1. Maruhashi T, Soga J, Fujimura N, Idei N, Mikami S, Iwamoto Y, Iwamoto A, Kajikawa M, Matsumoto T, Oda N, Kishimoto S, Matsui S, Hashimoto H, Aibara Y, Mohamad Yusoff F, Hidaka T, Kihara Y, Chayama K, Noma K, Nakashima A, Goto C, Tomiyama H, Takase B, Kohro T, Suzuki T, Ishizu T, Ueda S, Yamazaki T, Furumoto T, Kario K, Inoue T, Koba S, Watanabe K, Takemoto Y, Hano T, Sata M, Ishibashi Y, Node K, Maemura K, Ohya Y, Furukawa T, Ito H, Ikeda H, Yamashina A, Higashi Y. Endothelial dysfunction, increased arterial stiffness, and cardiovascular risk prediction in patients with coronary artery disease: FMD-J (flow-mediated dilation Japan) study A. J Am Heart Assoc. 2018; 7:e008588. PMID:
30005558.

2. Anderson TJ. Arterial stiffness or endothelial dysfunction as a surrogate marker of vascular risk. Can J Cardiol. 2006; 22(Suppl B):72B–80B.

3. Santos-Parker JR, Strahler TR, Bassett CJ, Bispham NZ, Chonchol MB, Seals DR. Curcumin supplementation improves vascular endothelial function in healthy middle-aged and older adults by increasing nitric oxide bioavailability and reducing oxidative stress. Aging (Albany NY). 2017; 9:187–208. PMID:
28070018.

4. Magda SL, Ciobanu AO, Florescu M, Vinereanu D. Comparative reproducibility of the noninvasive ultrasound methods for the assessment of vascular function. Heart Vessels. 2013; 28:143–150. PMID:
22241737.

5. Kajikawa M, Maruhashi T, Hida E, Iwamoto Y, Matsumoto T, Iwamoto A, Oda N, Kishimoto S, Matsui S, Hidaka T, Kihara Y, Chayama K, Goto C, Aibara Y, Nakashima A, Noma K, Higashi Y. Combination of flow-mediated vasodilation and nitroglycerine-induced vasodilation is more effective for prediction of cardiovascular events. Hypertension. 2016; 67:1045–1052. PMID:
26975705.

6. Tomiyama H, Yamashina A. Non-invasive vascular function tests: their pathophysiological background and clinical application. Circ J. 2010; 74:24–33. PMID:
19920359.
7. Cardoso SM, Pereira OR, Seca AM, Pinto DC, Silva AM. Seaweeds as preventive agents for cardiovascular diseases: from nutrients to functional foods. Mar Drugs. 2015; 13:6838–6865. PMID:
26569268.

8. Alissa EM, Ferns GA. Functional foods and nutraceuticals in the primary prevention of cardiovascular diseases. J Nutr Metab. 2012; 2012:569486. PMID:
22570771.

9. Sikand G, Kris-Etherton P, Boulos NM. Impact of functional foods on prevention of cardiovascular disease and diabetes. Curr Cardiol Rep. 2015; 17:39. PMID:
25899657.

10. Kim YH, Cho ML, Kim DB, Shin GH, Lee JH, Lee JS, Park SO, Lee SJ, Shin HM, Lee OH. The antioxidant activity and their major antioxidant compounds from Acanthopanax senticosus and A. koreanum. Molecules. 2015; 20:13281–13295. PMID:
26205054.

