Abstract
Purpose
Diquafosol is a pharmaceutical drug used for dry eye treatment with a novel mechanism of action. It is a purinergic P2Y2 receptor agonist that promotes the secretion of tears and healing of corneal epithelial wounds. However, its inhibitory effect on hyperosmotic stress-induced inflammation in human corneal epithelial cells (HCECs) remains unclear.
Methods
A hyperosmotic stress model was established by transferring HCECs from isosmotic (312 mOsm/kg to hyperosmotic medium (500 mOsm/kg). HCECs were incubated with 500 mOsm/kg hyperosmotic medium for 30 minutes, and then treated with diquafosol (0.6–6 mg/mL) for 4 or 24 hours. Cells were then harvested and analyzed by western blot, immunocytochemistry, and real-time polymerase chain reaction to evaluate the expression of interleukin-6, tumor necrosis factor-alpha, and the phosphorylation status of nuclear factor-kappa B.
Results
Diquafosol significantly decreased the mRNA and protein expression of hyperosmotic stress-induced tumor necrosis factor-alpha and interleukin-6. These results were supported by immunofluorescence staining and quantitative real-time polymerase chain reaction analysis. Furthermore, diquafosol inhibits nuclear factor-kappa B activation by suppressing the phosphorylation and degradation of the inhibitor of кB.
Conclusions
This study shows that diquafosol inhibits nuclear factor-kappa B signaling and inflammatory factors induced by hyperosmotic stress in HCECs. This suggests that using diquafosol for the improvement of dry eye syndrome could be effective in the treatment of inflammation-related corneal and conjunctival diseases.
Dry eye syndrome, or dry eye disease, is one of the most common reasons for patient visits to eye clinics. It is a multifactorial disease of the tears and ocular surface, resulting in discomfort, visual disturbances, and tear film instability, and it can potentially damage the ocular surface [1]. A comparison of age-specific prevalence data showed that the prevalence of dry eye syndrome ranged from 5% to more than 35% at various ages [23]. People with dry eye are significantly more likely to report problems with reading, performing professional work, computer use, and driving compared to people without dry eye [4].
A major mechanism of dry eye pathogenesis is hyperosmolarity, which is caused by tear deficiency or excessive tear evaporation or both. Hyperosmolarity stress is associated with a potent inflammatory response. Osmotic stress that is initiated by an increase in extracellular osmolarity occurs during normal cellular function in various tissues, including human conjunctival [567] and corneal epithelial cells [8910]. Tear hyperosmolarity is considered a causative factor in the ocular surface inflammation, cell damage, and irritation symptoms experienced by dry eye syndrome patients [11]. Extracellular hyperosmolarity is also believed to play a role in the inflammatory response [12]. Notably, these symptoms are also strongly associated with inflammatory cytokine secretion. Inflammatory cytokines that are linked to hyperosmotic stress-related pathologies include the tumor necrosis factor (TNF) family and interleukins [1213].
Diquafosol ophthalmic solution (3%) is a pharmaceutical drug used for the treatment of dry eye syndrome with a novel mechanism of action. It has been widely used in clinical practice to treat dry eye and is currently approved in Japan and South Korea. Diquafosol is a purinergic P2Y2 receptor agonist that promotes tear and mucin secretion in experimental dry eye models [14]. However, little is known about the therapeutic potential of diquafosol on the hyperosmotic stress-induced inflammatory response. The present study is focused on the effects of diquafosol on TNF-α and interleukin-6 (IL-6) expression in hyperosmotic stress-activated human corneal epithelial cells (HCECs).
Anti–TNF-α and anti–IL-6 antibodies were purchased from Abcam (Cambridge, MA, USA). Anti-β-actin and anti-Lamin B1 antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Antibodies against phospho-inhibitor of κB (I-κB) α, I-κBα, phospho–nuclear factor-kappa B (NF-κB) p65, and NF-κB p65 were purchased from Cell Signaling Technology (Beverly, MA, USA). Horseradish peroxidase-conjugated goat anti-rabbit and rabbit anti-goat IgG were purchased from Zymed Laboratories (San Francisco, CA, USA). The annexin V and dead cell assay kit was purchased from Merck Millipore (Billerica, MA, USA). Diquafosol was obtained from Santen (eye drops 3%; Santen Pharmaceutical, Osaka, Japan).
HCECs (2.040 pRSV-T) were purchased from the American Type Culture Collection (Manassas, VA, USA). Cells were maintained in DMEM/F12 containing 10% Fetal Bovine Serum (Gibco, Carlsbad, CA, USA), 5 µg/mL insulin, 5 µg/mL human transferrin, 5 nM selenium, and 1% penicillin/streptomycin. Cultures were incubated at 37℃ with 5% CO2. Hyperosmotic stress was induced by transferring HCECs from isosmotic (312 mOsm/kg) DMEM/F-12 growth media to hyperosmotic growth media (500 mOsm/kg).
