1. Heng YJ, Liong S, Permezel M, Rice GE, Di Quinzio MK, Georgiou HM. Human cervicovaginal fluid biomarkers to predict term and preterm labor. Front Physiol. 2015; 6:151.

2. Yoo JY, You YA, Kwon EJ, Park MH, Shim S, Kim YJ. Differential expression and methylation of integrin subunit alpha 11 and thrombospondin in the amnion of preterm birth. Obstet Gynecol Sci. 2018; 61:565–74.

3. Chan RL. Biochemical markers of spontaneous preterm birth in asymptomatic women. BioMed Res Int. 2014; 2014:164081.

4. Amabebe E, Chapman DR, Stern VL, Stafford G, Anumba DO. Mid-gestational changes in cervicovaginal fluid cytokine levels in asymptomatic pregnant women are predictive markers of inflammation-associated spontaneous preterm birth. J Reprod Immunol. 2018; 126:1–10.

5. Jefferson KK. The bacterial etiology of preterm birth. Adv Appl Microbiol. 2012; 80:1–22.

6. Dekker GA, Lee SY, North RA, McCowan LM, Simpson NA, Roberts CT. Risk factors for preterm birth in an international prospective cohort of nulliparous women. PLoS One. 2012; 7:e39154.

7. Son KA, Kim M, Kim YM, Kim SH, Choi SJ, Oh SY, et al. Prevalence of vaginal microorganisms among pregnant women according to trimester and association with preterm birth. Obstet Gynecol Sci. 2018; 61:38–47.

8. Kim YJ, Lee BE, Park HS, Kang JG, Kim JO, Ha EH. Risk factors for preterm birth in Korea: a multicenter prospective study. Gynecol Obstet Invest. 2005; 60:206–12.
9. Baer RJ, McLemore MR, Adler N, Oltman SP, Chambers BD, Kuppermann M, et al. Pre-pregnancy or first-trimester risk scoring to identify women at high risk of preterm birth. Eur J Obstet Gynecol Reprod Biol. 2018; 231:235–40.

10. Conde-Agudelo A, Papageorghiou AT, Kennedy SH, Villar J. Novel biomarkers for the prediction of the spontaneous preterm birth phenotype: a systematic review and meta-analysis. BJOG. 2011; 118:1042–54.

11. Paternoster DM, Muresan D, Vitulo A, Serena A, Battagliarin G, Dell’avanzo M, et al. Cervical phIGFBP-1 in the evaluation of the risk of preterm delivery. Acta Obstet Gynecol Scand. 2007; 86:151–5.

12. Liong S, Di Quinzio MK, Fleming G, Permezel M, Rice GE, Georgiou HM. New biomarkers for the prediction of spontaneous preterm labour in symptomatic pregnant women: a comparison with fetal fibronectin. BJOG. 2015; 122:370–9.

13. Son M, Miller ES. Predicting preterm birth: cervical length and fetal fibronectin. Semin Perinatol. 2017; 41:445–51.

14. Chandiramani M, Seed PT, Orsi NM, Ekbote UV, Bennett PR, Shennan AH, et al. Limited relationship between cervico-vaginal fluid cytokine profiles and cervical shortening in women at high risk of spontaneous preterm birth. PLoS One. 2012; 7:e52412.

15. DeFranco EA, Lewis DF, Odibo AO. Improving the screening accuracy for preterm labor: is the combination of fetal fibronectin and cervical length in symptomatic patients a useful predictor of preterm birth? A systematic review. Am J Obstet Gynecol. 2013; 208:233.e1–6.

16. Son GH, You YA, Kwon EJ, Lee KY, Kim YJ. Comparative analysis of midtrimester amniotic fluid cytokine levels to predict spontaneous very pre-term birth in patients with cervical insufficiency. Am J Reprod Immunol. 2016; 75:155–61.

