Abstract
In cases of chronic hyperglycemia, advanced glycation end-products (AGEs) are actively produced and accumulated in the circulating blood and various tissues. AGEs also accelerate the expression of receptors for AGEs, and they play an important role in the development of diabetic vascular complications through various mechanisms. Active interventions for glucose and related risk factors may help improve the clinical course of patients by reducing AGEs. This review summarizes recent updates on AGEs that have a significant impact on diabetic vascular complications.
The Sulwon Award for Scientific Achievement is the Korean Diabetes Association's highest scientific award and honors an individual who has excellently contributed to the progress in the field of diabetes and metabolism. Sulwon Award is named after an emeritus professor Eung Jin Kim, who founded Korean Diabetes Association. Prof. Young Seol Kim received the ninth Sulwon Award at 2017 International Conference on Diabetes and Metabolism, September 28 to 30, 2017 at Seoul, Korea.
Chronic hyperglycemia, a contributing factor in diabetic vascular complications, has been evaluated through experimental and large-scale clinical studies, such as the Diabetes Control and Complications Trial (DCCT) and the UK Prospective Diabetes Study (UKPDS) [123]. A follow-up DCCT study found that when the regulation of blood glucose was controlled strictly for 6.5 years at the initial stage of type 1 diabetes mellitus (T1DM), the effect lasted so long that the progression of microvascular complications was delayed for at least 8 years after the termination of the initial study, and the occurrence of cardiovascular events was delayed for 11 years [2]. According to the UKPDS follow-up study, patients with type 2 diabetes mellitus (T2DM) also had the legacy effect stemming from the strict regulation of blood sugar from the initial stage of the disease, which suggests that the phenomenon of metabolic memory exists in the vascular complications of diabetes [3].
Recent studies have suggested that advanced glycation end-products (AGEs) may be a key factor in the development of metabolic memory in diabetic complications, because AGEs are produced and accumulated irreversibly in the body, depending on the degree of blood sugar regulation and duration [4]. Once produced, the material is metabolized so slowly that it can well explain the phenomenon of metabolic memory or legacy effect [5]. Direct evidence of the relationship between the accumulation of AGEs and development of diabetic vascular complications was reported recently [6].
In this review, we summarized the recent AGE-related updates that have an important influence on the occurrence and deterioration of these diabetic vascular complications.
AGEs are produced by the Maillard reaction. A reducing sugar, such as glucose, reacts non-enzymatically with the amino group of proteins to produce a Schiff base that rearranges into Amadori compounds [5]. These Amadori adducts then very slowly undergo irreversible dehydration and condensation reactions, eventually producing AGEs, which are yellowish-brown materials with particular fluorescence [5].
AGEs are produced not only from glucose but also from the dicarbonyl compound produced from autoxidation and the degradation products of glucose, such as glyoxal, methylglyoxal, and 3-deoxyglucosone, or α-hydroxy aldehydes such as glyceraldehyde and glycolaldehyde [78]. In the case of chronic hyperglycemia, AGEs are actively produced and accumulate in the circulating blood and various tissues, resulting in vascular complications in diabetes [5].
AGEs accelerate the expression of receptors for advanced glycation end-products (RAGEs). The constant activation of the AGE-RAGE system is presumed to create the long-term metabolic memory or legacy effect [11]. Upon the recognition of AGEs by RAGE in endothelial cells, the production of oxidative stress is accelerated in the cells, and various cytokines and growth factors are secreted through the activation of nuclear factor κB, thereby inducing the expression of adhesion factor and causing an inflammatory response (Fig. 1) [12]. In patients with diabetes, RAGE expression is accelerated in atherosclerotic lesions in proportion to aggravation of blood sugar regulation [13].
The oxidative stress accelerated by AGE-RAGE inactivates nitric oxide and then either causes an inflammatory response or aggravates a thrombotic tendency that leads to the progression of arteriosclerosis [1415]. In endothelial cells, the AGE-RAGE system promotes the gene expression of p22phox and gp91phox, which are components of the reduced form of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, and accelerates the production of oxidative stress by activating the cell membrane transport of Rac family small GTPase 1 (Rac1) to cause endothelial cell dysfunction [16].
AGEs promote the production of vascular endothelial growth factor (VEGF) in the endothelial cells, which then induces pathological angiogenesis [17]. VEGF is known to be involved in the increasing atheroma in an atherosclerotic lesion [18]. This suggests that the AGE-RAGE system, through the production of VEGF, activates angiogenesis in atheroma and aggravates inflammation in the plaque, possibly by a hemorrhage or an increase in the atheroma [19].