To evaluate viability, cells were cultured in a 96-well plate and grown to 80%–90% confluence. HCECs were treated with various concentrations of diquafosol solution for 20 hours. After incubation, cell viability was determined by using the CCK-8 assay (Dojindo Laboratories, Kumamoto, Japan). Color development was measured at 450 nm using an ELISA microplate reader (Infinite M200; Tecan, Männedorf, Switzerland). Experiments were performed in triplicate. The percentage of apoptotic cells was determined with the annexin V and dead cell kit, according to the manufacturer's instructions. Briefly, harvested cells were washed with PBS and then mixed with 100 µL of the annexin V and dead cell assay kit reagents. Samples were incubated at room temperature for 20 minutes in the dark. Measurements were conducted in triplicate using a MUSE cell analyzer (Merck Millipore, Billerica, MA, USA).
To determine the a mount of mRNA expression, cells were exposed to hyperosmotic media (500 mOsm/kg DMEM/F-12, serum-free) for 30 minutes, followed by diquafosol for 4 hours, as previously described [15]. Total RNA was isolated from the cells with Trizol reagent (Life Technologies, Rockville, MD, USA), according to the manufacturer's instructions, and reverse-transcribed into complementary DNA with M-MLV reverse transcriptase (Promega, Madison, WI, USA). Real-time polymerase chain reaction (PCR) was performed using SYBR Premix Ex Taq (Perfect Real Time) Premix (Takara Bio, Otsu, Japan) and Takara Thermal Cycler Dice (TP850), according to the manufacturer's protocol (Takara Bio, Shiga, Japan). Relative quantification of mRNA expression was performed using TP850 software. Table 1 shows the gene-specific primers used in this study (Macrogen, Seoul, Korea). PCR products were electrophoresed on 1% agarose gels and visualized by GreenLight (BioAssay Co., Daejeon, Korea). PCR conditions are indicated in Table 1. All experiments were performed in triplicate.
To determine the expression of proteins, cells were exposed to hyperosmotic media (500 mOsm/kg DMEM/F-12, serum-free) for 30 minutes, followed by diquafosol for 24 hours. Protein extraction and western blotting were performed as described previously [15]. Membranes were incubated overnight at 4℃ with polyclonal antibodies against TNF-α and IL-6 and with a monoclonal antibody against β-actin in 0.1% Tween-20 Tris-buffered saline (TBS) containing 5% nonfat dried milk.
Membranes were then incubated with the appropriate horseradish peroxidase-conjugated secondary antibodies for 1 hour. Antibody binding was visualized using an enhanced chemiluminescence detection kit (ELPIS Biotech, Daejeon, Korea) and exposure to X-ray film. The experiments were performed in triplicate. Quantification of band intensity was performed using ImageJ software ver. 1.52 (National Institutes of Health, Bethesda, MD, USA).
To confirm the activation of NF-κB, cells were exposed to hyperosmotic media (500 mOsm/kg DMEM/F-12, serum- free) for 30 minutes, followed by diquafosol for 30 minutes. Proteins from the cytoplasm and nucleus were separated by using the NE-PER nuclear and cytoplasmic extraction reagent kit (#78835; ThermoFisher Scientific, Waltham, MA, USA). Brief ly, HCECs were harvested, cells were centrifuged at 16,000 × g for 5 minutes, and the supernatant was removed. Ice-cold CER-I and -II solutions were added to the pellet per the manufacturer's instructions to separate cytoplasmic proteins from nuclear-compartment proteins. Western blots for NF-κB, p-NF-κB, I-κB, p-I-κB, β-actin (as a cytosol marker) and Lamin B1 (as a nuclear marker) were performed to ensure that there was no contamination.
Harvested cells were fixed with 10% neutral buffered formalin solution at room temperature for 10 minutes. Cells were per meabilized wit h 0.3% Tr iton X-100 for 15 minutes and blocked overnight with 5% normal goat serum and bovine serum albumin in TBS at 4℃. Cells were then incubated with primary antibodies specific for TNF-α, IL-6, and p-NF-κB (diluted 1 : 50 to 1 : 100) in 5% bovine serum albumin overnight at 4℃ and washed three times in TBS. Cells were then incubated with the appropriate Alexa Fluor 488- or 555-conjugated secondary antibody at room temperature for 1 hour in the dark; nuclei were then counterstained with 4′,6-diamidino-2-phenylindole (DAPI, 1 µg/mL). Slides were mounted with ProLong Gold (ThermoFisher Scientific, Waltham, MA, USA), and images were captured by fluorescence microscopy (Axio Vision 4; Carl Zeiss, Jena, Germany). Fluorescence intensity was measured using color histograms generated with ImageJ software.
Data were evaluated by one-way analysis of variance using Tukey's post-hoc test. Analyses were performed using GraphPad PRISM software ver. 5.02 (GraphPad PRISM Software Inc., La Jolla, CA, USA). The criterion for statistical significance was set at p < 0.05. All statistical tests were two-tailed.
To investigate the cytotoxicity of diquafosol ophthalmic solution, we measured cell viability using the CCK-8 assay kit. Diquafosol displayed a dose-dependent toxicity in HCECs under starved conditions. No significant toxicity was observed at diquafosol concentrations of up to 6 mg/mL. However, cell viability notably decreased when HCECs were exposed to 30 mg/mL of diquafosol (Fig. 1A). We also measured the effects of diquafosol on apoptosis. Relative to control cells, the percentage of apoptotic HCECs increased with diquafosol in a dose-dependent manner, but the change was small and not significant (Fig. 1B).
To evaluate the anti-inflammatory activity of diquafosol, we investigated the effects of diquafosol on hyperosmotic stress-induced inflammatory gene expression. Real-time PCR analysis showed an increase in hyperosmotic stress-induced TNF-α and IL-6 mRNA concentration (Fig. 2A). However, pretreatment with diquafosol (1.5–6 mg/mL) resulted in the attenuation of the inflammatory response. In addition, western blot results revealed that TNF-α and IL-6 protein levels were markedly decreased by diquafosol in hyperosmotic stress-induced inflammation (Fig. 2B).