11. Davydov M, Krikorian AD. Eleutherococcus senticosus (Rupr. & Maxim.) Maxim. (Araliaceae) as an adaptogen: a closer look. J Ethnopharmacol. 2000; 72:345–393. PMID:
10996277.
12. Han Y, Zhang A, Sun H, Zhang Y, Meng X, Yan G, Liu L, Wang X. High-throughput ultra high performance liquid chromatography combined with mass spectrometry approach for the rapid analysis and characterization of multiple constituents of the fruit of Acanthopanax senticosus (Rupr. et Maxim.) Harms. J Sep Sci. 2017; 40:2178–2187. PMID:
28371133.
13. Saito T, Nishida M, Saito M, Tanabe A, Eitsuka T, Yuan SH, Ikekawa N, Nishida H. The fruit of Acanthopanax senticosus (Rupr. et Maxim.) Harms improves insulin resistance and hepatic lipid accumulation by modulation of liver adenosine monophosphate-activated protein kinase activity and lipogenic gene expression in high-fat diet-fed obese mice. Nutr Res. 2016; 36:1090–1097. PMID:
27865350.

14. Fujikawa T, Yamaguchi A, Morita I, Takeda H, Nishibe S. Protective effects of Acanthopanax senticosus Harms from Hokkaido and its components on gastric ulcer in restrained cold water stressed rats. Biol Pharm Bull. 1996; 19:1227–1230. PMID:
8889047.

15. Wang X, Hai CX, Liang X, Yu SX, Zhang W, Li YL. The protective effects of Acanthopanax senticosus Harms aqueous extracts against oxidative stress: role of Nrf2 and antioxidant enzymes. J Ethnopharmacol. 2010; 127:424–432. PMID:
19857565.

16. Yi JM, Hong SH, Kim JH, Kim HK, Song HJ, Kim HM. Effect of Acanthopanax senticosus stem on mast cell-dependent anaphylaxis. J Ethnopharmacol. 2002; 79:347–352. PMID:
11849840.

17. Jung HJ, Park HJ, Kim RG, Shin KM, Ha J, Choi JW, Kim HJ, Lee YS, Lee KT. In vivo anti-inflammatory and antinociceptive effects of liriodendrin isolated from the stem bark of Acanthopanax senticosus. Planta Med. 2003; 69:610–616. PMID:
12898415.
18. Lee S, Shin DS, Oh KB, Shin KH. Antibacterial compounds from the leaves of Acanthopanax senticosus. Arch Pharm Res. 2003; 26:40–42. PMID:
12568356.
19. Zhou AY, Song BW, Fu CY, Baranenko DD, Wang EJ, Li FY, Lu GW. Acanthopanax senticosus reduces brain injury in mice exposed to low linear energy transfer radiation. Biomed Pharmacother. 2018; 99:781–790. PMID:
29710476.

20. Bang JS, Chung YH, Chung SJ, Lee HS, Song EH, Shin YK, Lee YJ, Kim HC, Nam Y, Jeong JH. Clinical effect of a polysaccharide-rich extract of Acanthopanax senticosus on alcohol hangover. Pharmazie. 2015; 70:269–273. PMID:
26012258.
21. Lee YJ, Chung HY, Kwak HK, Yoon S. The effects of A. senticosus supplementation on serum lipid profiles, biomarkers of oxidative stress, and lymphocyte DNA damage in postmenopausal women. Biochem Biophys Res Commun. 2008; 375:44–48. PMID:
18671947.

22. Hwang YC, Jeong IK, Ahn KJ, Chung HY. The effects of Acanthopanax senticosus extract on bone turnover and bone mineral density in Korean postmenopausal women. J Bone Miner Metab. 2009; 27:584–590. PMID:
19452124.

23. Engler MB, Engler MM, Chen CY, Malloy MJ, Browne A, Chiu EY, Kwak HK, Milbury P, Paul SM, Blumberg J, Mietus-Snyder ML. Flavonoid-rich dark chocolate improves endothelial function and increases plasma epicatechin concentrations in healthy adults. J Am Coll Nutr. 2004; 23:197–204. PMID:
15190043.

24. Bucholz EM, Gooding HC, de Ferranti SD. Awareness of cardiovascular risk factors in U.S. young adults aged 18–39 years. Am J Prev Med. 2018; 54:e67–77. PMID:
29433955.

