Abstract
Purpose
To evaluate the effect of platelet rich plasma (PRP) on ocular surface damage caused by hyperosmotic conditions using retrospective clinical and experimental analyses.
Methods
Eighty eyes of moderate dry eye syndrome patients who had no responses using conventional treatments were included in the study. Before and 1, 3, and 6 months after the use of autologous PRP, the visual acuity, intraocular pressure, tear break-up time (TBUT), ocular staining score (OSS), and ocular surface disease index (OSDI) were compared. The changes in inflammatory factors of ocular surface cells were analyzed using a corneo-limbal epithelial cell culture and a hyperosmotic stress experimental model.
Results
Using retrospective clinical analyses, in 64 eyes (80%) after the use of autologous PRP, the symptom scores and symptoms were significantly reduced in the OSDI questionnaire when compared with the symptom scores and symptoms before treatment. The TBUT and OSS, which were objective indicators showed a significant increase of TBUT and significant decrease of OSS in 68 eyes (85%) and 72 eyes (90%), respectively. The expression of inflammatory factors such as interleukin-1, tumor necrosis factor-α, metalloproteinase (MMP)-1, and MMP-3 decreased in corneo-limbal epithelial cells under hyperosmotic conditions when PRP was added.
Conclusions
The use of autologous PRP showed significant improvement before and after treatment in the TBUT, OSS, symptom scores and symptoms, and OSDI. In addition, anti-inflammatory effects were demonstrated in hyperosmotic models simulating dry eye syndrome. Therefore, autologous PRP could be used effectively for the treatment of moderate dry eye syndrome.
References
1. Miljanović B, Dana R, Sullivan DA, Schaumberg DA. Impact of dry eye syndrome on vision-related quality of life. Am J Ophthalmol. 2007; 143:409–15.

2. Tong L, Waduthantri S, Wong TY, et al. Impact of symptomatic dry eye on vision-related daily activities. the Singapore Malay Eye Study. Eye (Lond). 2010; 24:1486–91.

3. Gupta A, Monroy D, Ji Z, et al. Transforming growth factor beta-1 and beta-2 in human tear fluid. Curr Eye Res. 1996; 15:605–14.

4. Nishida T, Nakamura M, Ofuji K, et al. Synergistic effects of abdominal P with insulin-like growth factor-1 on epithelial migration of the cornea. J Cell Physiol. 1996; 169:159–66.
5. Watanabe K, Nakagawa S, Nishida T. Stimulatory effects of abdominal and EGF on migration of corneal epithelial cells. Invest Ophthalmol Vis Sci. 1987; 28:205–11.
6. Barton K, Nava A, Monroy DC, Pflugfelder SC. Cytokines and tear function in ocular surface disease. Adv Exp Med Biol. 1998; 438:461–9.

7. Fukuda M, Fullard RJ, Willcox MD, et al. Fibronectin in the tear film. Invest Ophthalmol Vis Sci. 1996; 37:459–67.
8. Lopez Bernal D, Ubels JL. Artificial tear composition and abdominal of recovery of the damaged corneal epithelium. Cornea. 1993; 12:115–20.
9. Pancholi S, Tullo A, Khaliq A, et al. The effects of growth factors and conditioned media on the proliferation of human corneal abdominal cells and keratocytes. Graefes Arch Clin Exp Ophthalmol. 1998; 236:1–8.
10. Tsubota K, Xu KP, Fujihara T, et al. Decreased reflex tearing is abdominal with lymphocytic infiltration in lacrimal glands. J Rheumatol. 1996; 23:313–20.
11. Wilson SE, Liang Q, Kim WJ. Lacrimal gland HGF, KGF, and EGF mRNA levels increase after corneal epithelial wounding. Invest Ophthalmol Vis Sci. 1999; 40:2185–90.
12. Hartwig D, Harloff S, Liu L, et al. Epitheliotrophic capacity of a growth factor preparation produced from platelet concentrates on corneal epithelial cells: a potential agent for the treatment of ocular surface defects? Transfusion. 2004; 44:1724–31.