Non-hyperosmotic stress-stimulated cells exhibited weak staining, suggesting low basal levels of TNF-α and IL-6 (Fig. 3). After hyperosmotic stress stimulation, immunocytochemistry analysis revealed that TNF-α and IL-6 expression increased significantly. Diquafosol appears to block expression of TNF-α and IL-6 proteins in HCECs in response to hyperosmotic stress.
The NF-κB signaling pathway contributes to the inflammatory process of dry eye, so we investigated the effects of diquafosol on hyperosmotic stress-induced NF-κB activation. Phosphorylation of I-κB and nuclear translocation of NF-κB p65 were notably increased by hyperosmotic stress, whereas treatment with diquafosol decreased the hyperosmotic stress-induced levels of p-I-κB and nuclear translocation of NF-κB p65 in a dose-dependent manner (Fig. 4A). Hyperosmotic stress resulted in the translocation of p-NF-κB-p65 to the nucleus as seen by immunocytochemistry. Incubation with anti-NF-κB-p65 showed marked nuclear staining in HCECs under hyperosmotic stress conditions, whereas cells treated with diquafosol (6 mg/mL) showed only background staining (Fig. 4B). The action of diquafosol through inhibiton of NF-κB activity is schematically summarized (Fig. 4C).
Dry eye disease is the most common disorder of the eye, and its associated symptoms can cause corneal injury [16]. Dry eye disease is a condition caused by deficient tear production or an excessive loss of water from the tear film by evaporation. Increased tear osmolarity has been recognized as a hallmark of dry eye syndrome, and it appears to play an important role in the pathogenesis of ocular surface damage. Tear osmolarity has been reported to be the single best marker for dry eye disease [1718]. Tear film hyperosmolarity may cause pathological changes in the corneal epithelium, such as desquamation, fewer intercellular connections, loss of microplicae, cell membrane disruptions, and cellular swelling with decreased cytoplasmic density [19]. Other studies have reported that hyperosmotic stress not only stimulates the expression and production of inflammatory cytokines TNF-α, IL-8, and IL-1 in HCECs in vitro [89], but also stimulates the expression of these inflammatory mediators in an experimental dry eye mouse model in vivo [20]. Other studies have also shown that hyperosmolarity induces the expression of inflammatory cytokines such as TNF-α, IL-6, and monocyte chemotactic protein-1 (MCP-1) in cultured HCECs [1021]. In addition, an increase in these proteins has been seen in an HCEC culture model, an in vivo murine dry eye model, and in the tears of dry eye patients [2223].
The protective properties of diquafosol have been recognized in the treatment of dry eyes. Diquafosol stimulates the production of water, lipids, and mucin by activating the purinergic (P2Y2) receptor on the ocular surface [24]. However, little is known about the bioactivity and potential clinical implications of diquafosol on hyperosmotic stress-induced inflammation. In this study, we present evidence that diquafosol may exert beneficial effects on dry eye-mediated ocular inflammation. We show that diquafosol significantly suppressed the expression of inflammatory cytokines TNF-α and IL-6 in HCECs exposed to hyperosmotic stress. Our f indings demonstrate that hyperosmotic stress induces inflammation, and diquafosol is an effective agent that protects HCECs from hyperosmotic stress-induced inflammation. These results are consistent with previous studies in which the secretion of inf lammatory cytokines was signif icantly reduced in desiccation stress-induced dry eye models [25]. Results showing that diquafosol inhibits the production of inflammatory cytokines in a variety of dry eye models suggest that diquafosol may exert anti-inflammatory effects on various pathogenic factors associated with dry eye syndrome, such as tear hyperosmolarity or dry conditions.
NF-κB is a key transcription factor in the inflammatory response of various cells, and it could play an important role in the production of cytokines that have been implicated in the pathogenesis of dry eye disease. NF-κB dimers are retained in an inactive form in the cytosol, bound by I-κB proteins. Under various stimuli, including hyperosmotic stress, phosphorylation, and subsequent degradation of I-κB proteins occurs, allowing NF-κB to translocate to the nucleus where it activates various inflammatory genes [26]. In addition, phosphorylation of NF-κB subunits affects NF-κB transcriptional responses. Phosphorylation of t he p65 subunit of NF-κB leads to t he selective t ranscription of downstream inf lammatory cytokines [27]. Desiccating stress causes NF-κB activation in an in vivo model of dry eye disease; conversely, NF-κB inhibitors reduce IL-1β and IL-6 production and corneal epithelial damage in mice [28]. Through in vitro experiments using HCEC cells, we confirmed that NF-κB phosphorylation and nuclear localization are responses to hyperosmotic stress. Our results suggest that the activated NF-κB signaling pathways in our experimental dry eye model may play a role in the induction of inflammatory cytokines IL-6 and TNF-α. Previous studies reported that NF-κB inhibitors are useful for treating dry eye disorders and other disorders requiring the wetting of the eye. In our study, hyperosmotic stress-induced expression of p-NF-κB and p-I-κB and degradation of I-κB were blocked by diquafosol, which agrees with the results of immunocytochemistry analyses that show a significant nuclear translocation of p-NF-κB.