25. Nelson RH. Hyperlipidemia as a risk factor for cardiovascular disease. Prim Care. 2013; 40:195–211. PMID:
23402469.

26. Niva M. ‘All foods affect health’: understandings of functional foods and healthy eating among health-oriented Finns. Appetite. 2007; 48:384–393. PMID:
17166625.

27. Lim Y, Song TJ, Hwang W, Kim JY, Lee D, Kim YJ, Kwon O. Synergistic effects of Sanghuang-Danshen bioactives on arterial stiffness in a randomized clinical trial of healthy smokers: an integrative approach to in silico network analysis. Nutrients. 2019; 11:11.

28. Figueroa A, Wong A, Hooshmand S, Sanchez-Gonzalez MA. Effects of watermelon supplementation on arterial stiffness and wave reflection amplitude in postmenopausal women. Menopause. 2013; 20:573–577. PMID:
23615650.

29. Mucalo I, Jovanovski E, Rahelić D, Božikov V, Romić Z, Vuksan V. Effect of American ginseng (Panax quinquefolius L.) on arterial stiffness in subjects with type-2 diabetes and concomitant hypertension. J Ethnopharmacol. 2013; 150:148–153. PMID:
23973636.

30. Johnson SA, Figueroa A, Navaei N, Wong A, Kalfon R, Ormsbee LT, Feresin RG, Elam ML, Hooshmand S, Payton ME, Arjmandi BH. Daily blueberry consumption improves blood pressure and arterial stiffness in postmenopausal women with pre- and stage 1-hypertension: a randomized, double-blind, placebo-controlled clinical trial. J Acad Nutr Diet. 2015; 115:369–377. PMID:
25578927.

31. Avolio A. Arterial stiffness. Pulse (Basel). 2013; 1:14–28. PMID:
26587425.

32. Otsuka K, Fukuda S, Shimada K, Suzuki K, Nakanishi K, Yoshiyama M, Yoshikawa J. Serial assessment of arterial stiffness by cardio-ankle vascular index for prediction of future cardiovascular events in patients with coronary artery disease. Hypertens Res. 2014; 37:1014–1020. PMID:
25007768.

33. Laurent S, Boutouyrie P, Asmar R, Gautier I, Laloux B, Guize L, Ducimetiere P, Benetos A. Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension. 2001; 37:1236–1241. PMID:
11358934.

34. Sugawara J, Hayashi K, Yokoi T, Cortez-Cooper MY, DeVan AE, Anton MA, Tanaka H. Brachial-ankle pulse wave velocity: an index of central arterial stiffness? J Hum Hypertens. 2005; 19:401–406. PMID:
15729378.

35. Tsuchikura S, Shoji T, Kimoto E, Shinohara K, Hatsuda S, Koyama H, Emoto M, Nishizawa Y. Brachial-ankle pulse wave velocity as an index of central arterial stiffness. J Atheroscler Thromb. 2010; 17:658–665. PMID:
20467192.

36. Hung CS, Lin JW, Hsu CN, Chen HM, Tsai RY, Chien YF, Hwang JJ. Using brachial-ankle pulse wave velocity to associate arterial stiffness with cardiovascular risks. Nutr Metab Cardiovasc Dis. 2009; 19:241–246. PMID:
18815016.

37. Koyama N, Suzuki K, Furukawa Y, Arisaka H, Seki T, Kuribayashi K, Ishii K, Sukegawa E, Takahashi M. Effects of safflower seed extract supplementation on oxidation and cardiovascular risk markers in healthy human volunteers. Br J Nutr. 2009; 101:568–575. PMID:
18590590.

38. Tomiyama H, Kohro T, Higashi Y, Takase B, Suzuki T, Ishizu T, Ueda S, Yamazaki T, Furumoto T, Kario K, Inoue T, Koba S, Watanabe K, Takemoto Y, Hano T, Sata M, Ishibashi Y, Node K, Maemura K, Ohya Y, Furukawa T, Ito H, Ikeda H, Yamashina A. Reliability of measurement of endothelial function across multiple institutions and establishment of reference values in Japanese. Atherosclerosis. 2015; 242:433–442. PMID:
26291496.