17. Gulati S, Agrawal S, Raghunandan C, Bhattacharya J, Saili A, Agarwal S, et al. Maternal serum interleukin-6 and its association with clinicopathological infectious morbidity in preterm premature rupture of membranes: a prospective cohort study. J Matern Fetal Neonatal Med. 2012; 25:1428–32.

18. Curry AE, Vogel I, Skogstrand K, Drews C, Schendel DE, Flanders WD, et al. Maternal plasma cytokines in early- and mid-gestation of normal human pregnancy and their association with maternal factors. J Reprod Immunol. 2008; 77:152–60.

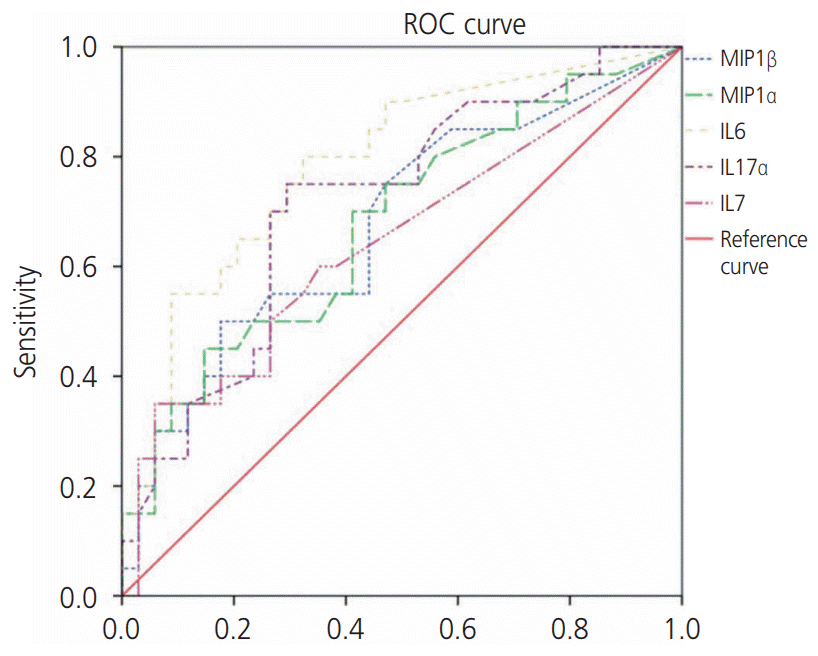
19. Chaemsaithong P, Romero R, Docheva N, Chaiyasit N, Bhatti G, Pacora P, et al. Comparison of rapid MMP-8 and interleukin-6 point-of-care tests to identify intraamniotic inflammation/infection and impending preterm delivery in patients with preterm labor and intact membranes. J Matern Fetal Neonatal Med. 2018; 31:228–44.

20. Musilova I, Bestvina T, Hudeckova M, Michalec I, Cobo T, Jacobsson B, et al. Vaginal fluid IL-6 concentrations as a point-of-care test is of value in women with preterm PROM. Am J Obstet Gynecol;Forthcoming. 2016.
21. Lee SM, Park KH, Jung EY, Kook SY, Park H, Jeon SJ. Inflammatory proteins in maternal plasma, cervicovaginal and amniotic fluids as predictors of intra-amniotic infection in preterm premature rupture of membranes. PLoS One. 2018; 13:e0200311.

22. Coleman MA, Keelan JA, McCowan LM, Townend KM, Mitchell MD. Predicting preterm delivery: comparison of cervicovaginal interleukin (IL)-1beta, IL-6 and IL-8 with fetal fibronectin and cervical dilatation. Eur J Obstet Gynecol Reprod Biol. 2001; 95:154–8.
23. Gibbs RS, Blanco JD, St Clair PJ, Castaneda YS. Quantitative bacteriology of amniotic fluid from women with clinical intraamniotic infection at term. J Infect Dis. 1982; 145:1–8.

24. Leitich H, Kaider A. Fetal fibronectin--how useful is it in the prediction of preterm birth? BJOG. 2003; 110 Suppl 20:66–70.