Our findings suggest potential benefits of diquafosol treatment in ocular surface disorders such as dry eye. Diquafosol partially protects the human corneal epithelium from hyperosmotic stress-induced inflammatory responses by reducing TNF-α and IL-6 levels. Diquafosol is a purinergic P2Y2 receptor agonist and enhances the corneal barrier function by increasing tear fluid secretion in dry eye disease [29]. However, inflammatory cytokines also contribute to dry eye disease [30]. Therefore, the inhibition of IL-6 and TNF-α protein expression indicates that diquafosol treatment may also improve dry eye symptoms, at least partly, through anti-inflammatory mechanisms. NF-κB inhibitors have been reported to inhibit pro-inflammatory cytokine production as well as reverse corneal epithelial damage [28]. A correlation between NF-κB inhibition and corneal epithelial recovery suggests that the anti-inflammatory effects of diquafosol can act as a partial mechanism to improve dry eye symptoms.
Figures and Tables
 | Fig. 1Effects of diquafosol (DQF) on the (A) viability and (B) apoptosis of human corneal epithelial cells. Cells were treated with different concentrations of DQF solution. After 20 hours, the apoptosis rate was assessed using the CCK-8 assay kit or the annexin V and dead cell assay kit. Results are expressed as the percentage of surviving cells over control cells. Data are expressed as the mean ± standard deviation from three separate experiments (p < 0.01 and p < 0.001, significantly different from the control). |
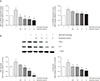 | Fig. 2Diquafosol downregulates mRNA and protein expression of tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) in human corneal epithelial cells. (A) Total RNA was extracted from human corneal epithelial cells. RNA levels were measured by real-time polymerase chain reaction. The cells were exposed to hyperosmotic media (500 mOsm/kg DMEM/F12, serum-free) for 30 minutes, followed by diquafosol (0.6–6 mg/mL) for 4 hours. The graph of multiple analyses shows the relative mRNA levels of TNF-α and IL-6. Glyceraldehyde-3-phosphate dehydrogenase was used as the reference gene. (B) Representative western blots of TNF-α and IL-6. Cells were exposed to hyperosmotic media (500 mOsm/kg DMEM/F12, serum-free) for 30 minutes, followed by diquafosol (0.6–6 mg/mL) for 24 hours. Expression levels of TNF-α and IL-6 were determined using β-actin as a control. The densities of bands relative to those of β-actin were measured using ImageJ software. Data are expressed as the mean ± standard deviation from three separate experiments (*p < 0.01 and **p < 0.01, significantly different from the control; #p < 0.05, ##p < 0.01, and ###p < 0.001, significantly different from the hyperosmotic group). HOS = hyperosmotic stress. |
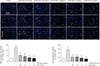 | Fig. 3Fluorescence immunocytochemistry for tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) in human corneal epithelial cells. Cells were exposed to hyperosmotic media (500 mOsm/kg DMEM/F12, serum-free) for 30 minutes, followed by diquafosol (DQF, 0.6–6 mg/mL) for 24 hours. Changes in the expression of cytoplasmic TNF-α and IL-6 were confirmed using fluorescence immunocytochemistry. Cells were counterstained with DAPI. The fluorescence intensity from green to blue was measured in ImageJ software using a color histogram. White scale bars: 100 µm (***p < 0.001, significantly different from the control; ###p < 0.001, significantly different from the hyperosmotic group). HOS = hyperosmotic stress. |
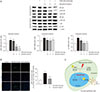 | Fig. 4Effects of diquafosol on the nuclear factor-kappa B (NF-κB) signaling pathway in human corneal epithelial cells. (A) Expression levels of NF-κB, p-NF-κB, I-κB, and p-I-κB proteins in nuclear and cytosolic fractions were assessed using western blotting. The cells were exposed to hyperosmotic media (500 mOsm/kg DMEM/F12, serum-free) for 30 minutes, followed by diquafosol (1.5–6 mg/mL) for 30 minutes. Lamin B1 and ß-actin were used as standard proteins for quantitating the levels of the proteins of interest. (B) Expression of p-NF-κB in the nuclear fraction was confirmed using fluorescence immunocytochemistry. Cells were counterstained with DAPI. Fluorescence intensity and the density of bands were measured using ImageJ software. (C) Schematic diagram showing the inhibitory effects of diquafosol on hyperosmotic stress-induced inflammatory cytokine production via the NF-κB signaling pathway in human corneal epithelial cells. Data are expressed as the mean ± standard deviation from three separate experiments (***p < 0.001, significantly different from the control; #p < 0.05, ##p < 0.01 and ###p < 0.001, significantly different from the hyperosmotic group). HOS = hyperosmotic stress. |
Acknowledgements
This study was supported by the Kyungpook National University Research Fund (2017) and the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (2015R1D1A1A01060777). This study was partially supported by a grant from the Dongguk University Research Fund (2017).
References
1. Gulati S, Jain S. Ocular pharmacology of tear film, dry eye, and allergic conjunctivitis. Handb Exp Pharmacol. 2017; 242:97–118.