39. Incalza MA, D'Oria R, Natalicchio A, Perrini S, Laviola L, Giorgino F. Oxidative stress and reactive oxygen species in endothelial dysfunction associated with cardiovascular and metabolic diseases. Vascul Pharmacol. 2018; 100:1–19. PMID:
28579545.

40. Lessiani G, Santilli F, Boccatonda A, Iodice P, Liani R, Tripaldi R, Saggini R, Davì G. Arterial stiffness and sedentary lifestyle: role of oxidative stress. Vascul Pharmacol. 2016; 79:1–5. PMID:
26044182.

41. Siti HN, Kamisah Y, Kamsiah J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vascul Pharmacol. 2015; 71:40–56. PMID:
25869516.

42. Kolluru GK, Siamwala JH, Chatterjee S. eNOS phosphorylation in health and disease. Biochimie. 2010; 92:1186–1198. PMID:
20363286.

43. Fukada K, Kajiya-Sawane M, Matsumoto Y, Hasegawa T, Fukaya Y, Kajiya K. Antiedema effects of Siberian ginseng in humans and its molecular mechanism of lymphatic vascular function in vitro. Nutr Res. 2016; 36:689–695. PMID:
27333960.

44. Chung BH, Kim S, Kim JD, Lee JJ, Baek YY, Jeoung D, Lee H, Choe J, Ha KS, Won MH, Kwon YG, Kim YM. Syringaresinol causes vasorelaxation by elevating nitric oxide production through the phosphorylation and dimerization of endothelial nitric oxide synthase. Exp Mol Med. 2012; 44:191–201. PMID:
22170035.

45. Efferth T, Koch E. Complex interactions between phytochemicals. The multi-target therapeutic concept of phytotherapy. Curr Drug Targets. 2011; 12:122–132. PMID:
20735354.

46. Calabrese EJ, Calabrese V, Tsatsakis A, Giordano JJ. Hormesis and Ginkgo biloba (GB): numerous biological effects of GB are mediated via hormesis. Ageing Res Rev.

47. Calabrese V, Santoro A, Trovato Salinaro A, Modafferi S, Scuto M, Albouchi F, Monti D, Giordano J, Zappia M, Franceschi C, Calabrese EJ. Hormetic approaches to the treatment of Parkinson's disease: perspectives and possibilities. J Neurosci Res. 2018; 96:1641–1662. PMID:
30098077.

48. Ozturk K, Guler AK, Cakir M, Ozen A, Demirci H, Turker T, Demirbas S, Uygun A, Gulsen M, Bagci S. Pulse wave velocity, intima media thickness, and flow-mediated dilatation in patients with normotensive normoglycemic inflammatory bowel disease. Inflamm Bowel Dis. 2015; 21:1314–1320. PMID:
25803507.

49. EUGenMed Cardiovascular Clinical Study Group. Regitz-Zagrosek V, Oertelt-Prigione S, Prescott E, Franconi F, Gerdts E, Foryst-Ludwig A, Maas AH, Kautzky-Willer A, Knappe-Wegner D, Kintscher U, Ladwig KH, Schenck-Gustafsson K, Stangl V. Gender in cardiovascular diseases: impact on clinical manifestations, management, and outcomes. Eur Heart J. 2016; 37:24–34. PMID:
26530104.

