1. Chen HH, Chao YH, Chen DY, Yang DH, Chung TW, Li YR, Lin CC. 2016; Oral administration of acarbose ameliorates imiquimod-induced psoriasis-like dermatitis in a mouse model. Int Immunopharmacol. 33:70–82. DOI:
10.1016/j.intimp.2016.02.001. PMID:
26874324.

3. Alharbi T, Alzahrani A, Hakami A, Almohammadi A, Alsaadi A, Magbel NB, AlJuaid N, Almasoudi H, Alruwaili T, Alkhezzi S. 2018; Psoriasis pathophysiology and impact on life. Int J Commun Med Public Health. 5:3663–3667. DOI:
10.18203/2394-6040.ijcmph20183383.

4. Conrad C, Gilliet M. 2018; Psoriasis: from pathogenesis to targeted therapies. Clin Rev Allergy Immunol. 54:102–113. DOI:
10.1007/s12016-018-8668-1. PMID:
29349534.

5. Lowes MA, Bowcock AM, Krueger JG. 2007; Pathogenesis and therapy of psoriasis. Nature. 445:866–873. DOI:
10.1038/nature05663. PMID:
17314973.

6. Li Y, Zhang G, Chen M, Tong M, Zhao M, Tang F, Xiao R, Wen H. 2019; Rutaecarpine inhibited imiquimod-induced psoriasis-like dermatitis via inhibiting the NF-κB and TLR7 pathways in mice. Biomed Pharmacother. 109:1876–1883. DOI:
10.1016/j.biopha.2018.10.062. PMID:
30551443.

7. Krueger JG, Ferris LK, Menter A, Wagner F, White A, Visvanathan S, Lalovic B, Aslanyan S, Wang EE, Hall D, Solinger A, Padula S, Scholl P. 2015; Anti-IL-23A mAb BI 655066 for treatment of moderate-to-severe psoriasis: Safety, efficacy, pharmacokinetics, and biomarker results of a single-rising-dose, randomized, double-blind, placebo-controlled trial. J Allergy Clin Immunol. 136:116–124.e7. DOI:
10.1016/j.jaci.2015.01.018. PMID:
25769911.

8. Cline A, Hill D, Lewallen R, Feldman SR. 2016; Current status and future prospects for biologic treatments of psoriasis. Exp Review Clin Immunol. 12:1273–1287. DOI:
10.1080/1744666X.2016.1202115. PMID:
27327580.

9. Talbott W, Duffy N. 2015; Complementary and alternative medicine for psoriasis: what the dermatologist needs to know. Am J Clin Dermatol. 16:147–165. DOI:
10.1007/s40257-015-0128-6. PMID:
25904522.

10. Farahnik B, Sharma D, Alban J, Sivamani RK. 2017; Topical botanical agents for the treatment of psoriasis: a systematic review. Am J Clin Dermatol. 18:451–468. DOI:
10.1007/s40257-017-0266-0. PMID:
28289986.

11. Liu QS, Luo XY, Jiang H, Xing Y, Yang MH, Yuan GH, Tang Z, Wang H. 2015; Salvia miltiorrhiza injection restores apoptosis of fibroblast-like synoviocytes cultured with serum from patients with rheumatoid arthritis. Mol Med Rep. 11:1476–1482. DOI:
10.3892/mmr.2014.2779. PMID:
25352238.

12. Fan Y, Luo Q, Wei J, Lin R, Lin L, Li Y, Chen Z, Lin W, Chen Q. 2018; Mechanism of salvianolic acid B neuroprotection against ischemia/reperfusion induced cerebral injury. Brain Res. 1679:125–133. DOI:
10.1016/j.brainres.2017.11.027. PMID:
29180227.

13. Wei J, Wu J, Xu W, Nie H, Zhou R, Wang R, Liu Y, Tang G, Wu J. 2018; Salvianolic acid B inhibits glycolysis in oral squamous cell carcinoma via targeting PI3K/AKT/HIF-1α signaling pathway. Cell Death Dis. 9:599. DOI:
10.1038/s41419-018-0623-9. PMID:
29789538. PMCID:
PMC5964095.

14. Ma ZG, Xia HQ, Cui SL, Yu J. 2017; Attenuation of renal ischemic reperfusion injury by salvianolic acid B via suppressing oxidative stress and inflammation through PI3K/Akt signaling pathway. Braz J Med Biol Res. 50:e5954. DOI:
10.1590/1414-431x20175954. PMID:
28513773. PMCID:
PMC5479385.

