INTRODUCTION
In order to obtain a successful endodontic treatment, the removal of microorganisms, its by-products, and pulp remnants from root canal walls during instrumentation is essential [
1]. A combination of endodontic instruments, mechanical action, and properties of irrigant solutions is responsible for this clinical step. Despite several advances in techniques, anatomical variations remain as the main limiting factor to obtain complete cleaning, often presenting remnant pulp tissue in root canal isthmuses, ramifications and internal walls, when analyzed histologically [
2].
Endodontic therapy must accomplish a satisfactory root canal preparation. For this purpose, a vast arsenal of nickel-titanium (NiTi) instruments with different designs, alloys, tapers, motions, surface treatment, and tip sizes have been developed. Overcoming the anatomical limitations and mitigate the adverse events inherent to instrumentation, such as deviations and fractures, especially in oval and curved canals, must be the primary goal during instrument development [
23].
Hero 642 NiTi rotary system (Micromega, Besançon, France) is composed of 0.02, 0.04, and 0.06 tapered instruments with a triple-helix cross-section. Reciproc system (VDW, Munich, Germany) was developed using a NiTi alloy (M-Wire) with additional surface heat treatment aiming to enhance its mechanical and physical properties [
3]. Both instrumentation systems have been demonstrating satisfactory results regarding root canal shaping and its anatomical original shape preservation [
345].
Experimental methods have been used to assess root canal shaping with these endodontic instrumentation systems [
67]. Studies using microcomputed tomography (µCT) showed large areas of the root canal remaining untouched after instrumentation [
78]. The apical histological examination allows the evaluation of these areas, especially in the cross-sectional plane, including pulp tissue remnants accumulation. Furthermore, analysis combinations allow measurements of the pulp tissue remnants present in untouched places by the endodontic instrumentation concerning the total area of the root canal [
910].
The use of rotary (multiple-instruments) or reciprocating (single-instrument) NiTi systems in endodontics has led to debates regarding root canal preparation, such as the instrument motion efficacy and the apical limit of instrumentation [
11]. In theory, instrument design and kinematics should ensure maximum contact with the canal walls and, combined with the action of irrigant solutions, obtain efficient shaping of the root canal, maintaining its original shape even after instrumentation [
6].
The present study aimed to evaluate the apical pulp tissue removal, using histological analysis, in mandibular molars distal roots after in vivo preparation using reciprocating and rotary instruments.
MATERIALS AND METHODS
This study is in compliant with Consolidated Standards of Reporting Trials (CONSORT) 2010 guidelines (
Figure 1) and was reviewed and approved by an ethical board of Piracicaba Dental School according to protocol number 786.618 and was performed under the ethical standards the Declaration of Helsinki. The informed written consent and its understanding were obtained for all participants included in the study ranging from the years 2015 to 2017 from a single-center Endodontic clinic. Thirty patients scheduled for extraction of first or second mandibular molars, due to periodontal problems or teeth without restoration possibilities, were selected and randomly divided by a member of the staff, blind to the study purposes, into 2 test groups (
n = 20) and one control group (
n = 10). The inclusion criteria were teeth presenting a positive response to cold sensibility test using Endo Ice (Hygienic Corporation, Akron, OH, USA) and distal roots with single root canals, curvature angle between 10 and 20 degrees and curvature radius lower than 10 mm, assessed in digital periapical radiographs (
Figure 2) [
12].
Figure 1
Consolidated Standards of Reporting Trials (CONSORT) 2010 flow diagram.
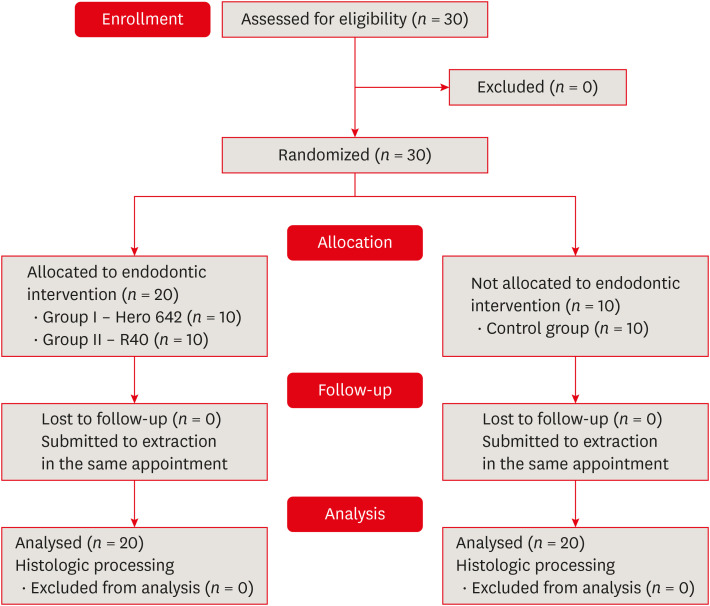
Figure 2
Representative diagrams used for curvature assessment previously to the operative procedures for distal roots included in the analysis. (A) Periapical radiograph used to curvature angle determination and (B) curvature radius determination [12].
