Abstract
Hemosiderosis is characterized by the deposition of excess iron in body tissues. The choroid plexus is an important part of the central nervous system that can be the primary site of iron overload. T2*-weighted gradient echo (GRE) sequence provides high sensitivity for demonstrating cerebral microhemorrhagic foci and iron deposition. In the present study, we describe the case of a 15-year-old boy with acute lymphoblastic leukemia, in whom repeated transfusion led to iron accumulation in the brain. GRE sequence effectively demonstrated hemosiderin deposition in the choroid plexus.
Hemosiderosis is a primary or secondary condition characterized by the deposition of excess iron, which occurs predominantly in hepatocytes and in the reticuloendothelial system (spleen, bone marrow, and liver). When these organs get saturated, deposition occurs in other tissues (1). Excessive iron deposition in the central nervous system (CNS) is rare, though it does sometimes occur in choroid plexus, the pituitary gland, the cortical surface, and less frequently in the basal ganglia (123).
CNS hemosiderosis is usually asymptomatic, but if not diagnosed early, it can cause pituitary dysfunction or neurologic sequelae (4567). In the past, the autopsy was the only diagnostic method for this disease. Recent advancements in magnetic resonance imaging (MRI) technique have enabled effective diagnosis of these diseases. In particular, T2*-weighted gradient-echo (GRE) imaging is a highly sensitive and specific diagnostic modality for hemosiderosis (4567).
Herein, we describe MRI findings of a child with acute lymphoblastic leukemia, in whom repeated transfusion led to iron accumulation in the choroid plexus and place emphasis on the role of GRE sequence for the detection of CNS iron deposition.
A 15-year-old boy was admitted to our pediatric department with complaints of dizziness, headache, and vomiting, which had developed in the past one month before admission. Two years back, he was diagnosed with acute lymphoblastic leukemia and treated by chemotherapy and received more than 40 red blood cell transfusions.
A physical examination at the time of admission revealed no remarkable vestibular function finding and his cerebrospinal fluid examination was normal. Laboratory findings revealed a remarkably elevated level of serum ferritin (3412 ng/mL, normal range 13 to 400 ng/mL). However, serum iron (183 mg/dL, normal range 33 to 193 mg/dL) and unsaturated iron-binding capacity (218 mg/dL, normal range 230 to 435 mg/dL) were essentially normal.
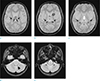
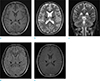
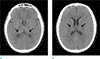
A brain MR examination was performed in a 1.5T machine (Signa HDxt, GE Healthcare, Milwaukee, WI, USA). Brain MRI revealed hypointensity in the choroid plexus of both the lateral ventricles, 3rd ventricle, 4th ventricle, and both foramina of Luschka on GRE (Fig. 1). There were no definite hypointensities suggesting iron deposition in the pituitary gland, pineal gland, and area postrema, although there were limitations due to 2D GRE acquisition and susceptibility artifacts in the parasellar area (Fig. 1). Axial T1- (T1WI) (Fig. 2a) and T2-weighted images (T2WI) (Fig. 2b, c), fluid-attenuated inversion recovery (FLAIR) (Fig. 2d), and postcontrast T1WI (Fig. 2e) showed no remarkable findings. Brain computed tomography (CT) performed a day later demonstrated no calcification or hemorrhage in choroid plexus (Fig. 3). The pituitary hormone function test was not performed because there were no symptoms suggestive of pituitary dysfunction. Other blood tests, including liver function tests were normal, so no other imaging tests were performed.
The patient was treated conservatively. On admission day 5, improvement in his clinical condition was noted and he was discharged.
Hemosiderosis or iron overload is a pathological condition characterized by deposition of excess iron (hemosiderin) in body tissues (1). Hemosiderosis can be classified as genetic or primary and as acquired or secondary hemosiderosis. Secondary hemosiderosis arises due to excessive iron absorption, repeated blood transfusions, or excessive oral intake (1). In particular, modern intensive combination chemotherapy for patients with childhood acute lymphoblastic leukemia requires more frequent red blood cell transfusions with the resultant increase in iron burden to unacceptable levels (89).
Iron overload initially results in iron accumulations in hepatocytes and macrophages of the reticuloendothelial system (spleen, bone marrow, and liver) and under the condition of saturation, deposition occurs in others tissues (1). In brain, iron deposition is typically observed at sites outside the blood-brain barrier, which include the pituitary gland, choroid plexus, pineal gland, and area postrema (23).
The choroid plexus is a highly vascularized papillary structure that protrudes into the ventricles. The CNS is separated from the systemic circulation by the blood-brain barrier, formed by capillary endothelium and the blood-cerebrospinal fluid barrier generated by choroid plexus epithelium. Capillaries of the choroid plexus are fenestrated, non-continuous, and permeable to small molecules and water. However, adjacent choroidal epithelial cells form tight junctions that selectively prevent the passage of ions and macromolecules (3). According to the autoradiographic distribution of 59Fe in adult rat brain, iron is selectively taken up by choroid plexus and extra-blood-brain barriers from the blood (2). Apparently, choroid plexus is hypothesized to play a role in protecting the brain from iron overloading through the buffering mechanism (2).