13. Knighton DR, Ciresi K, Fiegel VD, et al. Stimulation of repair in chronic, nonhealing, cutaneous ulcers using platelet derived wound healing formula. Surg Gynecol Obstet. 1990; 170:56–9.
14. Lynch SE, Colvin RB, Antoniades HN. Growth factors in wound healing. Single and synergistic effects in partial thickness porcine skin wounds. J Clin Invest. 1989; 84:640–6.
15. Alio JL, Abad M, Artola A, et al. Use of autologous platelet-rich plasma in the treatment of dormant corneal ulcers. Ophthalmology. 2007; 114:1286–93.e1.

16. Alio JL, Pastor S, Ruiz-Colecha J, et al. Treatment of ocular abdominal syndrome after LASIK with autologous platelet-rich plasma. J Refract Surg. 2007; 23:617–19.
17. Alio JL, Colecha JR, Pastor S, et al. Symptomatic dry eye treatment with autologous platelet-rich plasma. Ophthalmic Res. 2007; 39:124–9.

18. Kim KM, Shin YT, Kim HK. Effect of autologous platelet-rich plasma on persistent corneal epithelial defect after infectious keratitis. Jpn J Ophthalmol. 2012; 56:544–50.

19. Lee JH, Kim MJ, Ha SW, Kim HK. Autologous platelet-rich abdominal eye drops in the treatment of recurrent corneal erosions. Korean J Ophthalmol. 2016; 30:101–7.
20. Nelson JD, Craig JP, Akpek EK, et al. TFOS DEWS II introduction. Ocul Surf. 2017; 15:269–75.
21. Tsubota K, Goto E, Shimmura S, Shimazaki J. Treatment of abdominal epithelial defect by autologous serum application. Ophthalmology. 1999; 106:1984–9.
22. Goto E, Shimmura S, Shimazaki J, Tsubota K. Treatment of abdominal limbic keratoconjunctivitis by application of autologous serum. Cornea. 2001; 20:807–10.
23. del Castillo JM, de la Casa JM, Sardiña RC, et al. Treatment of abdominal corneal erosions using autologous serum. Cornea. 2002; 21:781–3.
24. Tananuvat N, Daniell M, Suillivan LJ, et al. Controlled study of the use of autologous serum in dry eye patients. Cornea. 2001; 20:802–6.

25. Ogawa Y, Okamoto S, Mori T, et al. Autologous serum eyedrops for the treatment of severe dry eye in patients with chronic graft-versus-host disease. Bone Marrow Transplant. 2003; 31:579–83.
26. Tsubota K, Goto E, Fujita H, et al. Treatment of dry eye by abdominal serum application in Sjögren's syndrome. Br J Ophthalmol. 1999; 83:390–5.
27. Anitua E, Andia I, Ardanza B, et al. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb Haemost. 2004; 91:4–15.

29. Klinger MH, Jelkmann W. Role of blood platelets in infection and inflammation. J Interferon Cytokine Res. 2002; 22:913–22.
30. Ho PC, Davis WH, Elliott JH, Cohen S. Kinetics of corneal abdominal regeneration and epidermal growth factor. Invest Ophthalmol. 1974; 13:804–9.
31. Gönül B, Koz M, Ersöz G, Kaplan B. Effect of EGF on the corneal wound healing of alloxan diabetic mice. Exp Eye Res. 1992; 54:519–24.

32. Rodeck U, Jost M, Kari C, et al. EGF-R dependent regulation of keratinocyte survival. J Cell Sci. 1997; 110(Pt 2):113–32.

33. Yashino Y, Garg R, Monroy D, et al. Production and secretion of transforming growth factor beta (TGF-β) by the human lacrimal gland. Curr Eye Res. 1996; 15:615–24.
34. Sammartino G, Tia M, Marenzi G, et al. Use of autologous plate-let-rich plasma (PRP) in periodontal defect treatment after abdominal of impacted mandibular third molars. J Oral Maxillofac Surg. 2006; 63:766–70.
35. Choi BH, Im CJ, Huh JY, et al. Effect of platelet-rich plasma on bone regeneration in autogenous bone graft. Int J Oral Maxillofac Surg. 2004; 33:56–9.