For the test groups, a single operator performed all endodontic procedures. After anesthesia and rubber dam isolation, endodontic access was obtained, the distal canal was located, and blood originated specifically from this root canal was assessed in order to confirm the presence of vital pulp tissue. Glide path was established using size 10 K-files (Dentsply-Maillefer, Ballaigues, Switzerland) and sizes 13, 16, and 19 Pathfile (Dentsply-Maillefer) to standardize the root canal surgical diameter before instrumentation. Cervical flaring was performed with a Reciproc R40 instrument with 3 in-and-out motions to a maximum length of 12 mm and followed by irrigation solely with 5 mL of saline solution, without irrigation solutions, in order to evaluate the isolated instrument action. Working length (WL) was determined using an apex locator (Novapex, Forum Engineering Technologies, Yehud, Israel), and when reading showed ‘apex’, the WL was added 1 mm. Teeth were then divided into 2 groups, and instrumentation followed.
In group I (n = 10), distal root canals were instrumented using VDW Silver Reciproc (VDW) with the complete Hero 642 rotary system, set at a speed of 300 rpm and torque of 2.5 N, in the following sequence.
• Size 20/0.02 at WL + 1 mm
• Size 25/0.02 at WL + 1 mm
• Size 25/0.04 until resistance to introduction
• Size 30/0.02 at WL + 1 mm
• Size 35/0.02 at WL + 1 mm
• Size 30/0.06 until resistance to introduction
• Size 40/0.02 at WL + 1 mm
• Size 45/0.02 at WL + 1 mm
The final foraminal instrumentation diameter, considering instrument taper in group I was 0.47 mm. The instrument was removed from the root canal immediately after reference length was achieved. At each instrument change, irrigation was performed using 5 mL saline solution with 40 mL in total.
In group II (n = 10), distal root canals were instrumented to WL + 1 mm, with the Reciproc R40 single-instrument in reciprocating motion system resulting in a final foraminal instrumentation diameter of 0.46 mm. Three in-and-out motions were followed by irrigation with 5 mL of saline solution and passage of a size 10 K-file between each instrument in WL according to manufacturer's instruction. This sequence was repeated as many times as necessary to reach the full WL. The volume of irrigant was evened in 40 mL to ensure there were no discrepancies between groups.
As a positive control (n = 10), distal roots without any endodontic procedure were selected for the evaluation of pulp tissue presence and predentin layer. Control samples were not included in quantitative analysis; once this group solely provided a histological pattern of pulp tissue content without endodontic instrumentation, but submitted to histological analysis. This procedure aimed to avoid the bias of samples pulp removal due to histological processing.
In sequence, teeth were extracted, labeled, and the distal roots processed for histological analysis. After the decalcification process, segments from 5-mm distal roots from the apex were cut transversely with a scalpel blade before inclusion in paraffin. For the morphometric study, five 6-µm histological sections were selected in the apex-crown direction for each root. In order to make the sample more homogeneous, the first cut was selected; the subsequent 50 cuts were discarded and cut 51 was selected for morphometry. Following this methodology, 5 cuts from each root were selected. The staining process used was hematoxylin and eosin (
Figure 3). Images were captured, scanned, and imported into ImageJ software (Reproductive Sciences Branch, National Institutes of Health, Bethesda, MD, USA).
Figure 3
Representative image (×50) (hematoxylin and eosin) of root canal cross-sectional perimeter used as a reference to evaluate surface considered as touched by instrumentation (arrows). The untouched area in the right shows the structure of normal predentin that was not removed by the action of the instruments, which was a frequent observation, especially in isthmus areas of oval root canal sections.
