Introduction
Methods
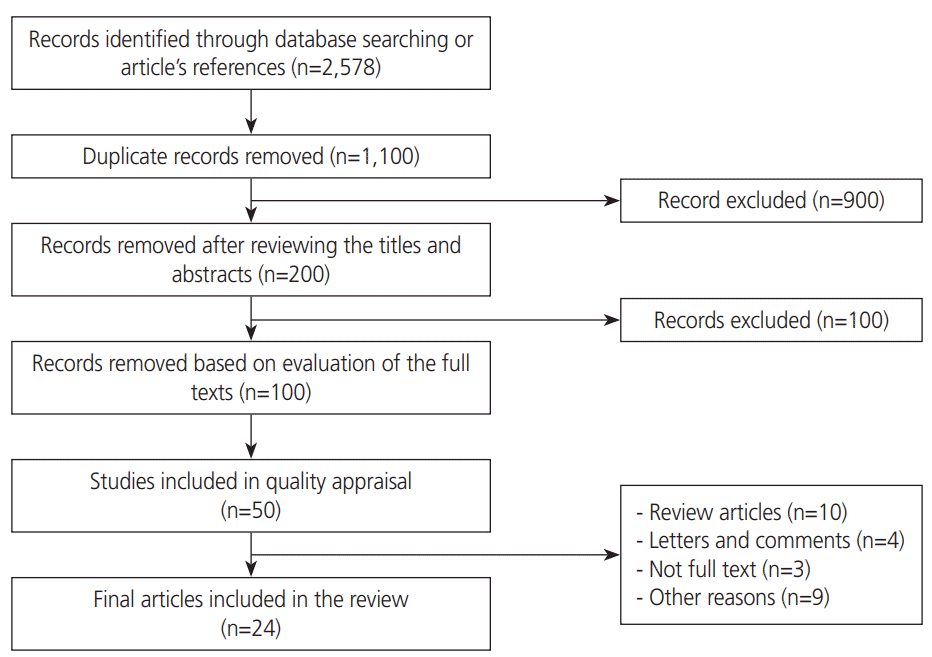
1. Search strategy
Table 1.
2. Inclusion and exclusion criteria
3. Study selection
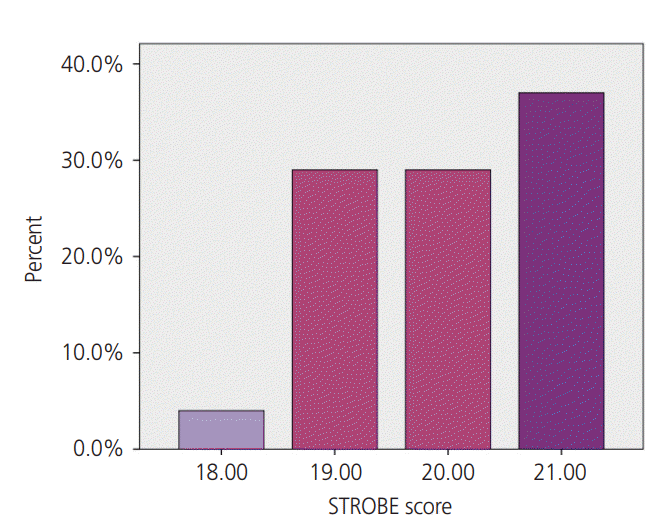
5. Data extraction
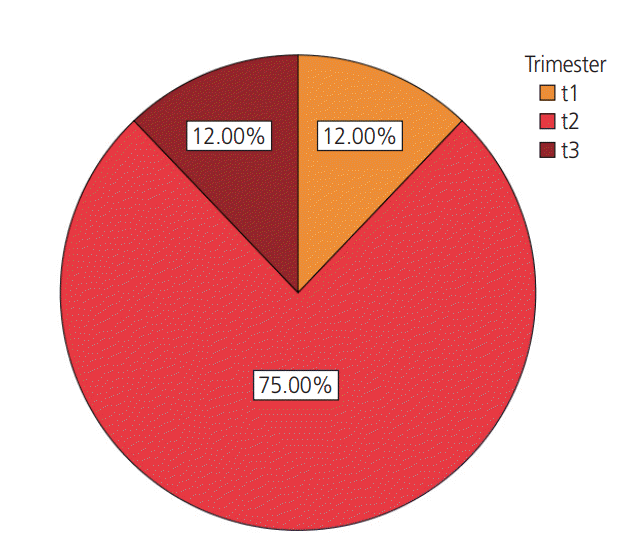
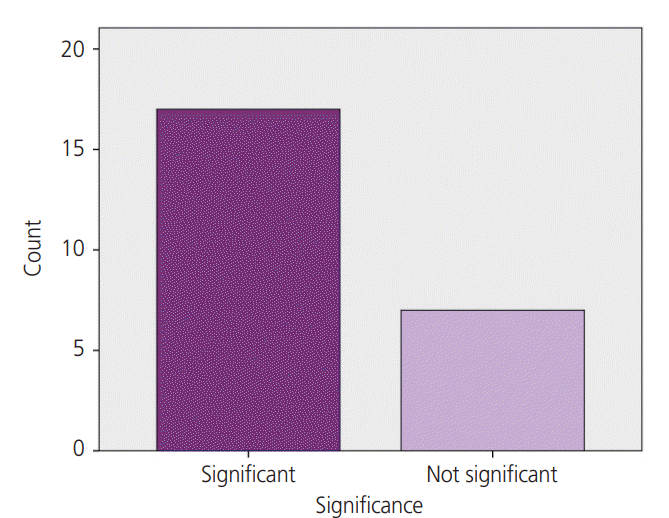
Results
Table 2.