15. Xia ZB, Yuan YJ, Zhang QH, Li H, Dai JL, Min JK. 2018; Salvianolic Acid B Suppresses Inflammatory Mediator Levels by Downregulating NF-κB in a Rat Model of Rheumatoid Arthritis. Med Sci Monit. 24:2524–2532. DOI:
10.12659/MSM.907084. PMID:
29691361. PMCID:
PMC5939601.

16. Chen YS, Lee SM, Lin YJ, Chiang SH, Lin CC. 2014; Effects of Danshensu and Salvianolic Acid B from Salvia miltiorrhiza Bunge (Lamiaceae) on cell proliferation and collagen and melanin production. Molecules. 19:2029–2041. DOI:
10.3390/molecules19022029. PMID:
24531218. PMCID:
PMC6271020.

17. El Malki K, Karbach SH, Huppert J, Zayoud M, Reissig S, Schüler R, Nikolaev A, Karram K, Münzel T, Kuhlmann CR, Luhmann HJ, von Stebut E, Wörtge S, Kurschus FC, Waisman A. 2013; An alternative pathway of imiquimod-induced psoriasis-like skin inflammation in the absence of interleukin-17 receptor a signaling. J Invest Dermatol. 133:441–451. DOI:
10.1038/jid.2012.318. PMID:
22951726.

18. Zhang S, Liu X, Mei L, Wang H, Fang F. 2016; Epigallocatechin-3-gallate (EGCG) inhibits imiquimod-induced psoriasis-like inflammation of BALB/c mice. BMC Complement Altern Med. 16:334. DOI:
10.1186/s12906-016-1325-4. PMID:
27581210. PMCID:
PMC5007807.

19. Zhang Q, Hu LQ, Li HQ, Wu J, Bian NN, Yan G. 2019; Beneficial effects of andrographolide in a rat model of autoimmune myocarditis and its effects on PI3K/Akt pathway. Korean J Physiol Pharmacol. 23:103–111. DOI:
10.4196/kjpp.2019.23.2.103. PMID:
30820154. PMCID:
PMC6384199.

23. Jiang YF, Liu ZQ, Cui W, Zhang WT, Gong JP, Wang XM, Zhang Y, Yang MJ. 2015; Antioxidant effect of salvianolic acid B on hippocampal CA1 neurons in mice with cerebral ischemia and reperfusion injury. Chin J Integr Med. 21:516–522. DOI:
10.1007/s11655-014-1791-1. PMID:
25081897.

24. Zhao GR, Zhang HM, Ye TX, Xiang ZJ, Yuan YJ, Guo ZX, Zhao LB. 2008; Characterization of the radical scavenging and antioxidant activities of danshensu and salvianolic acid B. Food Chem Toxicol. 46:73–81. DOI:
10.1016/j.fct.2007.06.034. PMID:
17719161.