In addition, the AGE-RAGE system inhibits the production of prostacyclin (PGI2) in endothelial cells and promotes the de novo synthesis of plasminogen activator inhibitor-1, thereby inhibiting fibrinolytic activity and then contributing to stabilization of the thrombus [19]. A decrease in cyclic adenosine monophosphate production through the AGE-RAGE system is expected to be involved in the thrombotic tendency [20].
AGEs are known to promote not only platelet aggregation but also the blood coagulation cascade through tissue factor production [21]. The thrombotic tendency induced by AGEs is considered the cause of acute coronary syndromes, such as unstable angina or acute myocardial infarction through atheroma rupture and subsequent thrombogenesis in the coronary artery [21].
The low density lipoproteins (LDLs) oxidized and denatured by AGEs are recognized by scavenger receptors, which then activate foam cell formation from macrophages and the secretion of chemokines or growth factors therein. The LDLs therefore accelerate the migration, proliferation, and transformation of smooth-muscle cells or the endothelial invasion by monocytes [22].
A clear progression of atherosclerosis and plaques accompanied by calcified lesions is observed in patients with diabetes [23]. In addition, the appearance of calcified lesions in atherosclerotic lesions is considered a predictive factor of future cardiovascular events and a marker of poor prognosis resistance to coronary intervention [23]. AGEs act on blood vessel wall cells, such as pericytes, promoting their differentiation into osteoblasts and thus causing calcification deposition [24]. Therefore, AGEs present in atherosclerotic lesions are presumed to be involved in the occurrence and progression of calcified lesions by acting on pericytes of the angiogenesis induced by an increase in atheroma [25].
The oxidative stress accelerated by the AGE-RAGE system not only causes vascular disorder by activating the renin-angiotensin system (RAS) but also aggravates organ failure, because RAS activation causes NADPH oxidase-derived oxidative stress that again leads to an increase in RAGE expression as well as AGE production [2627]. This fact suggests that inhibition of the AGE-RAGE system is involved in some of the long-term protective effects of RAS inhibitors [27].
It is known that in hyperglycemia, the de novo synthesis of diacylglycerol is accelerated and protein kinase Cβ (PKCβ) is activated, and sorbitol or fructose is accumulated in the cells as a result of polyol pathway hyperactivity [28]. PKCβ activation is presumed to accelerate the activity of NADPH oxidase and then causes vascular complications in diabetes in connection with the AGE-RAGE system [28]. Fructose has a 10-time stronger glycation activity than glucose, and AGE formation from intracellular protein and fructose may be involved in vascular disorders [29]. The mitochondrial-derived oxidative stress produced by the hyperglycemic load causes AGE production, PKCβ activation, and polyol pathway hyperactivity [28]. These three hypotheses, which have existed for many years, and the AGE-RAGE system have a mutual role in the development of diabetic vascular complications [30].
Serum levels of AGEs are known to constitute an independent risk factor of endothelial dysfunction in patients with T2DM [4]. In addition, serum levels of AGEs were shown to be an independent factor for predicting death from cardiovascular events in women with T2DM or in nondiabetic patients in an 18-year follow-up study [30]. It is also known that serum AGE levels are related to left ventricular diastolic dysfunction and vascular stiffness in patients with T1DM [31].
RAGE has three splice variants of full-length RAGE, an N-terminal variant that does not contain an AGE-binding domain, a soluble receptor for advanced glycation end-product (sRAGE), and a C-terminal splice variant that does not include transmembrane and effector domains [32]. The physiological function of sRAGE has not yet been fully characterized, but it is known to be produced through cleavage of cell-surface RAGE and is known to reflect endothelial cell damage [33]. However, it is known that sRAGE can modulate the AGE-RAGE system as an antagonist of full-length RAGE [34].
Although serum levels of sRAGEs are only about 1/1,000 to 1/5,000 of the AGE concentrations in circulation, sRAGE could become an indicator for predicting the degree of vascular disorders because it reflects the RAGE expression in tissues [30]. One study also demonstrated that serum sRAGE levels were significantly higher in T2DM patients than in non-diabetic subjects and positively associated with the presence of coronary artery disease [33].
Because the AGE reaction is also referred to as the browning reaction, the browning change of foods can be a measure of their AGE content. In general, it is known that a large amount of AGEs are produced when meats or fatty foods are heated or baked at a high temperature [35]. On the other hand, AGEs are not produced in a significant amount when foods are slowly steamed or heated with a lot of water for a long time [35]. For example, chicken shows a clear browning reaction, and grilled chicken skewers in particular have about 10 times higher levels of AGEs than chicken boiled in water [35].
