Abstract
Dysmenorrhea is the most common gynecologic condition in women during the reproductive period. Severe dysmenorrhea pain affects their social activities, sleep, and quality of life. Nevertheless, the proportion of women with dysmenorrhea do not receive adequate medical counseling or pharmacological treatments. Primary dysmenorrhea is diagnosed clinically, and the secondary causes that can cause pelvic pain should be identified. The treatment of choice for primary dysmenorrhea is non-steroidal anti-inflammatory drugs (NSAIDs). In order to maximize the therapeutic effect, it is necessary to ensure that the appropriate medication is administered in a proper way. NSAIDs can cause adverse effects, including gastrointestinal disorders. If side effects occur or are anticipated with NSAIDs, the use of hormonal contraceptives may be recommended when contraception is considered. In addition to these pharmacological treatments, heat, dietary, and behavioral therapies have been tried and reported to have some effects. However, further research is required for robust conclusions.
Go to : 
REFERENCES
1. Campbell MA, McGrath PJ. Use of medication by adolescents for the management of menstrual discomfort. Arch Pediatr Adolesc Med. 1997; 151:905–913.

2. Latthe P, Latthe M, Say L, Gulmezoglu M, Khan KS. WHO systematic review of prevalence of chronic pelvic pain: a neglected reproductive health morbidity. BMC Public Health. 2006; 6:177.

3. Burnett MA, Antao V, Black A, Feldman K, Grenville A, Lea R, Lefebvre G, Pinsonneault O, Robert M. Prevalence of primary dysmenorrhea in Canada. J Obstet Gynaecol Can. 2005; 27:765–770.

4. Sundell G, Milsom I, Andersch B. Factors influencing the prevalence and severity of dysmenorrhoea in young women. Br J Obstet Gynaecol. 1990; 97:588–594.

5. Baker FC, Driver HS, Rogers GG, Paiker J, Mitchell D. High nocturnal body temperatures and disturbed sleep in women with primary dysmenorrhea. Am J Physiol. 1999; 277:E1013–E1021.
7. Lee EH, Kim JI, Kim HW, Lee HK, Lee SH, Kang NM, Hur MH. Dysmenorrhea and menstrual attitudes in adult women. Korean J Women Health Nurs. 2003; 9:105–112.

8. Latthe P, Mignini L, Gray R, Hills R, Khan K. Factors predisposing women to chronic pelvic pain: systematic review. BMJ. 2006; 332:749–755.

9. Altunyurt S, Gol M, Altunyurt S, Sezer O, Demir N. Primary dysmenorrhea and uterine blood flow: a color Doppler study. J Reprod Med. 2005; 50:251–255.
11. Chan WY, Dawood MY, Fuchs F. Relief of dysmenorrhea with the prostaglandin synthetase inhibitor ibuprofen: effect on prostaglandin levels in menstrual fluid. Am J Obstet Gynecol. 1979; 135:102–108.

12. Dawood MY. Primary dysmenorrhea: advances in pathogenesis and management. Obstet Gynecol. 2006; 108:428–441.
13. Nigam S, Benedetto C, Zonca M, Leo-Rossberg I, Lubbert H, Hammerstein J. Increased concentrations of eicosanoids and platelet-activating factor in menstrual blood from women with primary dysmenorrhea. Eicosanoids. 1991; 4:137–141.
14. Buhimschi I, Yallampalli C, Dong YL, Garfield RE. Involvement of a nitric oxide-cyclic guanosine monophosphate pathway in control of human uterine contractility during pregnancy. Am J Obstet Gynecol. 1995; 172:1577–1584.

15. Moya RA, Moisa CF, Morales F, Wynter H, Ali A, Narancio E. Transdermal glyceryl trinitrate in the management of primary dysmenorrhea. Int J Gynaecol Obstet. 2000; 69:113–118.

16. Pittrof R, Lees C, Thompson C, Pickles A, Martin JF, Campbell S. Crossover study of glyceryl trinitrate patches for controlling pain in women with severe dysmenorrhoea. BMJ. 1996; 312:884.

19. French L. Dysmenorrhea. Am Fam Physician. 2005; 71:285–291.
20. Marjoribanks J, Proctor ML, Farquhar C. Nonsteroidal antiinflammatory drugs for primary dysmenorrhoea. Cochrane Database Syst Rev. 2003; 4:CD001751.