25. Goldenberg RL, Iams JD, Mercer BM, Meis PJ, Moawad A, Das A, et al. The Preterm Prediction Study: toward a multiple-marker test for spontaneous preterm birth. Am J Obstet Gynecol. 2001; 185:643–51.

26. Heng YJ, Di Quinzio MK, Permezel M, Rice GE, Georgiou HM. Interleukin-1 receptor antagonist in human cervicovaginal fluid in term pregnancy and labor. Am J Obstet Gynecol. 2008; 199:656.e1–7.

27. Kekki M, Kurki T, Kärkkäinen T, Hiilesmaa V, Paavonen J, Rutanen EM. Insulin-like growth factor-binding protein-1 in cervical secretion as a predictor of preterm delivery. Acta Obstet Gynecol Scand. 2001; 80:546–51.

28. Lockwood CJ, Senyei AE, Dische MR, Casal D, Shah KD, Thung SN, et al. Fetal fibronectin in cervical and vaginal secretions as a predictor of preterm delivery. N Engl J Med. 1991; 325:669–74.

29. Rizzo G, Capponi A, Rinaldo D, Tedeschi D, Arduini D, Romanini C. Interleukin-6 concentrations in cervical secretions identify microbial invasion of the amniotic cavity in patients with preterm labor and intact membranes. Am J Obstet Gynecol. 1996; 175:812–7.

30. Jacobsson B, Holst RM, Mattsby-Baltzer I, Nikolaitchouk N, Wennerholm UB, Hagberg H. Interleukin-18 in cervical mucus and amniotic fluid: relationship to microbial invasion of the amniotic fluid, intra-amniotic inflammation and preterm delivery. BJOG. 2003; 110:598–603.

31. Tanaka Y, Narahara H, Takai N, Yoshimatsu J, Anai T, Miyakawa I. Interleukin-1β and interleukin-8 in cervicovaginal fluid during pregnancy. Am J Obstet Gynecol. 1998; 179:644–9.

32. Sanchez-Ramos L, Mentel C, Bertholf R, Kaunitz AM, Delke I, Loge C. Human chorionic gonadotropin in cervicovaginal secretions as a predictor of preterm delivery. Int J Gynaecol Obstet. 2003; 83:151–7.

33. Kacerovsky M, Musilova I, Jacobsson B, Drahosova M, Hornychova H, Janku P, et al. Cervical fluid IL-6 and IL-8 levels in pregnancies complicated by preterm prelabor rupture of membranes. J Matern Fetal Neonatal Med. 2015; 28:134–40.

34. Ashford K, Chavan NR, Wiggins AT, Sayre MM, McCubbin A, Critchfield AS, et al. Comparison of serum and cervical cytokine levels throughout pregnancy between preterm and term births. AJP Rep. 2018; 8:–e113. –20.

35. Kiefer DG, Peltier MR, Keeler SM, Rust O, Ananth CV, Vintzileos AM, et al. Efficacy of midtrimester short cervix interventions is conditional on intraamniotic inflammation. Am J Obstet Gynecol. 2016; 214:276.e1–6.

