1. Dare AJ, Bartlett AS, Fraser JF. 2012; Critical care of the potential organ donor. Curr Neurol Neurosci Rep. 12:456–65. DOI:
10.1007/s11910-012-0272-9. PMID:
22618126.

3. Souter MJ, Eidbo E, Findlay JY, Lebovitz DJ, Moguilevitch M, Neidlinger NA, et al. 2018; Organ donor management: part 1. toward a consensus to guide anesthesia services during donation after brain death. Semin Cardiothorac Vasc Anesth. 22:211–22. DOI:
10.1177/1089253217749053. PMID:
29276852.

4. van Erp AC, van Dullemen LF, Ploeg RJ, Leuvenink HG. 2018; Systematic review on the treatment of deceased organ donors. Transplant Rev (Orlando). 32:194–206. DOI:
10.1016/j.trre.2018.06.001. PMID:
30049604.

5. Chamorro-Jambrina C, Muñoz-Ramírez MR, Martínez- Melgar JL, Pérez-Cornejo MS. 2017; Organ donor management: eight common recommendations and actions that deserve reflection. Med Intensiva. 41:559–68. DOI:
10.1016/j.medin.2017.01.012. PMID:
28318674.

6. Chudoba P, Krajewski W, Wojciechowska J, Kamińska D. 2017; Brain death-associated pathological events and therapeutic options. Adv Clin Exp Med. 26:1457–64. DOI:
10.17219/acem/65068. PMID:
29442469.

8. Berthelsen PG, Marik PE. 2015; Therapeutic hypothermia in deceased organ donors and kidney-graft function. N Engl J Med. 373:2686. DOI:
10.1056/NEJMc1511744. PMID:
27186763.

9. Miñambres E, Pérez-Villares JM, Chico-Fernández M, Zabalegui A, Dueñas-Jurado JM, Misis M, et al. 2015; Lung donor treatment protocol in brain dead-donors: a multicenter study. J Heart Lung Transplant. 34:773–80. DOI:
10.1016/j.healun.2014.09.024. PMID:
25447580. PMCID:
PMC6988327.

10. Nguyen DC, Loor G, Carrott P, Shafii A. 2019; Review of donor and recipient surgical procedures in lung transplantation. J Thorac Dis. 11(Suppl 14):S1810–6. DOI:
10.21037/jtd.2019.06.31. PMID:
31632758. PMCID:
PMC6783720.

12. de Perrot M, Fischer S, Liu M, Jin R, Bai XH, Waddell TK, et al. 2001; Prostaglandin E1 protects lung transplants from ischemia-reperfusion injury: a shift from pro- to anti-inflammatory cytokines. Transplantation. 72:1505–12. DOI:
10.1097/00007890-200111150-00006. PMID:
11707737.
13. Oto T, Rabinov M, Negri J, Marasco S, Rowland M, Pick A, et al. 2006; Techniques of reconstruction for inadequate donor left atrial cuff in lung transplantation. Ann Thorac Surg. 81:1199–204. DOI:
10.1016/j.athoracsur.2005.11.057. PMID:
16564243.

14. Slama A, Schillab L, Barta M, Benedek A, Mitterbauer A, Hoetzenecker K, et al. 2017; Standard donor lung procurement with normothermic ex vivo lung perfusion: a prospective randomized clinical trial. J Heart Lung Transplant. 36:744–53. DOI:
10.1016/j.healun.2017.02.011. PMID:
28314503.

16. Yu HC, Cho BH. 2006; How to do I make an organ procurement in deceased donor? J Korean Soc Transplant. 20:14–24.
18. Keutgen XM, Petrowsky H. 2014; Procurement for visceral organ transplantation: where to cannulate and how to perfuse? Curr Opin Organ Transplant. 19:92–9. DOI:
10.1097/MOT.0000000000000066. PMID:
24565959.
19. Makowka L, Stieber AC, Sher L, Kahn D, Mieles L, Bowman J, et al. 1988; Surgical technique of orthotopic liver transplantation. Gastroenterol Clin North Am. 17:33–51.

20. Nickkholgh A, Contin P, Abu-Elmagd K, Golriz M, Gotthardt D, Morath C, et al. 2013; Intestinal transplantation: review of operative techniques. Clin Transplant. 27 Suppl 25:56–65. DOI:
10.1111/ctr.12190. PMID:
23909503.