Because assessments of their eating history and eating habits suggest a correlation between the amount of AGE intake and serum AGE level, a decrease in the amount of food-derived AGE intake is considered to inhibit vascular complications in diabetic patients [3536]. In this regard, some experiments also reported that the long-term consumption of AGE-limiting foods in mice reduced oxidative stress markers and inhibited insulin resistance and organ failure, resulting in an extension of life span [37]. Therefore, the limitation of food-derived AGEs can become a significant therapeutic target for inhibiting organ failure due to diabetes or aging.
Indeed, when patients with T2DM consumed AGE-restricted diet, the serum levels of AGEs decreased by around 30% to 40% in 2 to 6 weeks, resulting in a 20% decrease in markers of vascular complications, such as soluble vascular cell adhesion molecule-1 or C-reactive protein, or inflammatory markers [38]. In addition, it is known that in diabetic patients who limited food-derived AGEs, the AGE formation or oxidative degeneration of blood LDL-cholesterol is inhibited, and vascular complications due to LDL-cholesterol are also decreased [39]. However, it is not yet known whether the long-term, AGE-limiting diets can inhibit vascular complications in diabetic patients and improve their prognosis. In addition, differences in the absorption capacity of the gastrointestinal tract; the binding capacity with RAGE; and organ failure, depending on the type of AGEs taken up by the body, should be considered.
Several drugs are known to modulate AGEs (Table 1). Statin could slow the AGE-RAGE-mediated activation of NADPH oxidase by inhibiting the prenylation of Rac1, which subsequently inhibits post-RAGE-induced signal transduction [40]. Pravastatin is known to reduce tubular damage in diabetic nephropathy in tubular cells and attenuate AGEs-induced apoptosis [41]. Atorvastatin is also known to inhibit AGE formation through its anti-oxidative activity [42].
Telmisartan, an angiotensin receptor blocker with selective peroxisome proliferator-activated receptor γ activation, is known to inhibit RAGE expression in endothelial cells, renal mesangial cells, and hepatocytes [43]. It is known to inhibit the expression of oxidative stress and inflammatory markers by inhibiting signal transduction by AGEs and expression of arteriosclerosis-related genes [4344]. It is also known that ramipril, an angiotensin-converting enzyme inhibitor, can regulate AGEs with a similar mechanism of action [45].
The AGE modulatory potential is also known in some anti-diabetic medications. Rosiglitazone could reduce the expression of RAGE on the myocardium and attenuates cardiac fibrosis and ventricular diastolic function in experimental models [46]. Exendin-4 could inhibit the AGE-RAGE-mediated damage in tubular cells to attenuate the development and progression of diabetic nephropathy [47]. Linagliptin also has beneficial effects on diabetic nephropathy, partially inhibiting AGE-RAGE-evoked oxidative stress generation [4849].
Therefore, these agents are regarded as AGE-RAGE modulators in a broad sense. However, it is still unclear whether the modulation of AGEs by these drugs plays an important role in the improvement of diabetic macrovascular complications, compared to the main effects of these drugs.
Recently, the efficacy of aminoguanidine has been evaluated as a novel intervention for AGEs. Studies have shown that aminoguanidine inhibits the formation of AGEs and inhibits micro- and macrovascular complications in apolipoprotein knockout mice [505152]. It is also known that AGE cross-link breakers such as ALT-711 or alagebrium may help prevent diabetic vascular complications [4552]. In addition, the effect of direct antagonism on AGE of sRAGE has been demonstrated in some experimental studies [32]. However, the effect of the AGE modulator on advanced complications is unclear. In addition, there are few intervention studies of the actual clinical usefulness.
The formation of AGEs in diabetes patients occurs as a consequence of chronically elevated blood glucose levels. Some of the AGEs are known to be ingested through food. These AGEs could be one of the major contributing factors to the development of diabetic complications. It increases oxidative stress in the body according to various mechanisms and induces an inflammatory reaction, which has an important influence on the occurrence and exacerbation of diabetic vascular complications.
Recently, several clinical and preclinical studies have shown that interventions for these AGEs may play an important role in the reduction and prevention of diabetic vascular complications. This can be achieved not only by proper blood glucose control but also through diet and medical intervention. Recently, new compounds for the regulation of AGEs in the body have been studied.
In the future, further research results will clarify the role of AGEs in diabetic vascular complications, and we hope that successful intervention will help prevent complications and improve the quality of life for diabetic patients.
Figures and Tables
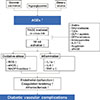 | Fig. 1Suggested mechanism of advanced glycation end-products (AGEs) in diabetic vascular complications. RAGE, receptor for advanced glycation end-product; NF-κB, nuclear factor κB; RAS, renin-angiotensin system; TZD, thiazolidinediones; GLP-1, glucagon like peptide-1; DPP-4, dipeptidylpeptide-4; sRAGE, soluble receptor for advanced glycation end-product; ROS, reactive oxygen species; eNOS, endothelial nitric oxide synthase; NADPH, nicotinamide adenine dinucleotide phosphate; IL, interleukin; TNF-α, tumor necrosis factor α. |
Table 1
Agents known to modulate AGEs