| Author | Geographic region | Study design |
GDM group |
Control group |
Sample | Time of collect test (wk) | Method of analysis test | Diagnostic criteria of GDM |
GDM group |
Control group |
Results (P-value) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Participants | BMI | Participants | BMI | IL-6 | IL-6 | ||||||||
| Wang et al. [21] | China | Case-control | 60 | NR | 50 | NR | Serum | 24–28 | ELISA | ADA | 96.66 (88.33–106.66)a) | 83.32 (78.34–88.33)a) | Significant (P<0.001) |
| Braga et al. [17] | Brazil | Observational | 78 | 27.8 (23.6–32.1)a) | 98 | 22.8 (20.9–27.3)a) | Serum | 24–28 | Multiplex immunoassay | Carpenter and Coustan | 0.175 (0.120–0.298)a) | 0.155 (0.100–0.325)a) | Not significant (P=0.77) |
| Sudharshana Murthy et al. [4] | India | Observational | 30 | 25.7b) | 30 | 25b) | Serum | 24–28 | ELISA | India criteria | 2.96±1.37c) | 2.88±1.21c) | Significant (NR) |
| Siddiqui et al. [13] | India | Case-control | 53 | 25.3±4.08c) | 50 | 23.28±2.95c) | Serum | 24–31 | ELISA (disclose) | ADA | 0.72±0.67d) | 0.39±0.70d) | Significant (P=0.001) |
| Šimják et al. [18] | Prague | Case-control | 12 | V1: 26.04±3.75d) | 12 | V1: 27.63±4.59d) | Serum | V1: 28–32 | Multiplex immunoassay | IADPSG | V1 :4.33±0.81d) | V1: 2.78±0.37d) | Not significant (NR) |
| V2: 27.59±3.54d) | V2: 28.74±4.63d) | V2: 36–38 | V2: 5.02±1.13d) | V2: 3.69±0.29d) | |||||||||
| Yang et al. [16] | China | Cohort | 21 | 31.60±3.70c) | 34 | 31.70±3.10c) | Serum | <13 | ELISA | Carpenter and Coustan | 17/5 (12–23)a) | 11 (5–7)a) | Significant (P=0.001) |
| Yu et al. [22] | China | Case-control | 80 | NR | 60 | NR | Serum | 24–29 | ELISA | CDADB | 59.44±3.95 | 22.81±1.54 | Significant (P=0.021) |
| Zhao et al. [23] | China | Cross sectional | 29 | 28.10±3.20c) | 32 | 24.10±3.00c) | Serum | 24–28 | ELISA | Carpenter and Coustan | 5.10±1.20c) | 2.96±0.70c) | Significant (P<0.001) |
| Abell et al. [24] | Australia | Cohort | 25 | 28.0 (26.0–31.0)a) | 78 | 29.5 (25.5–34.0)a) | Serum | 12–15 | ELISA | ADIPS | 2.03 (1.67–2.90)a) | 1.92 (1.57–2.56)a) | Not significant (P=0.428) |
| Zhang et al. [25] | China | Case-control | 60 | 38.68±9.50c) | 60 | 28.74±7.26c) | Serum | 24–28 | Multiplex immunoassay | ESCPG/ACOG | 5.85±1.41c) | 3.91±1.66c) | Significant (P=0.002) |
| Bossick et al. [26] | USA | Case-control | 18 | 37.10±7.70c) | 167 | 28.40±7.10c) | Serum | 13–28 | Multiplex immunoassay | ACOG/ADA | 5.40±2.00c) | 3.90±2.10c) | Significant (P=0.002) |
| Oztop et al. [27] | Turkey | Case-control | 30 | 26.4 (17.3–44.1)a) | 20 | 24 (21–26)a) | Serum | 24–26 | ELISA | ADA | 3.10±0.90c) | 2.70±0.70c) | Significant (P=0.04) |
| Qu et al. [28] | China | Case-control | 53 | 32.17±4.36 | 57 | 23.62±3.51 | Serum | 22–30 | ELISA | Carpenter and Coustan | 4.43±0.45c) | 2.57±0.19c) | Significant (P=0.0004) |