36. Englert SJ, Estep TH, Ellis-Stoll CC. Autologous platelet gel abdominal during cardiovascular surgery: effect on wound healing. J Extra Corpor Technol. 2005; 37:148–52.
37. Man D, Plosker H, Winland-Brown JE. The use of autologous pla-telet-rich plasma (platelet gel) and autologous platelet-poor plasma (fibrin glue) in cosmetic surgery. Plast Reconstr Surg. 2001; 107:229–37. discussion 238–9.

38. Bhanot S, Alex JC. Current applications of platelet gels in facial plastic surgery. Facial Plast Surg. 2002; 18:27–33.

39. Li DQ, Luo L, Chen Z, et al. JNK and ERK MAP kinases meidate induction of IL-1β, TNF-α and IL-8 following hyperosmolar stress in human limbal epithelial cells. Exp Eye Res. 2006; 82:588–96.
40. Li DQ, Chen X, Song XJ, et al. Stimulation of matrix metal-lopreteinases by hyperosmolarity via a JNK pathwasy in human corneal epithelial cells. Invest Ophthalmol Vis Sci. 2004; 45:4302–11.
41. Zeigler ME, Chi Y, Schmidt T, Varani J. Role of ERK and JNK pathways in regulating cell motility and matrix metalloproteinase 9 production in growth factor-stimulated human epidermal keratinocytes. J. Cell Physiol. 1999; 180:271–84.
42. Cho A, Graves J, Reidy MA. Mitogen-activated protein kinases mediate matrix metalloproteinase-9 expression in vascular smooth muscle cells. Arterioscler Thromb Vasc Biol. 2000; 20:2527–32.