| Agents | Suggested effects | References |
|---|---|---|
| Pravastatin | Reduce tubular damage in diabetic nephropathy in tubular cells and attenuate AGEs-induced apoptosis | [41] |
| Atorvastatin | Inhibit AGE formation through its anti-oxidative activity | [42] |
| Telmisartan | Inhibit the expression of oxidative stress and inflammatory markers by inhibiting signal transduction by AGEs and expression of arteriosclerosis-related genes | [43,44] |
| Ramipril | Inhibit the expression of oxidative stress and inflammatory markers by inhibiting signal transduction by AGEs | [45] |
| Rosiglitazone | Reduce the expression of RAGE on the myocardium and attenuate cardiac fibrosis and ventricular diastolic function | [46] |
| Exendin-4 | Inhibit the AGE-RAGE mediated damage in tubular cells to attenuate the development and progression of diabetic nephropathy | [47] |
| Linagliptin | Inhibit AGE-RAGE–evoked oxidative stress generation | [48,49] |
| Aminoguanidine | Prevent cardiac hypertrophy and arterial stiffening in diabetic cardiomyopathy | [50,51,52] |
| ALT-711 | AGE cross-link breaker | [52] |
| Alagebrium | AGE cross-link breaker | [45] |
| sRAGE | Competitive inhibitor of RAGE | [32] |
ACKNOWLEDGMENTS
Professor Young Seol Kim received the ninth Sulwon Award at the 2017 International Conference on Diabetes and Metabolism, September 28 to 30, 2017, at Seoul, Korea. The work was supported by a grant from the Korea Healthcare Technology R&D Project, Ministry of Health and Welfare Republic of Korea (HI10C2020).
References
1. Vlassara H, Palace MR. Diabetes and advanced glycation endproducts. J Intern Med. 2002; 251:87–101.