25. Zhang HS, Wang SQ. 2006; Salvianolic acid B from Salvia miltiorrhiza inhibits tumor necrosis factor-alpha (TNF-alpha)-induced MMP-2 upregulation in human aortic smooth muscle cells via suppression of NAD(P)H oxidase-derived reactive oxygen species. J Mol Cell Cardiol. 41:138–148. DOI:
10.1016/j.yjmcc.2006.03.007. PMID:
16713603.
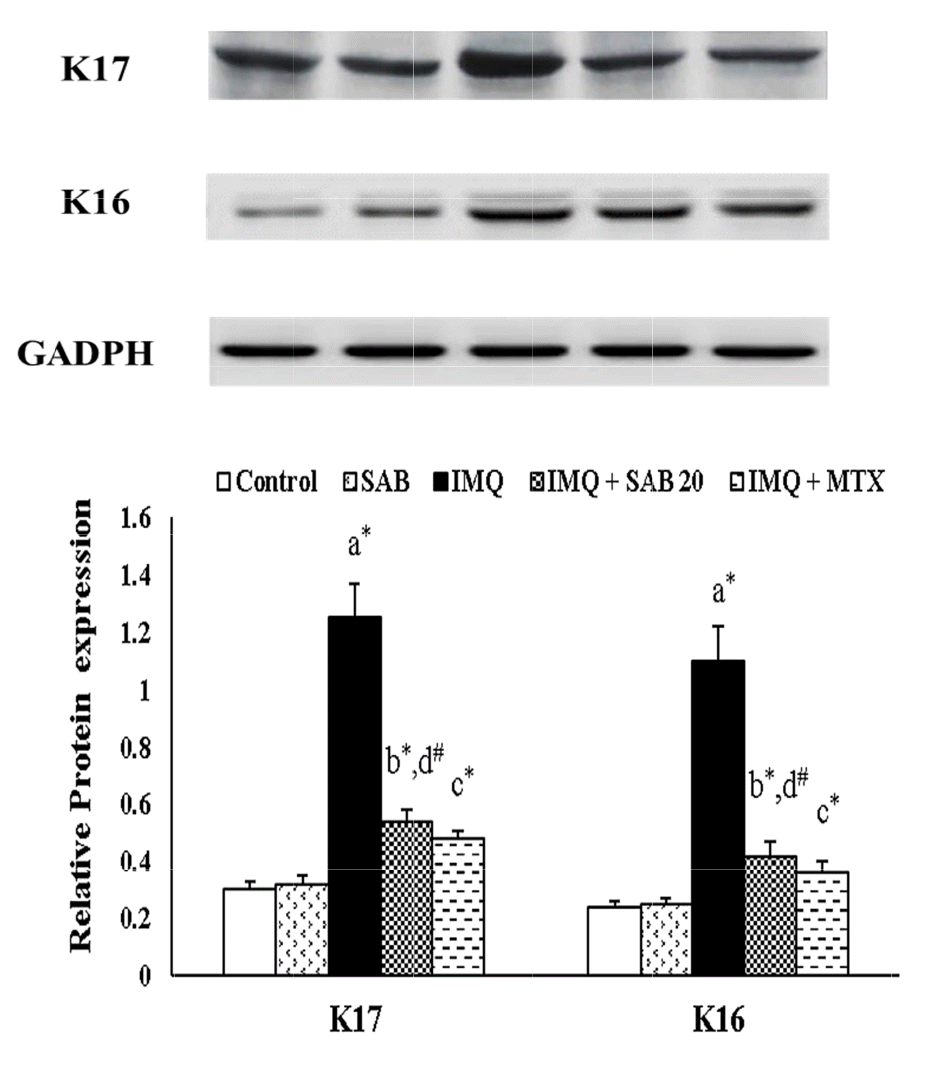
26. Jin L, Wang G. 2014; Keratin 17: a critical player in the pathogenesis of psoriasis. Med Res Rev. 34:438–454. DOI:
10.1002/med.21291. PMID:
23722817.

27. Leigh IM, Navsaria H, Purkis PE, McKay IA, Bowden PE, Riddle PN. 1995; Keratins (K16 and K17) as markers of keratinocyte hyperproliferation in psoriasis in vivo and in vitro. Br J Dermatol. 133:501–511. DOI:
10.1111/j.1365-2133.1995.tb02696.x. PMID:
7577575.
28. Zhang W, Dang E, Shi X, Jin L, Feng Z, Hu L, Wu Y, Wang G. 2012; The pro-inflammatory cytokine IL-22 up-regulates keratin 17 expression in keratinocytes via STAT3 and ERK1/2. PLoS One. 7:e40797. DOI:
10.1371/journal.pone.0040797. PMID:
22808266. PMCID:
PMC3396590.

29. Yang L, Fan X, Cui T, Dang E, Wang G. 2017; Nrf2 promotes keratinocyte proliferation in psoriasis through up-regulation of keratin 6, keratin 16, and keratin 17. J Invest Dermatol. 137:2168–2176. DOI:
10.1016/j.jid.2017.05.015. PMID:
28576737.

30. Du ZC, Xue T, Jiang M, Lu HY, Ye ZC, Ruan BJ, Xu CM, Jiang YH, Wei M, Wang G, Lu ZF, Lei XY, Wang L. 2016; Arctigenin attenuates imiquimod-induced psoriasis-like skin lesions via down-regulating keratin17. Int J Clin Exp Med. 9:1639–1647.
31. Zhang J, Li X, Wei J, Chen H, Lu Y, Li L, Han L, Lu C. 2018; Gallic acid inhibits the expression of keratin 16 and keratin 17 through Nrf2 in psoriasis-like skin disease. Int Immunopharmacol. 65:84–95. DOI:
10.1016/j.intimp.2018.09.048. PMID:
30293051.

32. Dogra S, Mahajan R. 2013; Systemic methotrexate therapy for psoriasis: past, present and future. Clin Exp Dermatol. 38:573–588. DOI:
10.1111/ced.12062. PMID:
23837932.