23. Budoff PW. Use of mefenamic acid in the treatment of primary dysmenorrhea. JAMA. 1979; 241:2713–2716.

24. Athanasiou S, Bourne TH, Khalid A, Okokon EV, Crayford TJ, Hagstrom HG, Campbell S, Collins WP. Effects of indomethacin on follicular structure, vascularity, and function over the periovulatory period in women. Fertil Steril. 1996; 65:556–560.
26. Wong CL, Farquhar C, Roberts H, Proctor M. Oral contraceptive pill as treatment for primary dysmenorrhoea. Cochrane Database Syst Rev. 2009; 2:CD002120.

27. Davis AR, Westhoff C, O'Connell K, Gallagher N. Oral contraceptives for dysmenorrhea in adolescent girls: a randomized trial. Obstet Gynecol. 2005; 106:97–104.
28. Proctor ML, Roberts H, Farquhar CM. Combined oral contraceptive pill (OCP) as treatment for primary dysmenorrhoea. Cochrane Database Syst Rev. 2001; 4:CD002120.
29. Zahradnik HP, Goldberg J, Andreas JO. Efficacy and safety of the new antiandrogenic oral contraceptive Belara. Contraception. 1998; 57:103–109.

31. Anderson FD, Gibbons W, Portman D. Safety and efficacy of an extended-regimen oral contraceptive utilizing continuous low-dose ethinyl estradiol. Contraception. 2006; 73:229–234.

32. Yucel N, Baskent E, Karamustafaoglu Balci B, Goynumer G. The levonorgestrel-releasing intrauterine system is associated with a reduction in dysmenorrhoea and dyspareunia, a decrease in CA 125 levels, and an increase in quality of life in women with suspected endometriosis. Aust N Z J Obstet Gynaecol. 2018; 58:560–563.

33. Lee JY. Therapeutic efficacy of Mirena in gynecologic. J Korean Med Assoc. 2019; 62:459–465.
34. Croxatto HB. Clinical profile of Implanon: a single-rod etonogestrel contraceptive implant. Eur J Contracept Reprod Health Care. 2000; 5(Suppl 2):21–28.
35. Roumen FJ. The contraceptive vaginal ring compared with the combined oral contraceptive pill: a comprehensive review of randomized controlled trials. Contraception. 2007; 75:420–429.

36. Harel Z, Biro FM, Kollar LM. Depo-Provera in adolescents: effects of early second injection or prior oral contraception. J Adolesc Health. 1995; 16:379–384.

37. Ling FW. Pelvic Pain Study Group. Randomized controlled trial of depot leuprolide in patients with chronic pelvic pain and clinically suspected endometriosis. Obstet Gynecol. 1999; 93:51–58.

38. Akin M, Price W, Rodriguez G Jr, Erasala G, Hurley G, Smith RP. Continuous, low-level, topical heat wrap therapy as compared to acetaminophen for primary dysmenorrhea. J Reprod Med. 2004; 49:739–745.
39. Akin MD, Weingand KW, Hengehold DA, Goodale MB, Hinkle RT, Smith RP. Continuous low-level topical heat in the treatment of dysmenorrhea. Obstet Gynecol. 2001; 97:343–349.

40. Wilson ML, Murphy PA. Herbal and dietary therapies for primary and secondary dysmenorrhoea. Nurs Times. 2001; 97:44.
41. Zhu X, Proctor M, Bensoussan A, Wu E, Smith CA. Chinese herbal medicine for primary dysmenorrhoea. Cochrane Database Syst Rev. 2008; 2:CD005288.

42. Pakniat H, Chegini V, Ranjkesh F, Hosseini MA. Comparison of the effect of vitamin E, vitamin D and ginger on the severity of primary dysmenorrhea: a single-blind clinical trial. Obstet Gynecol Sci. 2019; 62:462–468.

43. Jenabi E, Fereidooni B, Karami M, Masoumi SZ, Safari M, Khazaei S. The effect of bee propolis on primary dysmenorrhea: a randomized clinical trial. Obstet Gynecol Sci. 2019; 62:352–356.
44. Golomb LM, Solidum AA, Warren MP. Primary dysmenorrhea and physical activity. Med Sci Sports Exerc. 1998; 30:906–909.