| Gümüş et al. [29] | Turkey | Case-control | 101 | 27.00±5.00c) | 66 | 22.00±3.00c) | Serum | 24–28 | ELISA | ADA | 19.21±29.00c) | 15.49±19.13c) | Not significant (P=0.933) |
| Hassiakos et al. [30] | Greece | Case-control | 40 | 25.36e) | 94 | 23.16e) | Serum | 11–14 | Chemiluminescent immunoassay | IADPSG | 2.0 (1.60)f) | 1.5 (1.01)f) | Significant (P=0.001) |
| Kim et al. [31] | USA | Case-control | 32 | 24.1±0.9d) | 227 | 24.9±0.6d) | Serum | 24–28 | ELISA | Carpenter and Coustan | 2.6±0.2d) | 2.9±0.9d) | Not significant (NR) |
| Kuźmicki et al. [32] | Poland | Case-control | 46 | V1: 25.0 (24.2–28.0)a) | 45 | V1: 23.0 (20.4–26.2)a) | Serum | V1: 24–28 | ELISA | PDA | V1: 0.99 (0.86–1.27)a) | V1: 0.89 (0.75–1.07)a) | Significant (P<0.05) |
| V2: 25.0 (24.2–28.0)a) | V2: 23.0 (20.4–26.2)a) | V2: 29–32 | V2: 1.13 (0.98–1.57)a) | V2: 1.02 (0.83–1.22)a) | |||||||||
| Nergiz et al. [33] | Turkey | Case-control | 30 | 30.20±5.60c) | 30 | 29.10±30.00c) | Serum | >24 | ELISA | ACOG | 32.5 (24.8–48)a) | 7.1 (4.2–9.8)a) | Significant (P=0.001) |
| Özyer et al. [34] | Turkey | Case-control | 35 | 30.40±4.30c) | 70 | 25.30±3.80c) | Serum | 24–28 | Chemiluminescent sequential immunometric assay | Carpenter and Coustan/ADA | 2.0 (2.0–2.00)a) | 2.0 (2.0–2.51)a) | Not significant (P=0.08) |
| Mrizak et al. [35] | Tunisia | Case-control | 28 | 28.50±1.80c) | 24 | 29.7±2.23c) | Serum | 24–26 | ELISA | NDDG | 77.84±10.98c) | 53.17±1.45c) | Significant (P=0.002) |
| Abdel Gader et al. [36] | Saudi Arabia | Cross-sectional | 150 | 34.40±5.90c) | 100 | 30.60±4.60c) | Serum | 35–40 | ELISA | NDDG | 13.70+2.50c) | 13.90+15.30c) | Not significant (NR) |
| Morisset et al. [37] | Canada | Longitudinal | 20 | 28.20±7.50 | 27 | 24.20±4.30 | Serum | 26.1±3.7 | ELISA | CDA | 1.47±0.72 | 0.90±0.32 | Significant (P≤0.05) |
| Pöyhönen-Alho et al. [38] | Finland | Case-control | 38 | 30.6 | 21 | 26.9 | Plasma | 24–28 | ELISA | ADA | 4.9±2.21c) | 4.91±2.82c) | Not significant (P=0.58) |
| Kuzmicki et al. [39] | Poland | Case-control | 81 | 22.1 (20.5–24.9)a) | 82 | 23.1 (20.3–25.0)a) | Serum | 24–31 | ELISA | WHO | 1.0 (0.7–1.5)a) | 0.8 (0.5–1.1)a) | Significant (P=0.006) |
GDM, gestational diabetes mellitus; BMI, body mass index; IL, interleukin; V1, visit 1; V2, visit 2; ELISA, enzyme-linked immunosorbent assay; ADA, American Diabetes Association; IADPSG, International Association of Diabetes and Pregnancy Study Groups; CDADB, China Diabetes Association Diabetes Branch; ADIPS, Australasian Diabetes in Pregnancy Society; ESCPG, Endocrine Society Clinical Practice Guideline; ACOG, American College of Obstetricians and Gynecologists; PDA, Polish Diabetes Association; NDDG, National Diabetes Data Group; CDA, Canadian Diabetes Association; WHO, World Health Organization; NR, not reported; SD, standard deviation.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download