Abstract
Objective
We examined the factors affecting the false positive results in the accidental neonatal screening test (NST) within 3 days after birth and tried to find out the most relevant factors.
Methods
From January 2011 to December 2016, we reviewed the electronic medical records of 142 patients with a gestational age of 34 weeks or more who had false positives at the first NST. NST was performed by tandem mass spectrometry. We collected information on gestational age, birth weight, delivery method, Apgar score at the time of birth. Postnatal exam time, body weight, feeding volume and parenteral nutrition, antibiotics, and steroid administration were also investigated at the time of NST.
Results
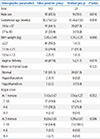
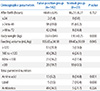
In the first NST, there were 102 cases of congenital adrenal hyperplasia, 27 cases of congenital hypothyroidism, nine cases of galactosemia and one case of homocystinuria. Gender, birth weight, and delivery method were statistically significant (P<0.05) at the time of birth using Pearson Chi-square method. There were significant differences in body weight, feeding volume, parenteral nutrition at the time of NST (P<0.05). In binary logistic regression analysis, the gender and the body weight seemed significant statistically.
Conclusion
Male sex and weight were identified as major factors of the false positive results in the NST performed within 3 days after birth, but further studies seemed to be needed. In the case of patients with short admission period or insufficient perinatal information, the first NST after birth should be performed at the time of outpatient department or after precise information collection.
Figures and Tables
Fig. 1
Incidences of major metabolic diseases at 1st neonatal screening test. Etc. includes hyperglycinemia, propionic academia, tyrosinemia.

References
1. Lee DH. Neonatal screening for inborn errors of metabolism. J Korean Pediatr Soc. 1987; 30:9–16.
2. Lee DH. Newborn screening of inherited metabolic disease in Korea. Korean J pediatr. 2006; 49:1125–1139.

3. Gurian EA, Kinnamon DD, Henry JJ, Waisbren SE. Expanded newborn screening for biochemical disorders: the effect of a false-positive result. Pediatrics. 2006; 117:1915–1921.

4. Schulze A, Lindner M, Kohlmüller D, Olgemöller K, Mayatepek E, Hoffmann GF. Expanded newborn screening for inborn errors of metabolism by electrospray ionization-tandem mass spectrometry: results, outcome, and implications. Pediatrics. 2003; 111(6 Pt 1):1399–1406.

5. Song JY, Son DW, Kim BI, Yang SW, Choi JH, Yoon CK, et al. Re-evaluation of TSH screening test in neonates. J Korean Pediatr Soc. 1993; 36:1502–1506.
6. Lee JM, Choi TY, Lee DW, Lee DH. Recheck rate, recall rate and reference range of the neonatal screening test for congenital hypothyroidism. J Clin Pathol Qual Control. 2001; 23:215–220.
7. Clinical and Laboratory Standards Institute. Newborn screening for preterm, low birth weight, and sick newborns; approved guideline. CLSI document NBS03-A. Wayne (PA): Clinical and Laboratory Standards Institute;2009.
8. Clinical and Laboratory Standards Institute. Newborn screening by tandem mass spectrometry; approved guideline. CLSI document NBS04-A. Wayne (PA): Clinical and Laboratory Standards Institute;2010.
9. Fant KE, Clark SJ, Kemper AR. Completeness and complexity of information available to parents from newborn-screening programs. Pediatrics. 2005; 115:1268–1272.

10. Green JM, Hewison J, Bekker HL, Bryant LD, Cuckle HS. Psychosocial aspects of genetic screening of pregnant women and newborns: a systematic review. Health Technol Assess. 2004; 8:iiiix–x. 1–109.

11. Kwon C, Farrell PM. The magnitude and challenge of false-positive newborn screening test results. Arch Pediatr Adolesc Med. 2000; 154:714–718.

12. Bodegård G, Fyrö K, Larsson A. Psychological reactions in 102 families with a newborn who has a falsely positive screening test for congenital hypothyroidism. Acta Paediatr Scand Suppl. 1983; 304:1–21.
13. Essex MJ, Klein MH, Cho E, Kalin NH. Maternal stress beginning in infancy may sensitize children to later stress exposure: effects on cortisol and behavior. Biol Psychiatry. 2002; 52:776–784.

15. Leslie LK, Boyce WT. Consultation with the specialist. The vulnerable child. Pediatr Rev. 1996; 17:323–326.

16. Choi TY, Kim JW, Min WK, Lee DH. Analysis of blood sample records for neonatal screening test and external quality assessment for inborn errors of metabolism in Korea. J Korean Soc Matern Child Health. 2003; 7:7–17.
17. Lee DH. The prevalence of pediatric endocrine and metabolic diseases in Korea. Korean J Pediatr. 2008; 51:559–563.

18. American College of Medical Genetics/American Society of Human Genetics Test and Technology Transfer Committee Working Group. Tandem mass spectrometry in newborn screening. American College of Medical Genetics/American Society of Human Genetics Test and Technology Transfer Committee Working Group. Genet Med. 2000; 2:267–269.
19. Matern D, Tortorelli S, Oglesbee D, Gavrilov D, Rinaldo P. Reduction of the false-positive rate in newborn screening by implementation of MS/MS-based second-tier tests: the Mayo Clinic experience (2004-2007). J Inherit Metab Dis. 2007; 30:585–592.

20. Shim KS, Hwang JS, Lim JS, Kim SY, Shin CH, Yang SW, et al. Re-evaluation of neonatal screening tests for inborn errors of metabolism with dried filter paper blood spots. J Korean Pediatr Soc. 1999; 42:1639–1644.
21. Ensenauer R, Vockley J, Willard JM, Huey JC, Sass JO, Edland SD, et al. A common mutation is associated with a mild, potentially asymptomatic phenotype in patients with isovaleric acidemia diagnosed by newborn screening. Am J Hum Genet. 2004; 75:1136–1142.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download