INTRODUCTION
METHODS
Patients
RNA extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
Immunohistochemistry and pathologic analysis
Statistical analysis
RESULTS
Patient characteristics
Table 1
Association between EC Notch1 and miR-34a expression and clinicopathological characteristics in 114 patients with TNBC

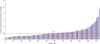 | Figure 1Quantitative real-time polymerase chain reaction analysis of miR-34a expression levels in 114 TNBC tissues. The relative miR-34a expression level (value of 2−∆Ct) in each sample was calculated based on a comparative Ct method with normalization to RNU6B RNA. Each data point represents the mean 2−∆Ct values from 3 independent assays.miR-34a = miRNA-34a; TNBC = triple-negative breast cancer; RNU6B = U6 small nuclear 6B.
|
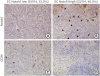 | Figure 2Immunohistochemical staining of CD34 and Notch1 in microvessels of TNBC tissues. Representative staining of Notch1 immunostaining (DAB substrate, brown) in (A) EC Notch1 low group and (B) EC Notch1 high group (original magnification ×200). Representative staining of CD34 immunostaining (DAB substrate, brown) in (C) EC Notch1 low group and (D) EC Notch1 high group (original magnification ×200). Arrowheads mark microvessels (Notch1-positive).TNBC = triple-negative breast cancer; DAB = diaminobenzidine; EC Notch1 = the ratio of Notch1 positive MVD to CD34 MVD; EC = endothelial cell; MVD = microvessel density.
|
 | Figure 3(A) The distribution of EC Notch1 and (B) expression level of EC Notch1 in 114 TNBC patients.EC Notch1 = the ratio of Notch1 positive MVD to CD34 MVD; EC = endothelial cell; MVD = microvessel density; TNBC = triple-negative breast cancer.
*p < 0.001.
|
Survival outcomes associated with expression of EC Notch1 and miR-34a
 | Figure 5Association between EC Notch1 and survival outcomes in TNBC.(A) OS, (B) DFS, (C) MFS in patients with TNBC; based on EC Notch1 expression level. (D) OS, (E) DFS, (F) MFS in patients with locally advanced TNBC; based on EC Notch1 expression level.
EC Notch1 = the ratio of Notch1 positive MVD to CD34 MVD; EC = endothelial cell; MVD = microvessel density; TNBC = triple-negative breast cancer; OS = overall survival; DFS = disease-free survival; MFS = metastasis-free survival; LN = lymph node.
|
 | Figure 6Association between miR-34a expression and survival outcomes in TNBC.(A) OS, (B) DFS, (C) MFS in patients with TNBC; based on miR-34a expression level. (D) OS, (E) DFS, (F) MFS in patients with locally advanced TNBC; based on miR-34a expression level.
miR-34a = miRNA-34a; TNBC = triple-negative breast cancer; OS = overall survival; DFS = disease-free survival; MFS = metastasis-free survival; LN = lymph node.
|
Combination of EC Notch1 and miR-34a, and their association with prognosis
 | Figure 8Kaplan–Meier analysis of survival of patients with TNBC stratified according to the expression of miR-34a and EC Notch1.(A) OS, (B) DFS, (C) MFS in patients with TNBC; based on the expression of miR-34a and EC Notch. (D) OS, (E) DFS, (F) MFS in patients with locally advanced TNBC; based on the expression of miR-34a and EC Notch1.
TNBC = triple-negative breast cancer; miR-34a = miRNA-34a; EC Notch1 = the ratio of Notch1 positive MVD to CD34 MVD; EC = endothelial cell; MVD = microvessel density; OS = overall survival; DFS = disease-free survival; MFS = metastasis-free survival; LN = lymph node.
|
Table 2
Univariate and multivariate analysis of survival outcomes





 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download