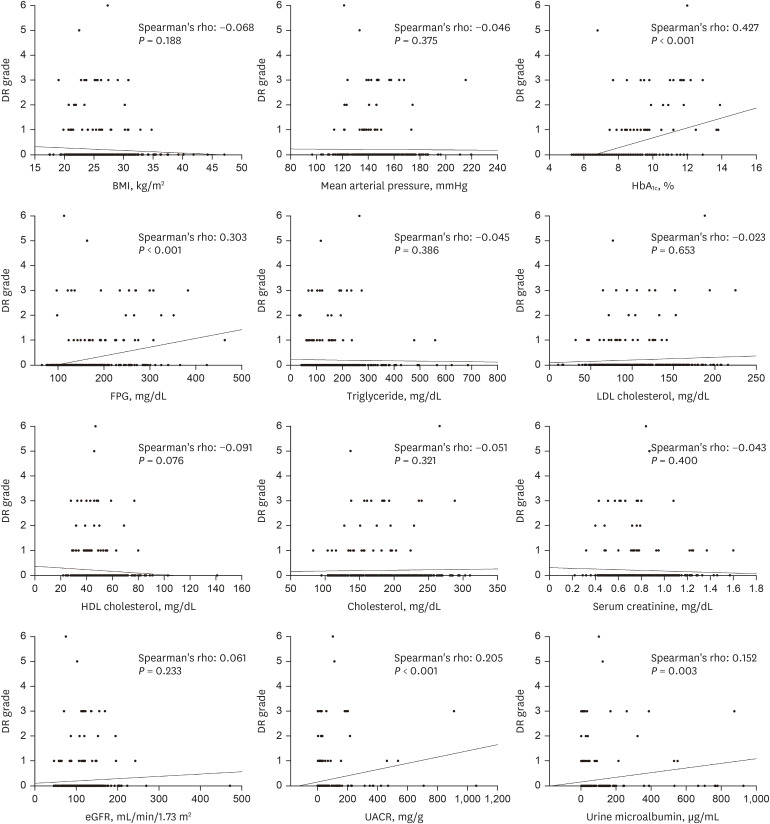
10. Park S, Rhee SY, Jeong SJ, Kim K, Chon S, Yu SY, et al. Features of long-standing Korean type 2 diabetes mellitus patients with diabetic retinopathy: a study based on standardized clinical data. Diabetes Metab J. 2017; 41(5):393–404. PMID:
29086538.

11. Chong YH, Fan Q, Tham YC, Gan A, Tan SP, Tan G, et al. Type 2 diabetes genetic variants and risk of diabetic retinopathy. Ophthalmology. 2017; 124(3):336–342. PMID:
28038984.

12. Stefánsson E, Bek T, Porta M, Larsen N, Kristinsson JK, Agardh E. Screening and prevention of diabetic blindness. Acta Ophthalmol Scand. 2000; 78(4):374–385. PMID:
10990036.

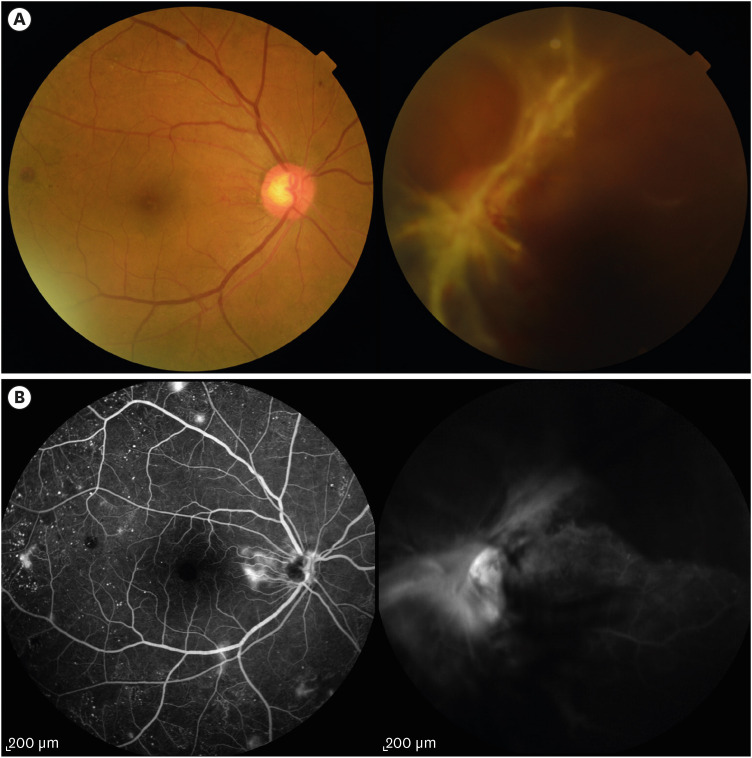
13. Wu L, Fernandez-Loaiza P, Sauma J, Hernandez-Bogantes E, Masis M. Classification of diabetic retinopathy and diabetic macular edema. World J Diabetes. 2013; 4(6):290–294. PMID:
24379919.

14. Wilkinson CP, Ferris FL 3rd, Klein RE, Lee PP, Agardh CD, Davis M, et al. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology. 2003; 110(9):1677–1682. PMID:
13129861.

15. Bressler SB, Odia I, Glassman AR, Danis RP, Grover S, Hampton GR, et al. Changes in diabetic retinopathy severity when treating diabetic macular edema with ranibizumab: DRCR.net protocol I 5-year report. Retina. 2018; 38(10):1896–1904. PMID:
30234859.
16. Roy Chowdhury S, Thomas RL, Dunseath GJ, Peter R, Rees DA, North RV, et al. Diabetic retinopathy in newly diagnosed subjects with type 2 diabetes mellitus: contribution of β-cell function. J Clin Endocrinol Metab. 2016; 101(2):572–580. PMID:
26652932.