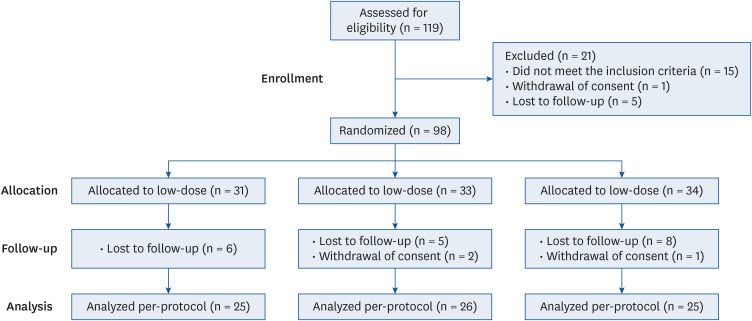
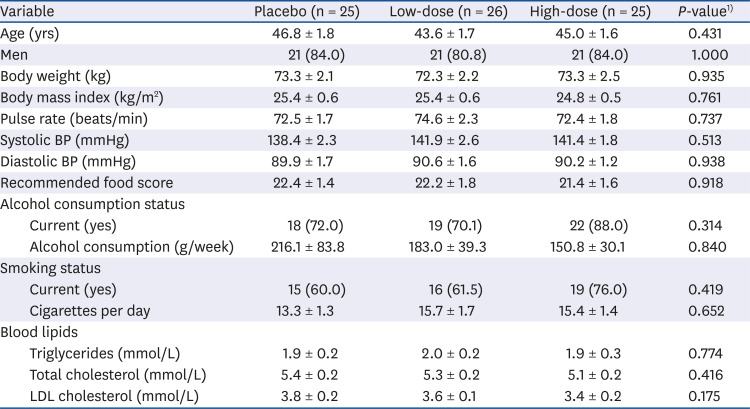
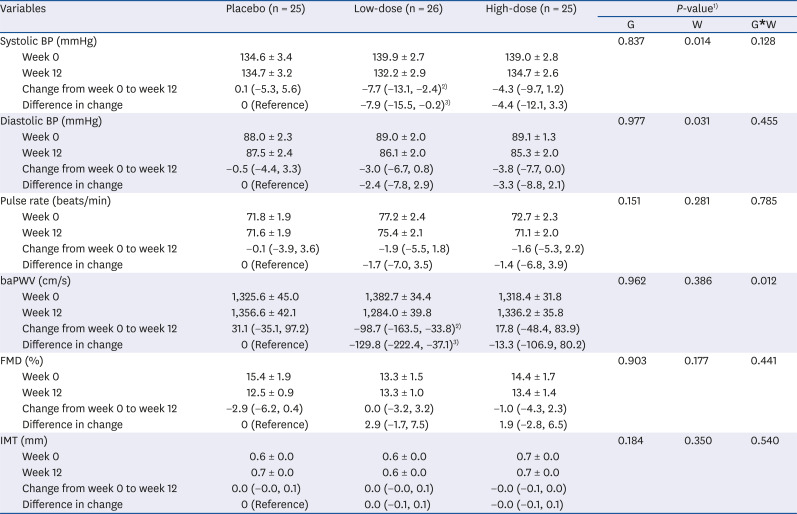
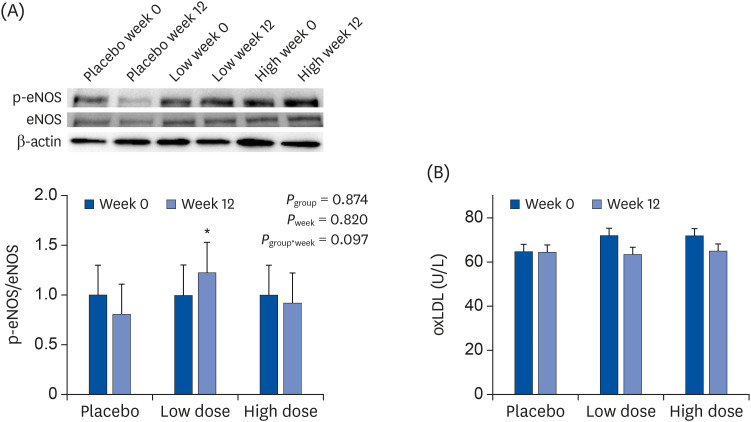




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download