21. Wunderlich H, Brockmann JG, Voigt R, Rauchfuss F, Pascher A, Brose S, et al. 2011; DTG procurement guidelines in heart beating donors. Transpl Int. 24:733–57. DOI:
10.1111/j.1432-2277.2011.01266.x. PMID:
21668528.

23. Yersiz H, Renz JF, Hisatake GM, Gordon S, Saggi BH, Feduska NJ Jr, et al. 2003; Multivisceral and isolated intestinal procurement techniques. Liver Transpl. 9:881–6. DOI:
10.1053/jlts.2003.50155. PMID:
12884206. PMCID:
PMC6445255.

24. Hakim NS SR, Gray D, Friend P, Colman A. 2010. Pancreas, islet and stem cell transplantation for diabetes. 2nd ed. Oxford University Press;Oxford, UK: DOI:
10.1093/med/9780199565863.001.0001.
25. Yang HC, Gifford RR, Dafoe DC, Neumyer MM, Thiele BL. 1991; Arterial reconstruction of the pancreatic allograft for transplantation. Am J Surg. 162:262–4. DOI:
10.1016/0002-9610(91)90083-P. PMID:
1928589.

26. Morris PJ, Tilney NL, editors. PM. 1984. Techniques of multiple organ harvesting. Progress in transplantation. Edinburgh. Churchill Livingstone;burgh, UK:
27. Knechtle S, Morris P. 2008. Kidney transplantation. 6th ed. Saunders Elsevier;Philadelphia, PA:




 PDF
PDF Citation
Citation Print
Print




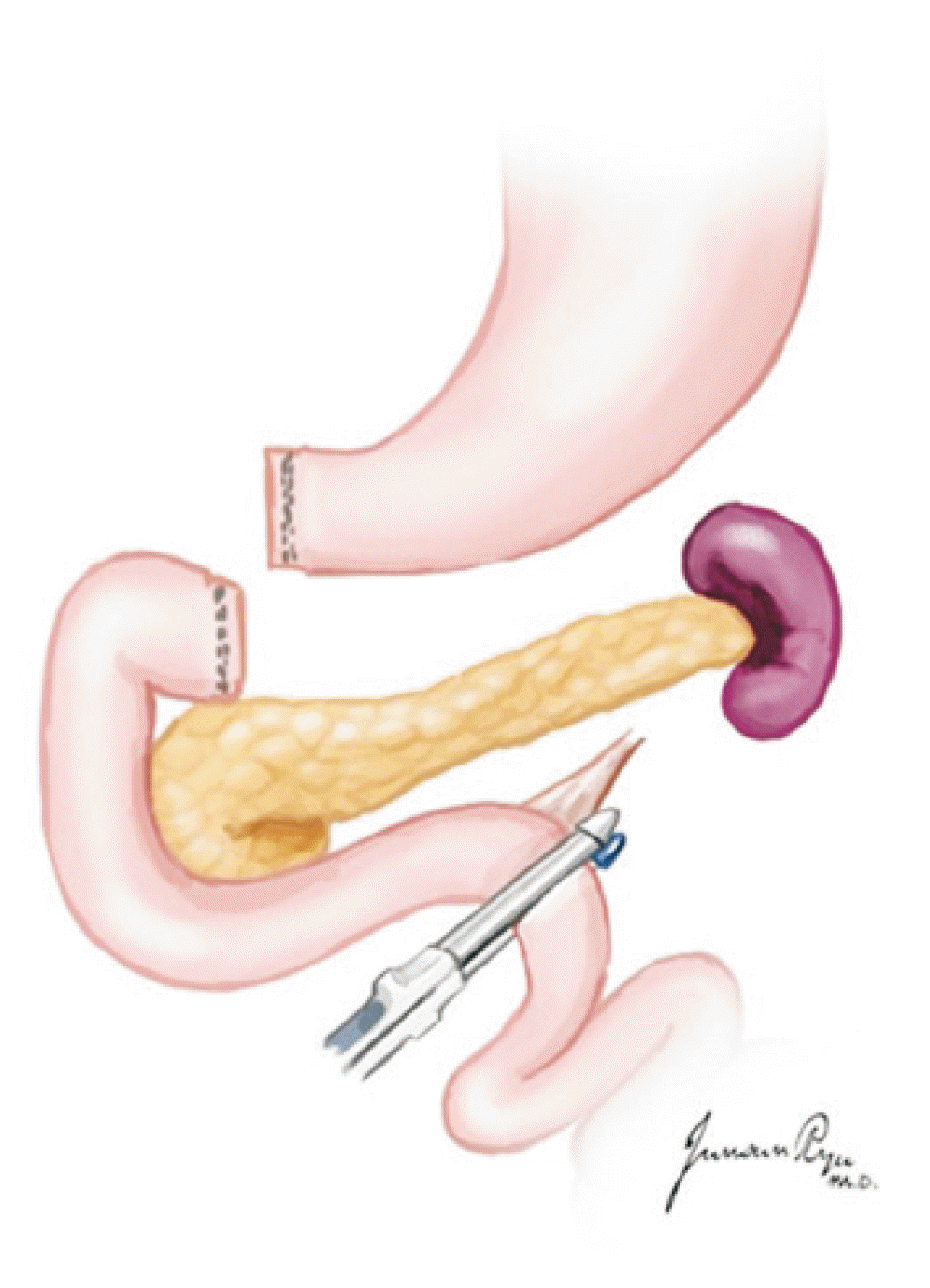