2. Writing Team for the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: the Epidemiology of Diabetes Interventions and Complications (EDIC) study. JAMA. 2003; 290:2159–2167.
3. Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-Year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008; 359:1577–1589.

4. Singh R, Barden A, Mori T, Beilin L. Advanced glycation end-products: a review. Diabetologia. 2001; 44:129–146.

5. Yamagishi S, Imaizumi T. Diabetic vascular complications: pathophysiology, biochemical basis and potential therapeutic strategy. Curr Pharm Des. 2005; 11:2279–2299.

6. Koska J, Saremi A, Howell S, Bahn G, De Courten B, Ginsberg H, Beisswenger PJ, Reaven PD. VADT Investigators. Advanced glycation end products, oxidation products, and incident cardiovascular events in patients with type 2 diabetes. Diabetes Care. 2018; 41:570–576.

7. Lo TW, Westwood ME, McLellan AC, Selwood T, Thornalley PJ. Binding and modification of proteins by methylglyoxal under physiological conditions. A kinetic and mechanistic study with N alpha-acetylarginine, N alpha-acetylcysteine, and N alpha-acetyllysine, and bovine serum albumin. J Biol Chem. 1994; 269:32299–32305.

8. Ahmed N, Thornalley PJ, Dawczynski J, Franke S, Strobel J, Stein G, Haik GM. Methylglyoxal-derived hydroimidazolone advanced glycation end-products of human lens proteins. Invest Ophthalmol Vis Sci. 2003; 44:5287–5292.

9. Uribarri J, Cai W, Sandu O, Peppa M, Goldberg T, Vlassara H. Diet-derived advanced glycation end products are major contributors to the body's AGE pool and induce inflammation in healthy subjects. Ann N Y Acad Sci. 2005; 1043:461–466.

10. Yamagishi S, Ueda S, Okuda S. Food-derived advanced glycation end products (AGEs): a novel therapeutic target for various disorders. Curr Pharm Des. 2007; 13:2832–2836.

11. Bierhaus A, Hofmann MA, Ziegler R, Nawroth PP. AGEs and their interaction with AGE-receptors in vascular disease and diabetes mellitus. I. The AGE concept. Cardiovasc Res. 1998; 37:586–600.

12. Naka Y, Bucciarelli LG, Wendt T, Lee LK, Rong LL, Ramasamy R, Yan SF, Schmidt AM. RAGE axis: animal models and novel insights into the vascular complications of diabetes. Arterioscler Thromb Vasc Biol. 2004; 24:1342–1349.
13. Schmidt AM, Stern D. Atherosclerosis and diabetes: the RAGE connection. Curr Atheroscler Rep. 2000; 2:430–436.

14. Xu B, Ji Y, Yao K, Cao YX, Ferro A. Inhibition of human endothelial cell nitric oxide synthesis by advanced glycation end-products but not glucose: relevance to diabetes. Clin Sci (Lond). 2005; 109:439–446.

15. Bucala R, Tracey KJ, Cerami A. Advanced glycosylation products quench nitric oxide and mediate defective endothelium-dependent vasodilatation in experimental diabetes. J Clin Invest. 1991; 87:432–438.

16. Yamagishi S, Nakamura N, Suematsu M, Kaseda K, Matsui T. Advanced glycation end products: a molecular target for vascular complications in diabetes. Mol Med. 2015; 21:Suppl 1. S32–S40.

17. Treins C, Giorgetti-Peraldi S, Murdaca J, Van Obberghen E. Regulation of vascular endothelial growth factor expression by advanced glycation end products. J Biol Chem. 2001; 276:43836–43841.

18. Chung NA, Lydakis C, Belgore F, Li-Saw-Hee FL, Blann AD, Lip GY. Angiogenesis, thrombogenesis, endothelial dysfunction and angiographic severity of coronary artery disease. Heart. 2003; 89:1411–1415.

19. Yamagishi S, Adachi H, Takeuchi M, Enomoto M, Furuki K, Matsui T, Nakamura K, Imaizumi T. Serum level of advanced glycation end-products (AGEs) is an independent determinant of plasminogen activator inhibitor-1 (PAI-1) in nondiabetic general population. Horm Metab Res. 2007; 39:845–848.

20. Yamagishi S. Role of advanced glycation end products (AGEs) and receptor for AGEs (RAGE) in vascular damage in diabetes. Exp Gerontol. 2011; 46:217–224.

21. Singh VP, Bali A, Singh N, Jaggi AS. Advanced glycation end products and diabetic complications. Korean J Physiol Pharmacol. 2014; 18:1–14.