The linear perimeter of each root canal cross-section was then traced using the software ‘freehand’ selection tool to determine the perimeter of action of the tested endodontic instrument by the difference of touched/untouched image aspect (
Figure 4). Values obtained in pixels were converted to percentage touched/untouched. The same images and criteria were then used to determine the area of the root canal cross-section touched/untouched by the instrument. The software ‘oval’ selection tool was used to obtain the instrument area of action subtracted from the total root canal cross-section area.
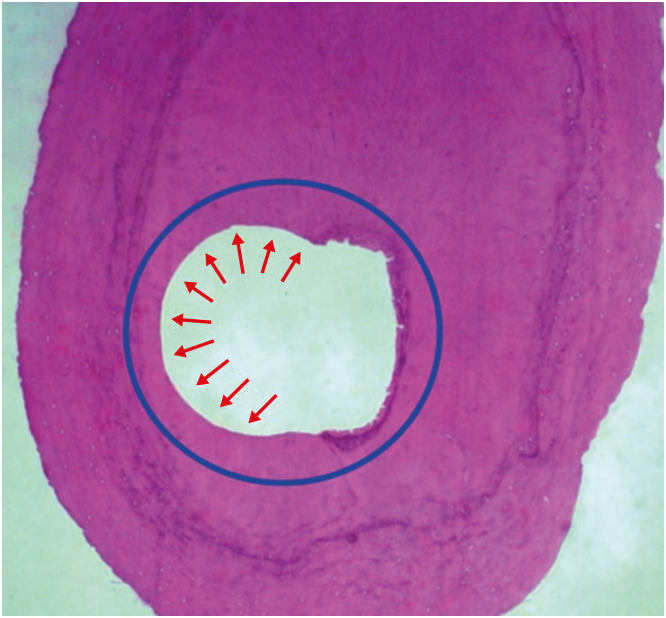
Figure 4
Histological images used for the assessment of perimeter and area. (A) Representative images (×50) (hematoxylin and eosin) with touched perimeter traced in yellow used to calculate touched/untouched perimeter ratio. (B) Representative images (×50) with total area traced in yellow used to calculate percentage area occupied by the instrument (blue circle).
Besides, the control group selected cuts were analyzed regarding its root canal shape according to a previously reported study [
13] in order to obtain information about the selected sample homogeneity. The same software ‘freehand’ tool provided the ratio of long to short canal diameter and the software ‘line’ tool used to determine the measurement in pixels. This analysis was not possible to be performed in the test groups due to instrumentation results, which altered its original shape.
Data were submitted to statistical analysis using the t-test according to obtained values using SPSS Statistics 24 (IBM, Armonk, NY, USA). Significance level was set at 5% (p < 0.05). The sample size was determined using the software SAS 9.4 (SAS Institute Inc., Cary, NC, USA) to express a power test of over 80%.
RESULTS
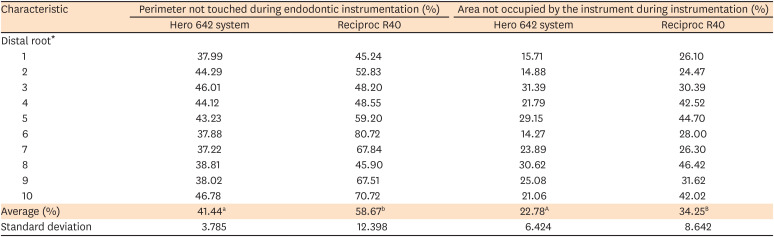
The percentage average of the 5 histological cuts for each distal root, regarding perimeter and area not touched during endodontic instrumentation, is presented in
Table 1. The cross-sectional perimeter percentage of the root canal untouched by the instrument for Hero 642 was significantly lower in comparison to Reciproc R40 (
p = 0.002), indicating a higher contact with dentinal walls of Hero 642 during endodontic instrumentation. The cross-sectional area percentage of the root canal not occupied by the instrument for Hero 642 was also significantly lower in comparison to Reciproc R40 (
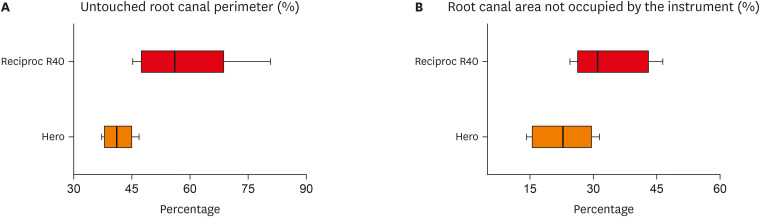
p = 0.002). Box-plot representations of perimeter not touched by instrumentation and area not occupied by the instrument are shown in
Figure 5. Sample normal data distribution showed significant differences for both perimeter and area values.
