Abstract
Purpose
Methods
Results
Figures and Tables
 | Figure 1Classification of matrix metalloproteinase-9 (MMP-9) test results. Distinct blue line in the result zone guarantees both the accuracy of test procedures and the validity of test results. Absence of red line is defined as ‘Negative (N)’ and marked red line parallel to blue line is defined as ‘Strong positive (S)’, with in-between subdivisions proportionally to the clarity of the red line. The red line gradually becomes more prominent from the left to the right configurations. |
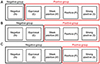 | Figure 2Categorization of the patients based on the results of matrix metalloproteinase-9 (MMP-9). (A) Result of MMP-9 negative was identified as negative group and the result of MMP-9 equivocal, weak positive, positive, and strong positive were categorized as positive group. (B) Result of MMP-9 negative and equivocal were categorized as negative group and the result of MMP-9 weak positive, positive, and strong positive were categorized as positive group. (C) Result of MMP-9 negative, equivocal, and weak positive were categorized as negative group while the other results of MMP-9 were categorized as positive group. |
 | Figure 3The difference of ocular surface disease index (OSDI) between the groups. (A) OSDI scores when result of matrix metalloproteinase-9 (MMP-9) negative was identified as negative. Note the significant difference of the average of OSDI scores between the negative (N) and positive (EWPS) (*p < 0.05). No significant difference of OSDI score was found when result of MMP-9 negative and equivocal were assumed as negative (B) nor result of MMP-9 negative, equivocal, and weak positive were assumed as negative (C). |
Table 2
Comparison of the demographics and test results between MMP-9 negative group and MMP-9 positive group when assuming result of MMP-9 negative as negative group

Values are presented as mean ± standard deviation unless otherwise indicated.
MMP = matrix metalloproteinase; BCVA = best corrected visual acuity; logMAR = logarithm of minimal angle of resolution; TBUT = tear breakup time; OSS = Oxford stain score; OSDI = Ocular surface disease index.
*The comparison was performed by using Student t-test; †the comparison was performed by using Pearson's chi-squared test; ‡the comparison was performed by division based on the subjects, not by eyes. If MMP-9 results in both eyes are not identical, more severe MMP-9 results are selected for the analysis. Eight patients are included in the negative group (N) and 59 patients are included in the positive group (EWPS).
Table 3
Comparison of the demographics and test results between MMP-9 negative group and MMP-9 positive group when assuming result of MMP-9 negative and equivocal as negative group

Values are presented as mean ± standard deviation unless otherwise indicated.
MMP = matrix metalloproteinase; BCVA = best corrected visual acuity; logMAR = logarithm of minimal angle of resolution; TBUT = tear breakup time; OSS = Oxford stain score; OSDI = Ocular surface disease index.
*The comparison was performed by using Student t-test; †the comparison was performed by using Pearson's chi-squared test; ‡the comparison was performed by division based on the subjects, not by eyes. If MMP-9 results in both eyes are not identical, more severe MMP-9 results are selected for the analysis. Twenty six patients are included in the negative group (NE) and 41 patients are included in the positive group (WPS).
Table 4
Comparison of the demographics and test results between MMP-9 negative group and MMP-9 positive group when assuming result of MMP-9 negative, equivocal and weak positive as negative group

Values are presented as mean ± standard deviation unless otherwise indicated.
MMP = matrix metalloproteinase; BCVA = best corrected visual acuity; logMAR = logarithm of minimal angle of resolution; TBUT = tear breakup time; OSS = Oxford stain score; OSDI = Ocular surface disease index.
*The comparison was performed by using student t-test; †the comparison was performed by using Pearson's chi-squared test; ‡the comparison was performed by division based on the subjects, not by eyes. If MMP-9 results in both eyes are not identical, more severe MMP-9 results are selected for the analysis. Thirty three patients are included in the negative group (NEW) and 34 patients are included in the positive group (PS).




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download