22. Takenaka K, Yamagishi S, Matsui T, Nakamura K, Imaizumi T. Role of advanced glycation end products (AGEs) in thrombogenic abnormalities in diabetes. Curr Neurovasc Res. 2006; 3:73–77.

23. Chait A, Bornfeldt KE. Diabetes and atherosclerosis: is there a role for hyperglycemia? J Lipid Res. 2009; 50:Suppl. S335–S339.

24. Wei Q, Ren X, Jiang Y, Jin H, Liu N, Li J. Advanced glycation end products accelerate rat vascular calcification through RAGE/oxidative stress. BMC Cardiovasc Disord. 2013; 13:13.

25. Cipollone F, Iezzi A, Fazia M, Zucchelli M, Pini B, Cuccurullo C, De Cesare D, De Blasis G, Muraro R, Bei R, Chiarelli F, Schmidt AM, Cuccurullo F, Mezzetti A. The receptor RAGE as a progression factor amplifying arachidonate-dependent inflammatory and proteolytic response in human atherosclerotic plaques: role of glycemic control. Circulation. 2003; 108:1070–1077.
26. Cheng CL, Tang Y, Zheng Z, Liu X, Ye ZC, Wang C, Lou TQ. Advanced glycation end-products activate the renin-angiotensin system through the RAGE/PI3-K signaling pathway in podocytes. Clin Invest Med. 2012; 35:E282.

27. Geronikaki A, Gavalas A, Dislian V, Giannoglou G. Inhibition of renin-angiotensin system and advanced glycation end products formation: a promising therapeutic approach targeting on cardiovascular diseases. Cardiovasc Hematol Agents Med Chem. 2007; 5:249–264.

28. Gleissner CA, Galkina E, Nadler JL, Ley K. Mechanisms by which diabetes increases cardiovascular disease. Drug Discov Today Dis Mech. 2007; 4:131–140.

29. Yamagishi S, Nakamura K, Matsui T, Noda Y, Imaizumi T. Receptor for advanced glycation end products (RAGE): a novel therapeutic target for diabetic vascular complication. Curr Pharm Des. 2008; 14:487–495.

30. Nakamura K, Yamagishi S, Adachi H, Kurita-Nakamura Y, Matsui T, Yoshida T, Imaizumi T. Serum levels of sRAGE, the soluble form of receptor for advanced glycation end products, are associated with inflammatory markers in patients with type 2 diabetes. Mol Med. 2007; 13:185–189.

31. Berg TJ, Snorgaard O, Faber J, Torjesen PA, Hildebrandt P, Mehlsen J, Hanssen KF. Serum levels of advanced glycation end products are associated with left ventricular diastolic function in patients with type 1 diabetes. Diabetes Care. 1999; 22:1186–1190.

32. Yonekura H, Yamamoto Y, Sakurai S, Petrova RG, Abedin MJ, Li H, Yasui K, Takeuchi M, Makita Z, Takasawa S, Okamoto H, Watanabe T, Yamamoto H. Novel splice variants of the receptor for advanced glycation end-products expressed in human vascular endothelial cells and pericytes, and their putative roles in diabetes-induced vascular injury. Biochem J. 2003; 370(Pt 3):1097–1109.

33. Nakamura K, Yamagishi S, Adachi H, Kurita-Nakamura Y, Matsui T, Yoshida T, Sato A, Imaizumi T. Elevation of soluble form of receptor for advanced glycation end products (sRAGE) in diabetic subjects with coronary artery disease. Diabetes Metab Res Rev. 2007; 23:368–371.

34. Hanford LE, Enghild JJ, Valnickova Z, Petersen SV, Schaefer LM, Schaefer TM, Reinhart TA, Oury TD. Purification and characterization of mouse soluble receptor for advanced glycation end products (sRAGE). J Biol Chem. 2004; 279:50019–50024.

35. Uribarri J, Woodruff S, Goodman S, Cai W, Chen X, Pyzik R, Yong A, Striker GE, Vlassara H. Advanced glycation end products in foods and a practical guide to their reduction in the diet. J Am Diet Assoc. 2010; 110:911–916.

36. Luevano-Contreras C, Chapman-Novakofski K. Dietary advanced glycation end products and aging. Nutrients. 2010; 2:1247–1265.