Go to : 
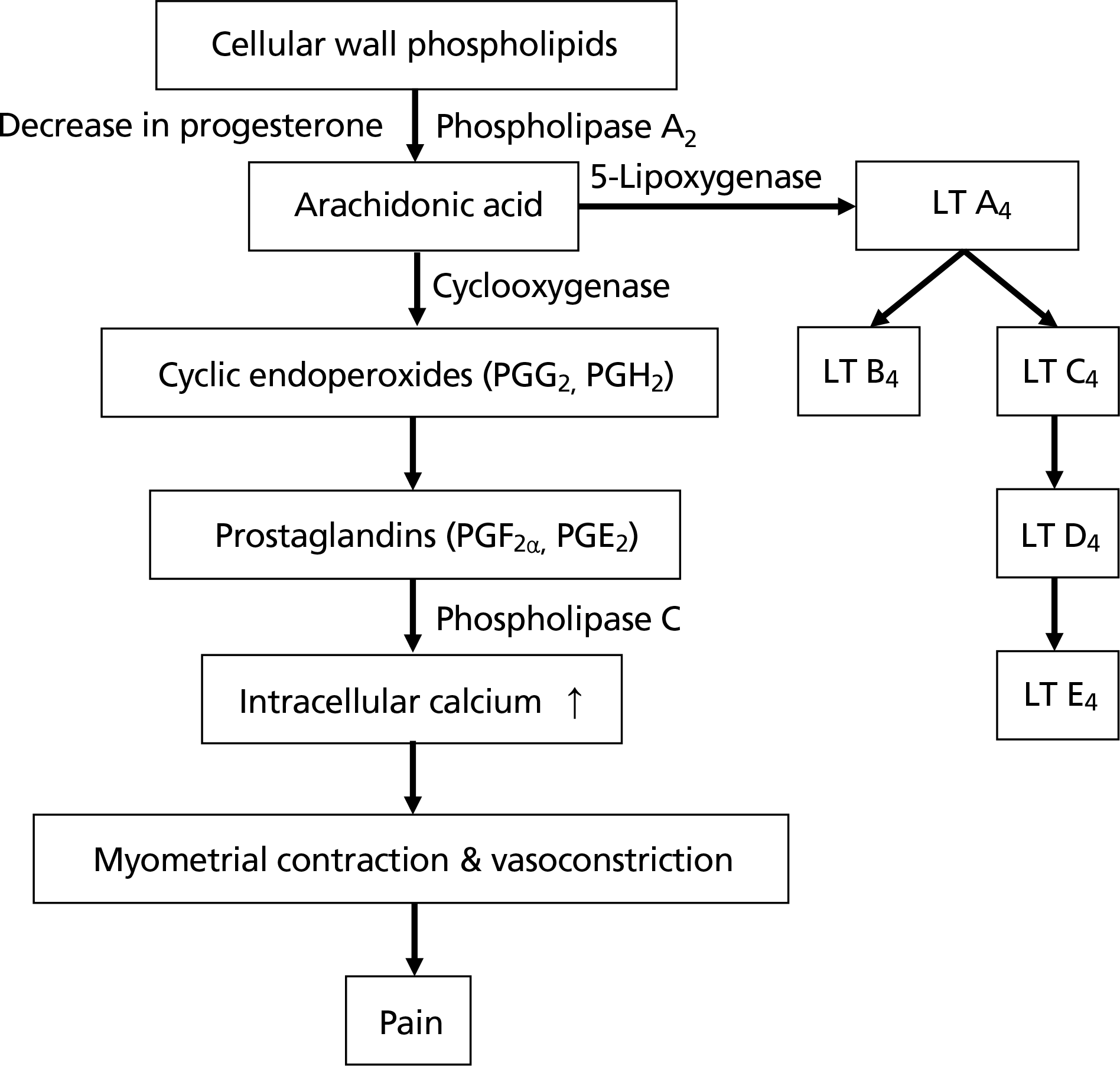 | Figure 1.Metabolic pathways of uterine contraction in dysmenorrhea. LT, leukotriene; PGG2, prostaglandin G2; PGH2, prostaglandin H2; PGF2α, prostaglandin F2α; PGE2, prostaglandin E2. |
Table 1.
Charact teristics of primary and secondary dysmenorrhea
Table 2.
Non-steroidal anti-inflammatory agents to treat dysmenorrhea




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download