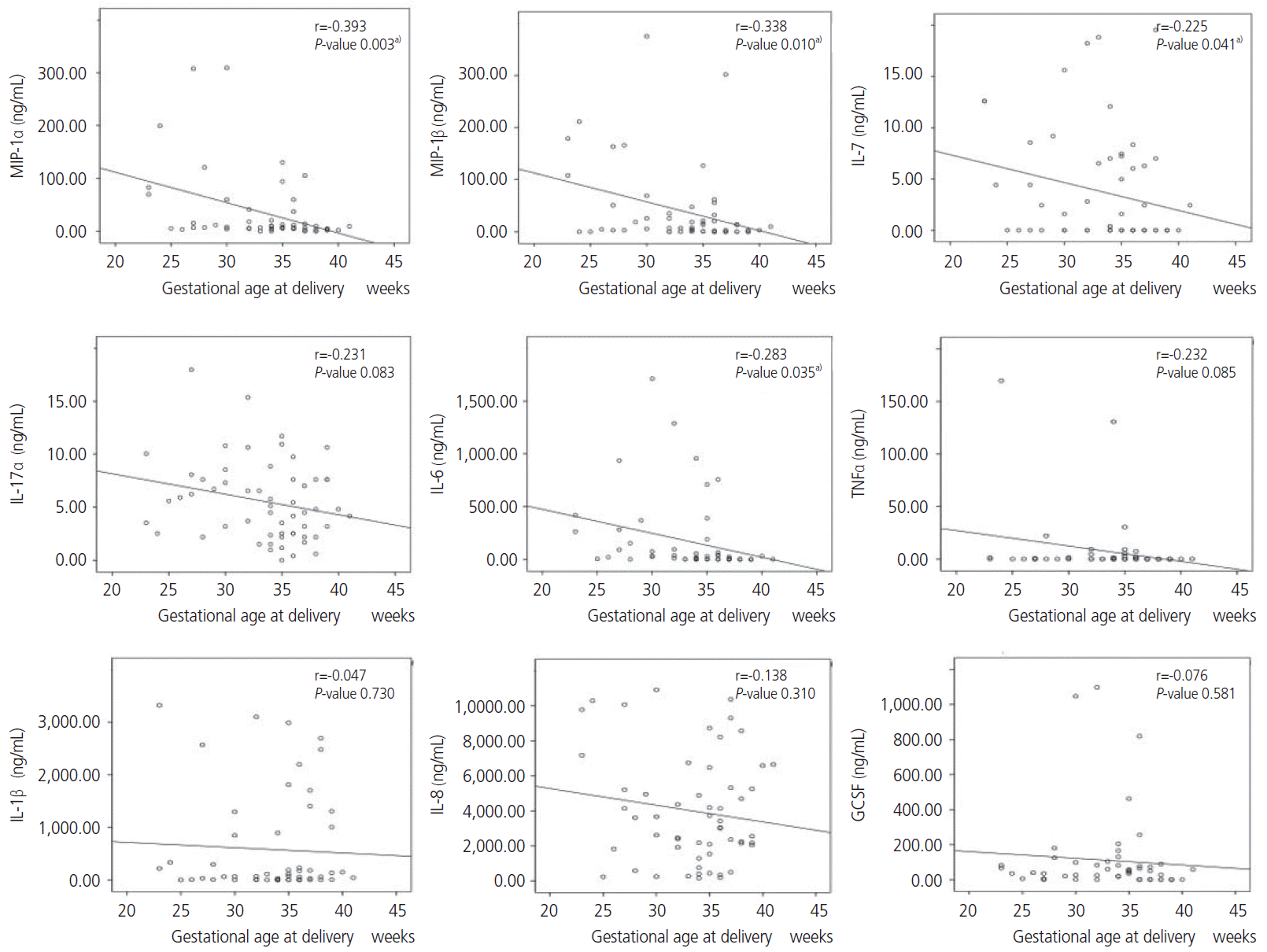
36. Romero R, Gomez R, Galasso M, Munoz H, Acosta L, Yoon BH, et al. Macrophage inflammatory protein-1 alpha in term and preterm parturition: effect of microbial invasion of the amniotic cavity. Am J Reprod Immunol. 1994; 32:108–13.
37. Lee SM, Park KH, Jung EY, Cho SH, Ryu A. Prediction of spontaneous preterm birth in women with cervical insufficiency: comprehensive analysis of multiple proteins in amniotic fluid. J Obstet Gynaecol Res. 2016; 42:776–83.

38. Herrera-Muñoz A, Fernández-Alonso AM, Fischer-Suárez N, Chedraui P, Pérez-López FR. Maternal serum cytokine levels in pregnancies complicated with threatened preterm labour. Gynecol Endocrinol. 2017; 33:408–12.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download