2. McCarty CA, Bansal AK, Livingston PM, et al. The epidemiology of dry eye in Melbourne, Australia. Ophthalmology. 1998; 105:1114–1119.

3. Kuang TM, Tsai SY, Hsu WM, et al. Correctable visual impairment in an elderly Chinese population in Taiwan: the Shihpai Eye Study. Invest Ophthalmol Vis Sci. 2007; 48:1032–1037.


4. Miljanovic B, Dana R, Sullivan DA, Schaumberg DA. Impact of dry eye syndrome on vision-related quality of life. Am J Ophthalmol. 2007; 143:409–415.


5. Versura P, Profazio V, Schiavi C, Campos EC. Hyperosmolar stress upregulates HLA-DR expression in human conjunctival epithelium in dry eye patients and in vitro models. Invest Ophthalmol Vis Sci. 2011; 52:5488–5496.


6. Bellotti M, Bast W, Berra A, Bonetto FJ. Effects of osmolarity on human epithelial conjunctival cells using an electrical technique. Graefes Arch Clin Exp Ophthalmol. 2011; 249:1875–1882.


7. Julio G, Lluch S, Pujol P, Merindano MD. Effects of tear hyperosmolarity on conjunctival cells in mild to moderate dry eye. Ophthalmic Physiol Opt. 2012; 32:317–323.