37. Liu G, Zong G, Hu FB, Willett WC, Eisenberg DM, Sun Q. Cooking methods for red meats and risk of type 2 diabetes: a prospective study of U.S. Women. Diabetes Care. 2017; 40:1041–1049.

38. Luevano-Contreras C, Garay-Sevilla ME, Wrobel K, Malacara JM, Wrobel K. Dietary advanced glycation end products restriction diminishes inflammation markers and oxidative stress in patients with type 2 diabetes mellitus. J Clin Biochem Nutr. 2013; 52:22–26.
39. Wu CH, Huang SM, Lin JA, Yen GC. Inhibition of advanced glycation endproduct formation by foodstuffs. Food Funct. 2011; 2:224–234.

40. Kwok JM, Ma CC, Ma S. Recent development in the effects of statins on cardiovascular disease through Rac1 and NADPH oxidase. Vascul Pharmacol. 2013; 58:21–30.

41. Ishibashi Y, Yamagishi S, Matsui T, Ohta K, Tanoue R, Takeuchi M, Ueda S, Nakamura K, Okuda S. Pravastatin inhibits advanced glycation end products (AGEs)-induced proximal tubular cell apoptosis and injury by reducing receptor for AGEs (RAGE) level. Metabolism. 2012; 61:1067–1072.

42. Yamagishi S. Possible involvement of advanced glycation end products in carry-over benefits of atorvastatin in ASCOT-BPLA. Eur Heart J. 2008; 29:1922.

43. Yamagishi S, Nakamura K, Matsui T. Potential utility of telmisartan, an angiotensin II type 1 receptor blocker with peroxisome proliferator-activated receptor-gamma (PPAR-gamma)-modulating activity for the treatment of cardiometabolic disorders. Curr Mol Med. 2007; 7:463–469.
44. Yamagishi S, Nakamura K, Matsui T. Regulation of advanced glycation end product (AGE)-receptor (RAGE) system by PPAR-gamma agonists and its implication in cardiovascular disease. Pharmacol Res. 2009; 60:174–178.

45. Coughlan MT, Thallas-Bonke V, Pete J, Long DM, Gasser A, Tong DC, Arnstein M, Thorpe SR, Cooper ME, Forbes JM. Combination therapy with the advanced glycation end product cross-link breaker, alagebrium, and angiotensin converting enzyme inhibitors in diabetes: synergy or redundancy? Endocrinology. 2007; 148:886–895.

46. Ihm SH, Chang K, Kim HY, Baek SH, Youn HJ, Seung KB, Kim JH. Peroxisome proliferator-activated receptor-gamma activation attenuates cardiac fibrosis in type 2 diabetic rats: the effect of rosiglitazone on myocardial expression of receptor for advanced glycation end products and of connective tissue growth factor. Basic Res Cardiol. 2010; 105:399–407.
47. Ojima A, Ishibashi Y, Matsui T, Maeda S, Nishino Y, Takeuchi M, Fukami K, Yamagishi S. Glucagon-like peptide-1 receptor agonist inhibits asymmetric dimethylarginine generation in the kidney of streptozotocin-induced diabetic rats by blocking advanced glycation end product-induced protein arginine methyltranferase-1 expression. Am J Pathol. 2013; 182:132–141.

48. Nakashima S, Matsui T, Takeuchi M, Yamagishi SI. Linagliptin blocks renal damage in type 1 diabetic rats by suppressing advanced glycation end products-receptor axis. Horm Metab Res. 2014; 46:717–721.

49. Matsui T, Nakashima S, Nishino Y, Ojima A, Nakamura N, Arima K, Fukami K, Okuda S, Yamagishi S. Dipeptidyl peptidase-4 deficiency protects against experimental diabetic nephropathy partly by blocking the advanced glycation end products-receptor axis. Lab Invest. 2015; 95:525–533.

50. Thornalley PJ, Yurek-George A, Argirov OK. Kinetics and mechanism of the reaction of aminoguanidine with the alpha-oxoaldehydes glyoxal, methylglyoxal, and 3-deoxyglucosone under physiological conditions. Biochem Pharmacol. 2000; 60:55–65.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download