Table 1
Percentage average of the histological cuts for each molar distal root, regarding perimeter and area not touched during endodontic instrumentation
|
Characteristic |
Perimeter not touched during endodontic instrumentation (%) |
Area not occupied by the instrument during instrumentation (%) |
|
Hero 642 system |
Reciproc R40 |
Hero 642 system |
Reciproc R40 |
|
Distal root*
|
|
|
|
|
|
1 |
37.99 |
45.24 |
15.71 |
26.10 |
|
2 |
44.29 |
52.83 |
14.88 |
24.47 |
|
3 |
46.01 |
48.20 |
31.39 |
30.39 |
|
4 |
44.12 |
48.55 |
21.79 |
42.52 |
|
5 |
43.23 |
59.20 |
29.15 |
44.70 |
|
6 |
37.88 |
80.72 |
14.27 |
28.00 |
|
7 |
37.22 |
67.84 |
23.89 |
26.30 |
|
8 |
38.81 |
45.90 |
30.62 |
46.42 |
|
9 |
38.02 |
67.51 |
25.08 |
31.62 |
|
10 |
46.78 |
70.72 |
21.06 |
42.02 |
|
Average (%) |
41.44a
|
58.67b
|
22.78A
|
34.25B
|
|
Standard deviation |
3.785 |
12.398 |
6.424 |
8.642 |
Figure 5
Box-plot representation of (A) percentual perimeter not touched by instrumentation and (B) areas not occupied by the instrument.
Considering untouched surfaces, the
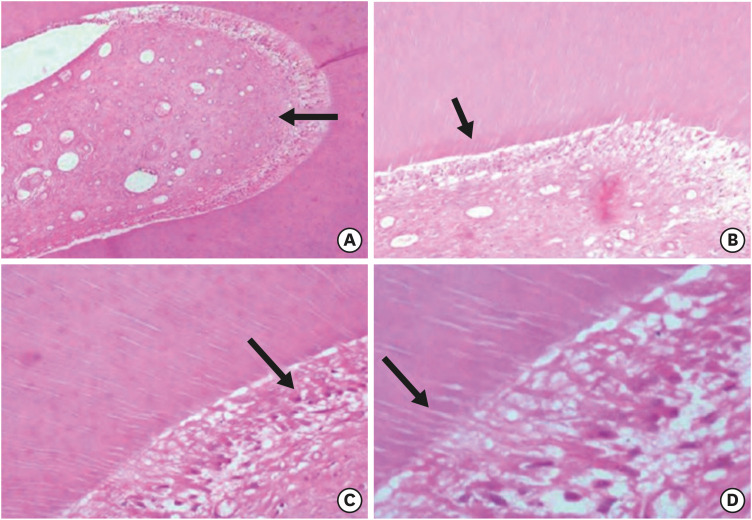
t-test obtained a test power of 81.9%. Whole anatomical structural characteristics were obtained in the control group histological samples (
Figure 6). Regarding the root canal shape evaluated in the control group cross-sections, 80% of the specimens were classified as having a round-shaped root canal, 10% as an oval-shaped root canal, and 10% as a long oval-shaped canal. Due to the single-appointment study characteristics, there was not any patient absence or washout.
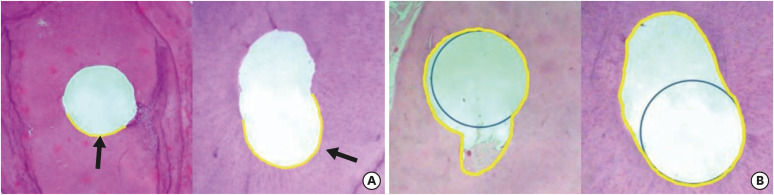
Figure 6
Representative images of the control group (hematoxylin and eosin) without any instrumentation procedure. Arrows are indicating (A) pulp tissue (×50), (B) predentin layer (×100), (C) odontoblast layer (×200), and (D) showing dentin tubules (×400).
DISCUSSION
The main objective of endodontic treatment is to promote root canal system disinfection combining instruments' mechanical action and physical-chemical action of irrigant solutions [
14]. The present study aimed to evaluate the apical pulp tissue removal, using histological analysis, in mandibular molars distal roots after
in vivo preparation using reciprocating and rotary instruments considering similar final foraminal instrumentation diameters. The number of patients included in this study were small and could be interpreted as a limitation of our study. However, the restricted inclusion criteria justified this bias, once only distal mandibular molars presenting vital pulp tissue confirmed both using sensibility test and bleeding were included. Our results must be interpreted carefully regarding the clinical situation once we considered solely the instrument mechanical action without the use of an active irrigation protocol, which is an advantage in order to investigate the role of each root canal therapy step.