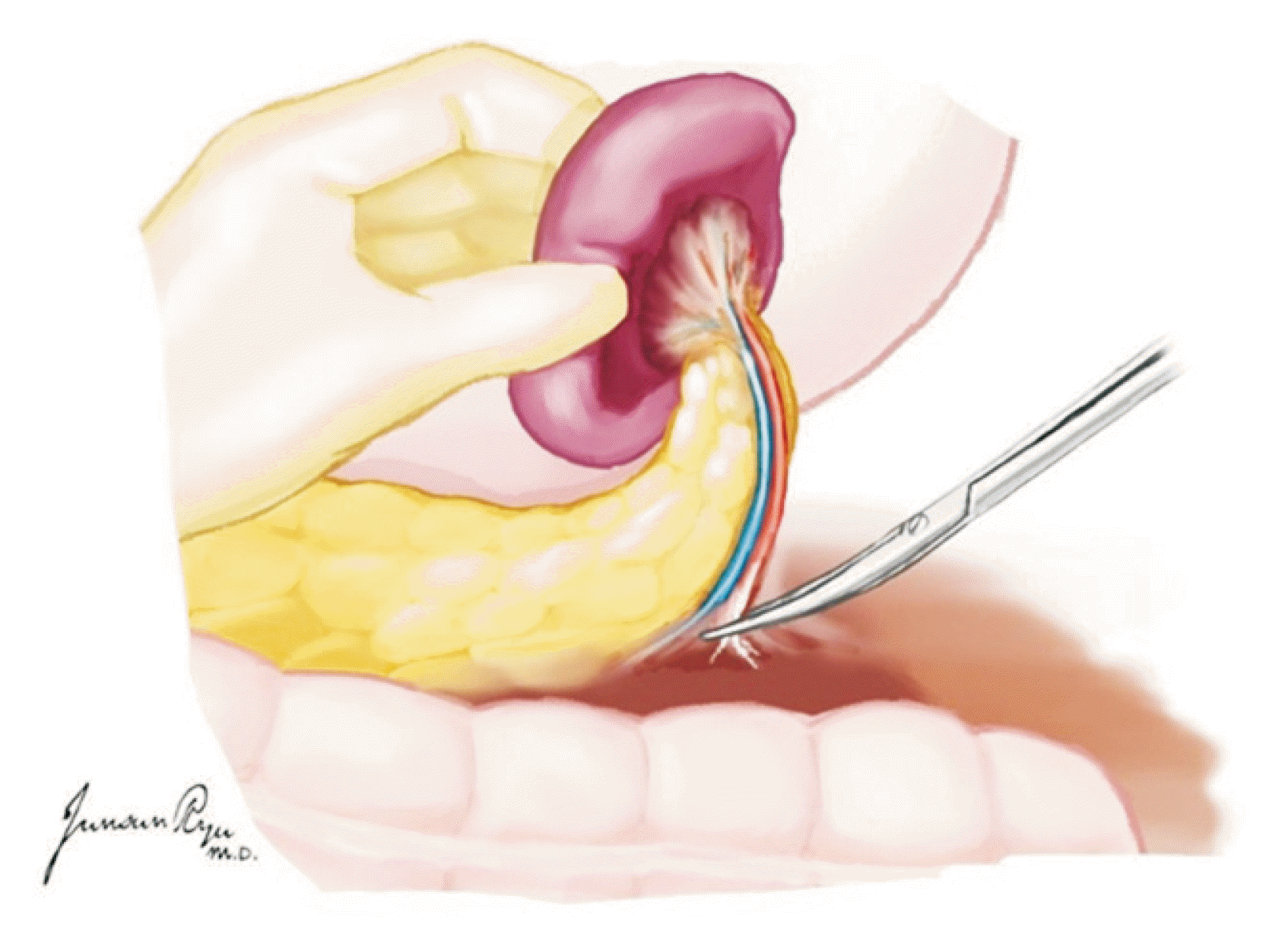
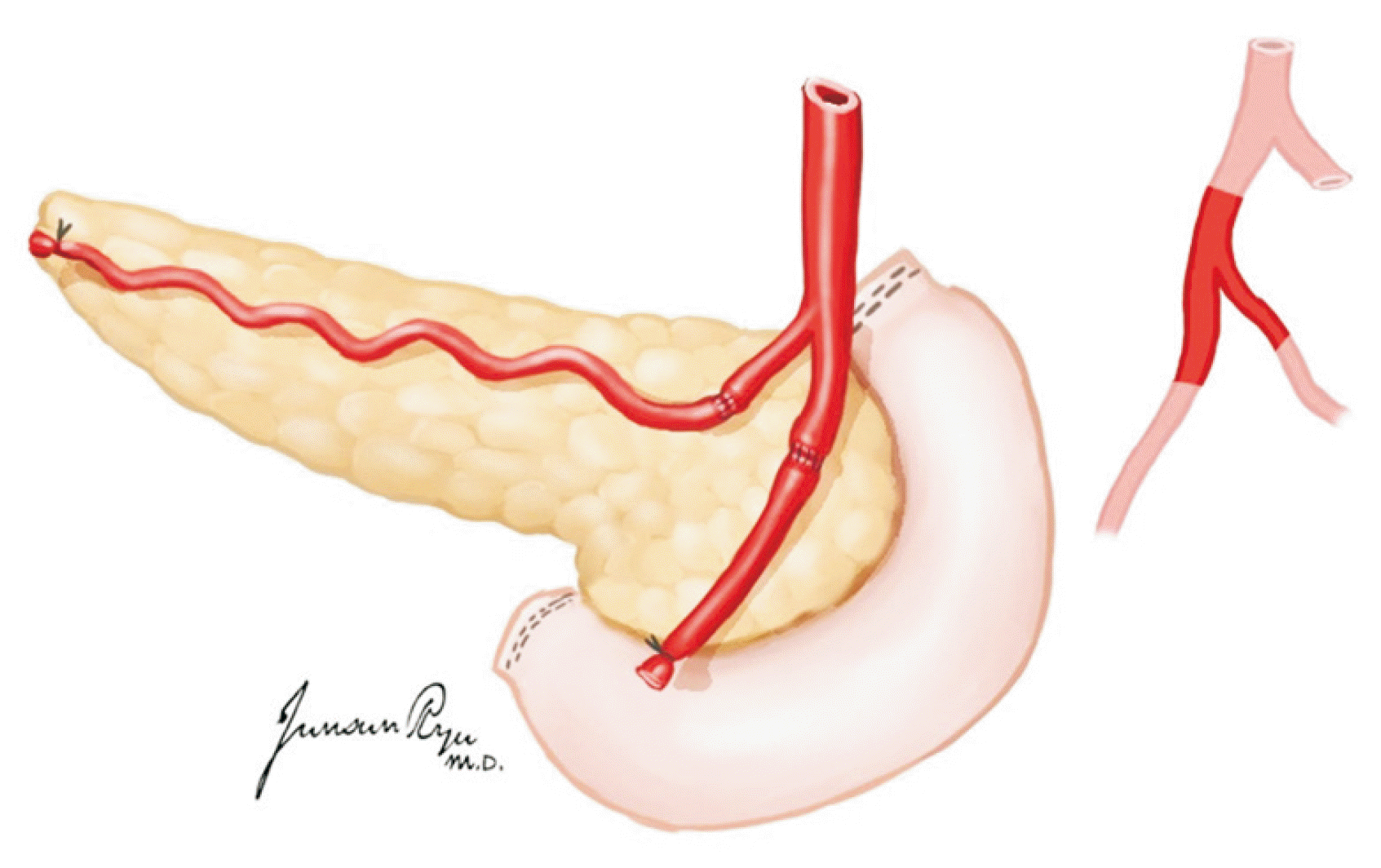