Transfusion related iron overload among pediatric oncology patients has been reported to be 19-40% (89). Factors affecting iron burden in different studies are the high-risk disease, the total amount of blood transfused, intensity of treatment, age of the patient, and body surface area (89). Iron deposits can occur anywhere in the body, but involvement of choroid plexus is an extremely rare condition with very few reported cases in the literature (456). The exact incidence remains unknown and relatively little is known about the natural history or prognosis of choroid plexus hemosiderosis. In previously reported cases, choroid plexus hemosiderosis in transfusion dependent patients was usually asymptomatic, before the appearance of endocrine signs due to pituitary involvement (456). A case of a patient with choroid plexus hemosiderosis who complained of headache has been reported (5). However, it should not be assumed that iron deposition in choroid plexus is a cause of the patient's symptoms as this may be an incidental finding. Since choroid plexus plays a role in preventing iron accumulation in the brain, the presence of choroid plexus hemosiderosis can be considered as an early signal of CNS iron deposition. Considering that hemosiderosis may ultimately lead to irreversible organ damage, its early identification in choroid plexus or the pineal gland is of importance, even if the patient is still asymptomatic (4567). Brain MRI including gradient echo (GRE) sequence demonstrated increased sensitivity in detecting CNS hemosiderosis due to its ability to detect hemosiderin based on the susceptibility effect. In our case, GRE images showed diffuse hypointensity in the choroid plexus of both the lateral ventricles, 3rd ventricle, 4th ventricle, and both the foramina of Luschka, which are underestimated on T2WI. Accordingly, we found GRE sequence superior to T2WI in this respect, as has been previously reported (34567), which makes it a better proposition for the detection of choroid plexus hemosiderosis. In a similar context, susceptibility weighted imaging is a velocity-compensated, GRE sequence that combines magnitude and phase information to accentuate the visibility of susceptible foci and is more sensitive than conventional GRE sequences for detecting microhemorrhages, subarachnoid hemorrhage, deoxygenated blood, blood degradation products, calcium, and iron deposits in brain (710). Though the technique was not performed in our patient, this sequence may be helpful in patients with suspicion of iron deposition in the CNS.
In conclusion, we present the brain MRI findings of choroid plexus hemosiderosis in a 15-year-old male patient with ALL, who had received repeated blood transfusions. Considering that hemosiderosis may ultimately lead to irreversible organ damage, its early identification in choroid plexus or the pituitary gland is of importance, even if the patient is still asymptomatic. GRE sequence enables early diagnosis of CNS hemosiderosis and provides a means of fully evaluating the distribution of hemosiderin deposition.
Figures and Tables
Fig. 1
MR images of choroid plexus hemosiderosis in a 15-year-old male with acute lymphoblastic leukemia in whom repeated blood transfusions led to iron overload. (a-e) Axial T2*-weighted gradient echo images demonstrate the presence of hypointense hemosiderin deposits in the choroid plexus of both the lateral ventricles (black arrows), both the foramina of Monro (open arrow), 3rd ventricle (white arrow), 4th ventricle (arrowhead), and both the foramina of Luschka (asterisks).
References
1. Fleming RE, Ponka P. Iron overload in human disease. N Engl J Med. 2012; 366:348–359.
2. Morris CM, Keith AB, Edwardson JA, Pullen RG. Uptake and distribution of iron and transferrin in the adult rat brain. J Neurochem. 1992; 59:300–306.
3. Rouault TA, Zhang DL, Jeong SY. Brain iron homeostasis, the choroid plexus, and localization of iron transport proteins. Metab Brain Dis. 2009; 24:673–684.
4. Duprez T, Maiter D, Cosnard G. Transfusional hemochromatosis of the choroid plexus in beta-thalassemia major. J Comput Assist Tomogr. 2001; 25:487–488.
5. Sossa DE, Chiang F, Verde AR, Sossa DG, Castillo M. Transfusional iron overload presenting as choroid plexus hemosiderosis. JBR-BTR. 2013; 96:39.
6. Kira R, Ohga S, Takada H, Gondo K, Mihara F, Hara T. MR choroid plexus sign of iron overload. Neurology. 2000; 55:1340.
7. Fearnley JM, Stevens JM, Rudge P. Superficial siderosis of the central nervous system. Brain. 1995; 118(Pt 4):1051–1066.
8. Eng J, Fish JD. Insidious iron burden in pediatric patients with acute lymphoblastic leukemia. Pediatr Blood Cancer. 2011; 56:368–371.
9. Halonen P, Mattila J, Suominen P, Ruuska T, Salo MK, Makipernaa A. Iron overload in children who are treated for acute lymphoblastic leukemia estimated by liver siderosis and serum iron parameters. Pediatrics. 2003; 111:91–96.
10. Haacke EM, DelProposto ZS, Chaturvedi S, et al. Imaging cerebral amyloid angiopathy with susceptibility-weighted imaging. AJNR Am J Neuroradiol. 2007; 28:316–317.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download