In this
in vivo study, we preserved supporting tissues (periodontal ligament, cementum, and alveolar bone) during instrumentation. Unlike in previous
in vitro studies, teeth were only extracted and processed for histological analysis after root canal preparation [
2151617]. The method used for the diagnosis of pulp vitality [
2] enabled standardized control of intra-radicular tissue content, which was confirmed histologically in the control specimens. Besides, aiming to reproduce clinical conditions, instrumentation of the specimens
in vivo also served to preserve dentin structure and hydration, once the loss of water could have bias effects on the mechanical behavior of this structure [
18].
With the evolution of rotary and reciprocating instruments, safer technologies were brought to the root canal preparation. Apical enlargement has presented advantages during root canal instrumentation, such as increased penetration of irrigant solution at the apical level [
19], improved pulp remnants removal, and reduction of non-instrumented areas of the root canal [
2021]. Several studies with different methodologies have assessed the Hero 642 and Reciproc systems [
13622]. Related advantages of reciprocating instruments are the prevention of cross-infection and reduced risk of fracture (due to their single-use nature), along with faster preparation of the root canal [
23].
In a µCT analysis, a previous study [
10] demonstrated the presence of pulp tissue remnants and dentin debris within the apex after instrumentation using Reciproc R40 instruments. In another study, when this critical region was analyzed histologically [
9], the most substantial amount of remaining pulp tissue after instrumentation was found in the apical third. In the present study, the WL was established 1 mm beyond the apex locator reading ‘apex’ in order to even the tested groups' final instrumentation diameter, once they present different tip designs. The present study aimed to evaluate the apical pulp tissue removal inside the root canal using histological analysis, so matching the apical diameter was necessary in order to be able to establish comparisons between different instruments.
Another study using µCT and histologic analysis revealed that instruments with a larger tip size and smaller tapers self-adjusting file prepared significantly more root canal walls than larger-tapered XP-endo Shaper instruments [
9]. This performance might be related to the design of the instrument, but there was no significant difference in the shaping capacity of the 2 systems on histological analysis. Different results were obtained in the present study, where a more considerable amount of debris was found in the apical region histological specimens after preparation using Reciproc (size 40/0.06) (larger taper) group than in those of the Hero 642 (size 45/0.02). These results may be attributed to 2 reasons: first, the larger number of instruments and in-and-out movements used in the Hero 642 system group may be the main reason for our results; second, the different kinematics of rotatory and reciprocating movements during root canal preparation. Although it is not possible to determine precisely the influence of each one separately, these variables combined may justify the different debridement of the root canal apical third of the instruments included in this study. A previous study, using µCT, showed no difference between these kinematics regarding changes in dentin volume or surface area after instrumentation [
8]. Another study comparing kinematics concluded that both rotatory and reciprocating instruments respected the original canal curvature and were safe to use [
3]. Future studies, with other instruments, combining kinematics, and with different methods of analysis, are undoubtedly necessary to the development of an instrument or system of instruments able to touch, clean and shape the largest possible area of the root canal system regardless of anatomic complexity.
The present study was focused exclusively on the action of endodontic instruments in the apical root canal preparation, and its results should be carefully interpreted without extrapolation to the entire root canal instrumentation. A sample selection using cone-beam computed tomography would be an ideal preoperative examination and inclusion criteria, unfortunately for our study, this exam was not available. Thus, the irrigation consisted of using the same volume for both groups of an inert saline solution, unlike other studies that used sodium hypochlorite, a solution that has a solvent effect [
9] on pulp tissues [
21524]. Irrigation solution with solvent effect was avoided in order to evaluate instrumentation outcomes solely. Proper irrigation during root canal preparation is essential to obtain successful endodontic outcomes, especially in the apical region where irrigation is critical in removing debris [
46]. Another critical consideration to take into account is the dissolution of vital pulp expected of the irrigant solution in different concentrations. Further investigation is necessary to obtain better results regarding instruments to be mechanically more active, the observations presented in this study support the fact the neither tested systems provided excellent pulp tissue removal.









 PDF
PDF Citation
Citation Print
Print





 XML Download
XML Download