17. Hainsworth DP, Bebu I, Aiello LP, Sivitz W, Gubitosi-Klug R, Malone J, et al. Risk factors for retinopathy in type 1 diabetes: the DCCT/EDIC study. Diabetes Care. 2019; 42(5):875–882. PMID:
30833368.

18. Stratton IM, Cull CA, Adler AI, Matthews DR, Neil HA, Holman RR. Additive effects of glycaemia and blood pressure exposure on risk of complications in type 2 diabetes: a prospective observational study (UKPDS 75). Diabetologia. 2006; 49(8):1761–1769. PMID:
16736131.

19. Anand KV, Lal CA, Ac G, Js K, Saurabh A. Assessment of the relationship between microalbuminuria, diabetic nephropathy & diabetic retinopathy. J Assoc Physicians India. 2016; 64(1):92.
20. Yun JS, Lim TS, Cha SA, Ahn YB, Song KH, Choi JA, et al. Clinical course and risk factors of diabetic retinopathy in patients with type 2 diabetes mellitus in Korea. Diabetes Metab J. 2016; 40(6):482–493. PMID:
27766793.

21. Romero-Aroca P, Baget-Bernaldiz M, Navarro-Gil R, Moreno-Ribas A, Valls-Mateu A, Sagarra-Alamo R, et al. Glomerular filtration rate and/or ratio of urine albumin to creatinine as markers for diabetic retinopathy: a ten-year follow-up study. J Diabetes Res. 2018; 2018:5637130. PMID:
29682579.

22. Hsieh YT, Tsai MJ, Tu ST, Hsieh MC. Association of abnormal renal profiles and proliferative diabetic retinopathy and diabetic macular edema in an Asian population with type 2 diabetes. JAMA Ophthalmol. 2018; 136(1):68–74. PMID:
29167896.

23. Cha DR, Kang YS, Han SY, Jee YH, Han KH, Han JY, et al. Vascular endothelial growth factor is increased during early stage of diabetic nephropathy in type II diabetic rats. J Endocrinol. 2004; 183(1):183–194. PMID:
15525586.

24. Pawlak K, Mysliwiec M, Pawlak D. Oxidative stress, phosphate and creatinine levels are independently associated with vascular endothelial growth factor levels in patients with chronic renal failure. Cytokine. 2008; 43(1):98–101. PMID:
18455421.

25. Lee WJ, Sobrin L, Lee MJ, Kang MH, Seong M, Cho H. The relationship between diabetic retinopathy and diabetic nephropathy in a population-based study in Korea (KNHANES V-2, 3). Invest Ophthalmol Vis Sci. 2014; 55(10):6547–6553. PMID:
25205863.

26. Lim LS, Cheung CY, Sabanayagam C, Lim SC, Tai ES, Huang L, et al. Structural changes in the retinal microvasculature and renal function. Invest Ophthalmol Vis Sci. 2013; 54(4):2970–2976. PMID:
23572105.

27. Kwon SH, Shin JP, Kim IT, Park DH. Association of plasma semaphorin 3A with phenotypes of diabetic retinopathy and nephropathy. Invest Ophthalmol Vis Sci. 2016; 57(7):2983–2989. PMID:
27273597.

28. Miljanovic B, Glynn RJ, Nathan DM, Manson JE, Schaumberg DA. A prospective study of serum lipids and risk of diabetic macular edema in type 1 diabetes. Diabetes. 2004; 53(11):2883–2892. PMID:
15504969.

29. Knickelbein JE, Abbott AB, Chew EY. Fenofibrate and diabetic retinopathy. Curr Diab Rep. 2016; 16(10):90. PMID:
27525681.

30. Keech AC, Mitchell P, Summanen PA, O'Day J, Davis TM, Moffitt MS, et al. Effect of fenofibrate on the need for laser treatment for diabetic retinopathy (FIELD study): a randomised controlled trial. Lancet. 2007; 370(9600):1687–1697. PMID:
17988728.

31. Tserentsoodol N, Gordiyenko NV, Pascual I, Lee JW, Fliesler SJ, Rodriguez IR. Intraretinal lipid transport is dependent on high density lipoprotein-like particles and class B scavenger receptors. Mol Vis. 2006; 12:1319–1333. PMID:
17110915.
32. The effect of intensive diabetes treatment on the progression of diabetic retinopathy in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial. Arch Ophthalmol. 1995; 113(1):36–51. PMID:
7826293.
33. ACCORD Eye Study Group, Chew EY, Ambrosius WT, Davis MD, Danis RP, Gangaputra S, et al. Effects of medical therapies on retinopathy progression in type 2 diabetes. N Engl J Med. 2010; 363(3):233–244. PMID:
20587587.

34. Fong DS, Aiello L, Gardner TW, King GL, Blankenship G, Cavallerano JD, et al. Retinopathy in diabetes. Diabetes Care. 2004; 27(Suppl 1):S84–7. PMID:
14693935.

35. Harris Nwanyanwu K, Talwar N, Gardner TW, Wrobel JS, Herman WH, Stein JD. Predicting development of proliferative diabetic retinopathy. Diabetes Care. 2013; 36(6):1562–1568. PMID:
23275374.

36. Hwang H, Chae JB, Kim JY, Moon BG, Kim DY. Changes in optical coherence tomography findings in patients with chronic renal failure undergoing dialysis for the first time. Retina. 2019; 39(12):2360–2368. PMID:
30180144.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download