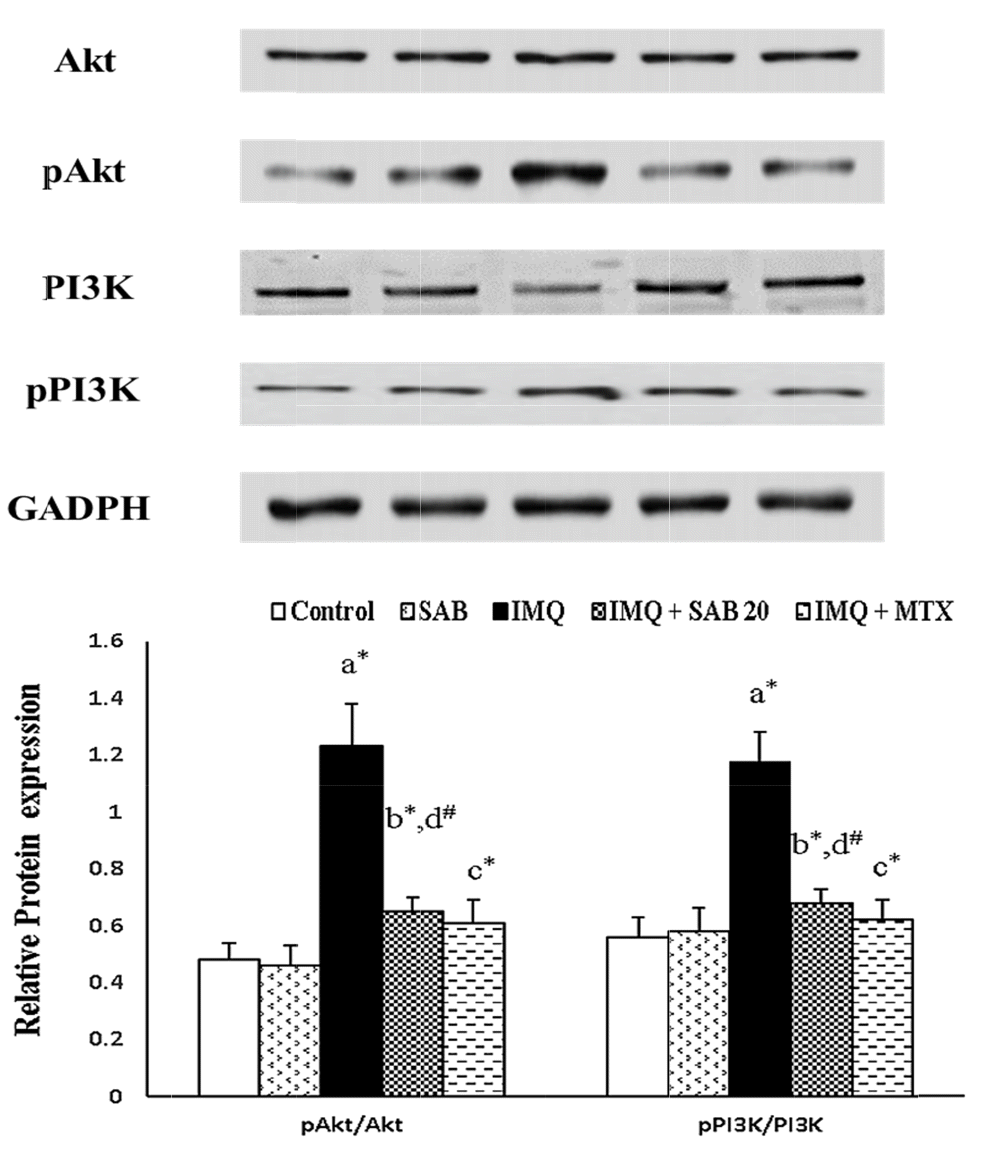
34. Chamcheu JC, Adhami VM, Esnault S, Sechi M, Siddiqui IA, Satyshur KA, Syed DN, Dodwad SM, Chaves-Rodriquez MI, Longley BJ, Wood GS, Mukhtar H. 2017; Dual inhibition of PI3K/Akt and mTOR by the dietary antioxidant, delphinidin, ameliorates psoriatic features in vitro and in an imiquimod-induced psoriasis-like disease in mice. Antioxid Redox Signal. 26:49–69. DOI:
10.1089/ars.2016.6769. PMID:
27393705. PMCID:
PMC5206770.

35. Chamcheu JC, Chaves-Rodriquez MI, Adhami VM, Siddiqui IA, Wood GS, Longley BJ, Mukhtar H. 2016; Upregulation of PI3K/AKT/mTOR, FABP5 and PPARβ/δ in human psoriasis and imiquimod-induced murine psoriasiform dermatitis model. Acta Derm Venereol. 96:854–856. DOI:
10.2340/00015555-2359. PMID:
26833029. PMCID:
PMC5540143.





 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download