8. Li DQ, Chen Z, Song XJ, et al. Stimulation of matrix metalloproteinases by hyperosmolarity via a JNK pathway in human corneal epithelial cells. Invest Ophthalmol Vis Sci. 2004; 45:4302–4311.


9. Li DQ, Luo L, Chen Z, et al. JNK and ERK MAP kinases mediate induction of IL-1beta, TNF-alpha and IL-8 following hyperosmolar stress in human limbal epithelial cells. Exp Eye Res. 2006; 82:588–596.
10. Cavet ME, Harrington KL, Ward KW, Zhang JZ. Mapracorat, a novel selective glucocorticoid receptor agonist, inhibits hyperosmolar-induced cytokine release and MAPK pathways in human corneal epithelial cells. Mol Vis. 2010; 16:1791–1800.


11. Al-Ayyoubi S, Gali-Muhtasib H. Differential apoptosis by gallotannin in human colon cancer cells with distinct p53 status. Mol Carcinog. 2007; 46:176–186.


12. Neuhofer W. Role of NFAT5 in inflammatory disorders associated with osmotic stress. Curr Genomics. 2010; 11:584–590.



13. Schwartz L, Guais A, Pooya M, Abolhassani M. Is inflammation a consequence of extracellular hyperosmolarity? J Inflamm (Lond). 2009; 6:21.



14. Nichols KK, Yerxa B, Kellerman DJ. Diquafosol tetrasodium: a novel dry eye therapy. Expert Opin Investig Drugs. 2004; 13:47–54.


15. Gum SI, Kim YH, Jung JC, et al. Cyclosporine A inhibits TGF-β2-induced myofibroblasts of primary cultured human pterygium fibroblasts. Biochem Biophys Res Commun. 2017; 482:1148–1153.


16. Kanellopoulos AJ, Asimellis G. In pursuit of objective dry eye screening clinical techniques. Eye Vis (Lond). 2016; 3:1.


17. Lemp MA, Bron AJ, Baudouin C, et al. Tear osmolarity in the d iagnosis and management of d ry eye d isease. Am J Ophthalmol. 2011; 151:792–798.

18. Corrales RM, Villarreal A, Farley W, et al. Strain-related cytokine profiles on the murine ocular surface in response to desiccating stress. Cornea. 2007; 26:579–584.


19. Gilbard JP, Carter JB, Sang DN, et al. Morphologic effect of hyperosmolarity on rabbit corneal epithelium. Ophthalmology. 1984; 91:1205–1212.


20. Luo L, Li DQ, Corrales RM, Pflugfelder SC. Hyperosmolar saline is a proinflammatory stress on the mouse ocular surface. Eye Contact Lens. 2005; 31:186–193.

21. Pan Z, Wang Z, Yang H, et al. TRPV1 activation is required for hypertonicity-stimulated inflammatory cytokine release in human corneal epithelial cells. Invest Ophthalmol Vis Sci. 2011; 52:485–493.



22. Lam H, Bleiden L, de Paiva CS, et al. Tear cytokine profiles in dysfunctional tear syndrome. Am J Ophthalmol. 2009; 147:198–205.


23. Enriquez-de-Salamanca A, Castellanos E, Stern ME, et al. Tear cytokine and chemokine analysis and clinical correlations in evaporative-type dry eye disease. Mol Vis. 2010; 16:862–873.
24. Koh S. Clinical utility of 3% diquafosol ophthalmic solution in the treatment of dry eyes. Clin Ophthalmol. 2015; 9:865–872.



25. Park JH, Moon SH, Kang DH, et al. Diquafosol sodium inhibits apoptosis and inf lammation of corneal epithelial cells via activation of Erk1/2 and RSK: in vitro and in vivo dry eye model. Invest Ophthalmol Vis Sci. 2018; 59:5108–5115.
26. Lan W, Petznick A, Heryati S, et al. Nuclear Factor-κB: central regulator in ocular surface inflammation and diseases. Ocul Surf. 2012; 10:137–148.


27. Shi H, Berger EA. Characterization of site-specific phosphorylation of NF-κB p65 in retinal cells in response to high glucose and cytokine polarization. Mediators Inflamm. 2018; 2018:3020675.

28. Guzman M, Keitelman I, Sabbione F, et al. Desiccating stress-induced disruption of ocular surface immune tolerance drives dry eye disease. Clin Exp Immunol. 2016; 184:248–256.







 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download