43. Hayashida T, Decaestecker M, Schnaper HW. Cross-talk between ERK MAP kinase and Smad signaling pathways enhances TGF-beta dependent responses in human mesangial cells. FASEB J. 2003; 17:1576–8.
44. Gospodarowicz D, Bialecki H, Thakral TK. The angiogenic abdominal of the fibroblast and epidermal growth factor. Exp Eye Res. 1979; 28:501–14.
Figure 1.
The relative gene expression of inflammatory markers: IL-1, MMP-1, −3, TNF-α in three different groups of human limbal epithelial cell. The second group with adding 10% PRP was showed significant increase of inflammatory markers IL-1 and TNF-α. However, in the third group under hyperosmolar condition with 10% PRP, the gene expression of inflammatory marker was decreased into below or normal level. IL-1 = interleukin-1; MMP = matrix metalloproteinase; TNF-α= tumor necrosis factor-α; PRP = platelet rich plasma. * p < 0.05, Mann-Whitney U test.
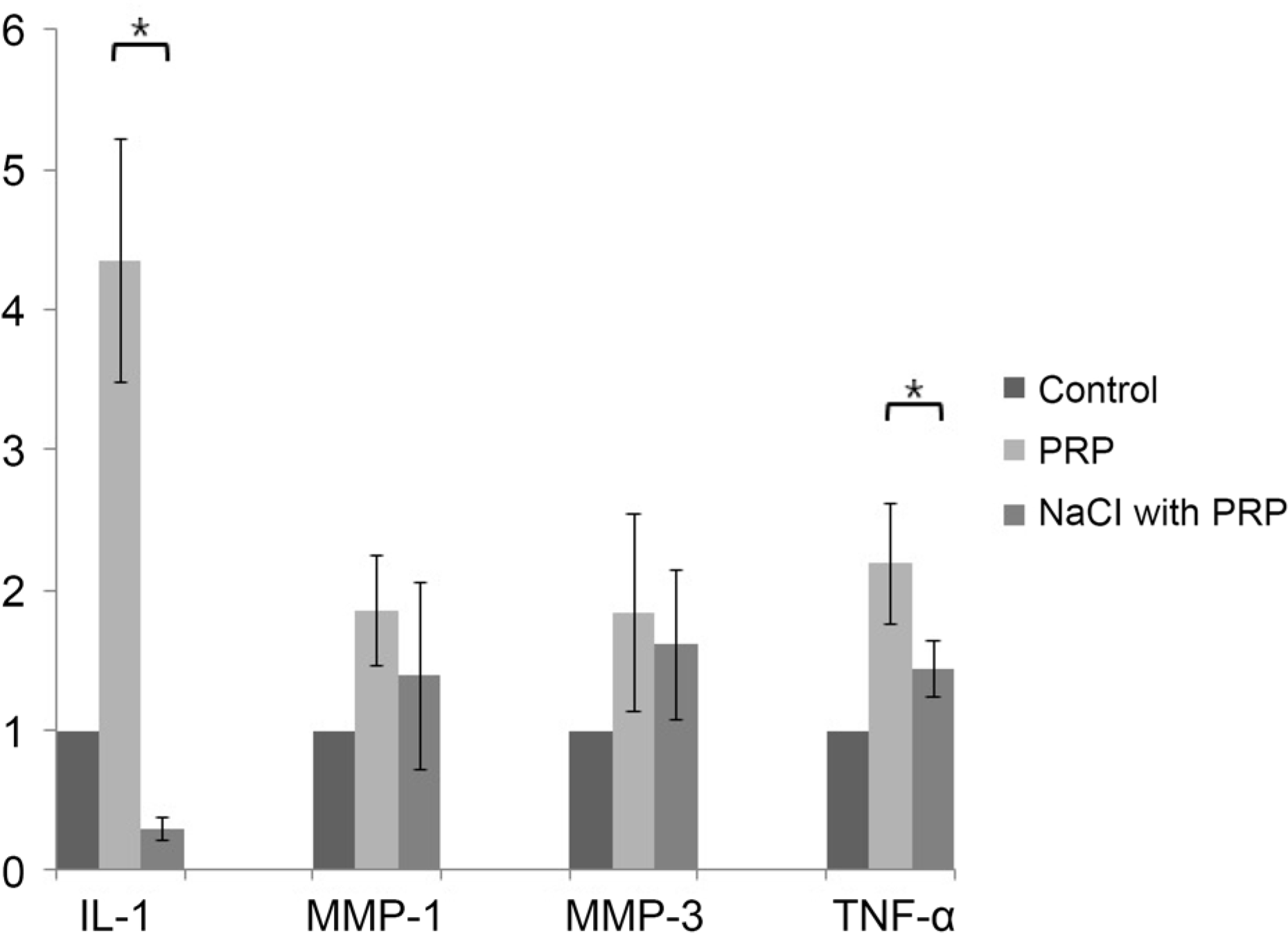
Table 1.
Demographic and clinical data of patients
Table 2.
Changes of tear film, cornea and conjunctival fluorescein stain, and symptom after autologous platelet-rich plasma therapy in all patients with dry eye syndrome
| Variable | Before treatment | 1 month after treatment | p-value* | 3 months after treatment | p-value* | 6 months after treatment | p-value* |
|---|---|---|---|---|---|---|---|
| BCVA | 0.72 ± 0.27 | 0.72 ± 0.29 | 0.898 | 0.74 ± 0.28 | 0.299 | 0.73 ± 0.27 | 0.678 |
| IOP (mmHg) | 14.83 ± 3.84 | 14.73 ± 3.46 | 0.777 | 14.55 ± 3.82 | 0.486 | 14.40 ± 4.01 | 0.302 |
| TBUT (seconds) | 3.45 ± 1.89 | 5.03 ± 1.90 | <0.001 | 6.03 ± 1.94 | <0.001 | 6.25 ± 1.97 | <0.001 |
| OSS | 4.55 ± 3.12 | 2.63 ± 2.30 | <0.001 | 2.43 ± 2.82 | <0.001 | 2.28 ± 2.31 | <0.001 |
| OSDI | 52.13 ± 25.84 | 41.21 ± 23.39 | <0.001 | 40.11 ± 23.01 | 0.008 | 40.58 ± 26.88 | 0.040 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download