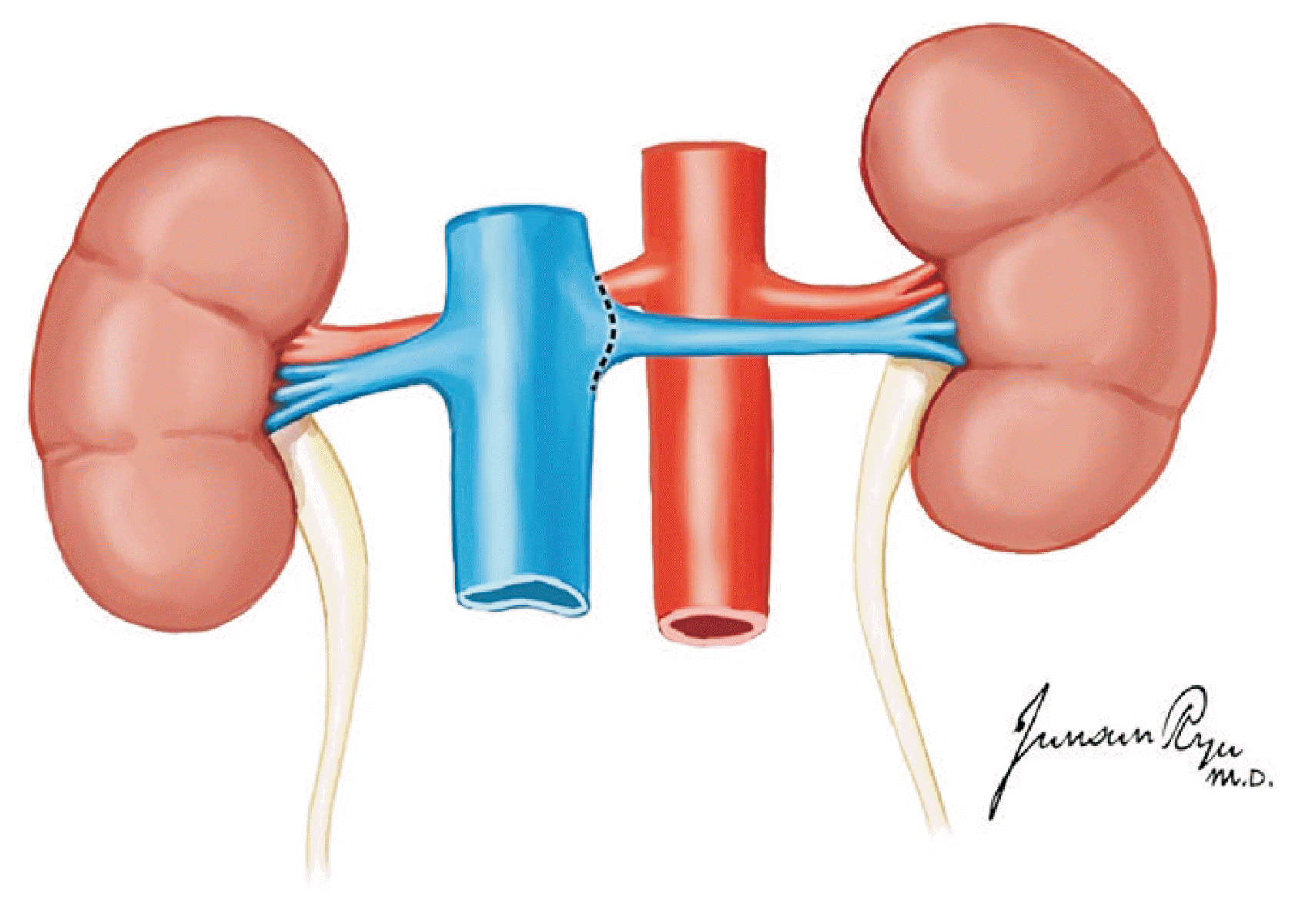
 XML Download
XML Download