Abstract
Purpose
We determined the effects of silicone tube tip fixation on the nasal wall using an absorbable suture during silicone tube intubation in patients with congenital nasolacrimal duct obstruction.
Methods
Patients (55:71 eyes) diagnosed with congenital nasolacrimal duct obstruction and who underwent silicone tube intubation were divided into two groups: those in which the silicone tube was fixed to the nasal wall using an absorbable suture (fixed group) or not (non-fixed group). We investigated the percentage of silicone tube displacement, the time for displacement, the success of surgery, and the method of removing the silicone tube after surgery.
Results
The mean age, percentage of early displacement within 1 month, time to displacement, and success rate of surgery were 2.4 years, 0% (0/35), 1.4 months, and 100% (35/35) in the fixed group (35 patients) and 1.8 years, 44% (16/36), 0.8 months, and 97% (35/36) in the non-fixed group (36 patients), respectively. Both groups were able to remove the silicone tube simply through lacrimal punctum at the outpatient clinic.
Conclusions
In patients with congenital nasolacrimal duct obstruction, silicone tube fixation at the nasal wall after silicone tube intubation can prevent early displacement of silicone tubes within 1 month. This is a simple and effective technique because it re-moves the silicone tube through the lacrimal punctum without general anesthesia or intravenous anesthesia.
References
2. Sevel D. Development and congenital abnormalities of the abdominal apparatus. J Pediatr Ophthalmol Strabismus. 1981; 18:13–9.
4. Guerry D 3rd, Kendig EL Jr. Congenital impatency of the abdominal duct. Arch Ophthal. 1948; 39:193–204.
5. Lee SY, Chung HS, Kim HB, et al. The incidence of congenital abdominal duct obstruction in Korean neonates. J Korean Ophthalmol Soc. 1989; 30:5–8.
6. Paul TO. Medical management of congenital nasolacrimal duct obstruction. J Pediatr Ophthalmol Strabismus. 1985; 22:68–70.

7. Petersen RA, Robb RM. The natural course of congenital abdominal of the nasolacrimal duct. J Pediatr Ophthalmol Strabismus. 1978; 15:246–50.
8. Dortzbach RK, France TD, Kushner BJ, Gonnering RS. Silicone intubation for obstruction of the nasolacrimal duct in children. Am J Ophthalmol. 1982; 94:585–90.

9. Leone CR Jr, Van Gemert JV. The success rate of silicone abdominal in congenital lacrimal obstruction. Ophthalmic Surg. 1990; 21:90–2.
10. Kraft SP, Crawford JS. Silicone tube intubation in disorders of the lacrimal system in children. Am J Ophthalmol. 1982; 94:290–9.

11. Beigi B, O'Keefe M. Results of Crawford tube intubation in children. Acta Ophthalmol (Copenh). 1993; 71:405–7.

12. Veloudios A, Harvey JT, Philippon M. abdominal placement of abdominal nasolacrimal tubes. Ophthalmic Surg. 1991; 22:225–7.
13. Kim KS, Park TK, Choi WC. Intranasal endoscopic diagnosis and treatment in congenital nasolacrimal duct obstruction. J Korean Ophthalmol Soc. 2001; 42:7–12.
14. Anderson RL, Edwards JJ. Indications, complications, and results with silicone stents. Ophthalmology. 1979; 86:1474–87.

15. Lekskul A, Khamapirad B, Nimvorapun T. Simple technique for silicone intubation in congenital nasolacrimal duct obstruction. J Med Assoc Thai. 2004; 87:1082–6.
16. Lee TS, Jang M. A modified technique of bicanalicular silicone tube intubation in congenital nasolacrimal duct obstruction. J Korean Ophthalmol Soc. 2009; 50:984–8.

17. Ratliff CD, Meyer DR. Silicone intubation without intranasal abdominal for treatment of congenital nasolacrimal duct obstruction. Am J Ophthalmol. 1994; 118:781–5.
18. Migliori ME, Putterman AM. Silicone intubation for the treatment of congenital lacrimal duct obstruction: successful results abdominal the tubes after six weeks. Ophthalmology. 1988; 95:792–5.
19. Patrinely JR, Anderson RL. A review of lacrimal drainage surgery. Ophthalmic Plast Reconstr Surg. 1986; 2:97–102.

20. Park J, Lee YJ, Kim SJ, Jang JW. Factors affecting the outcome of silicone intubation for congenital nasolacrimal duct obstruction. J Korean Ophthalmol Soc. 2011; 52:266–71.

21. Kim J, Kang SM. The clinical outcomes of early silicone tube abdominal in congenital nasolacrimal duct obstruction. J Korean Ophthalmol Soc. 2018; 59:393–6.
22. Peterson NJ, Weaver RG, Yeatts RP. Effect of short-duration abdominal intubation in congenital nasolacrimal duct obstruction. Ophthal Plast Reconstr Surg. 2008; 24:167–71.
23. Sabermoghaddam AA, Hosseinpoor SS. Preventing silicone tube extrusion after nasolacrimal duct intubation in children. J Ophthalmic Vis Res. 2010; 5:280–3.
Figure 1.
Post-operative photograph of right nose in 8 years old patient. Silicone tube (red arrow) is loosely fixed on mucocuta-neous junction of nasal lateral wall by absorbable suture (yellow arrow).
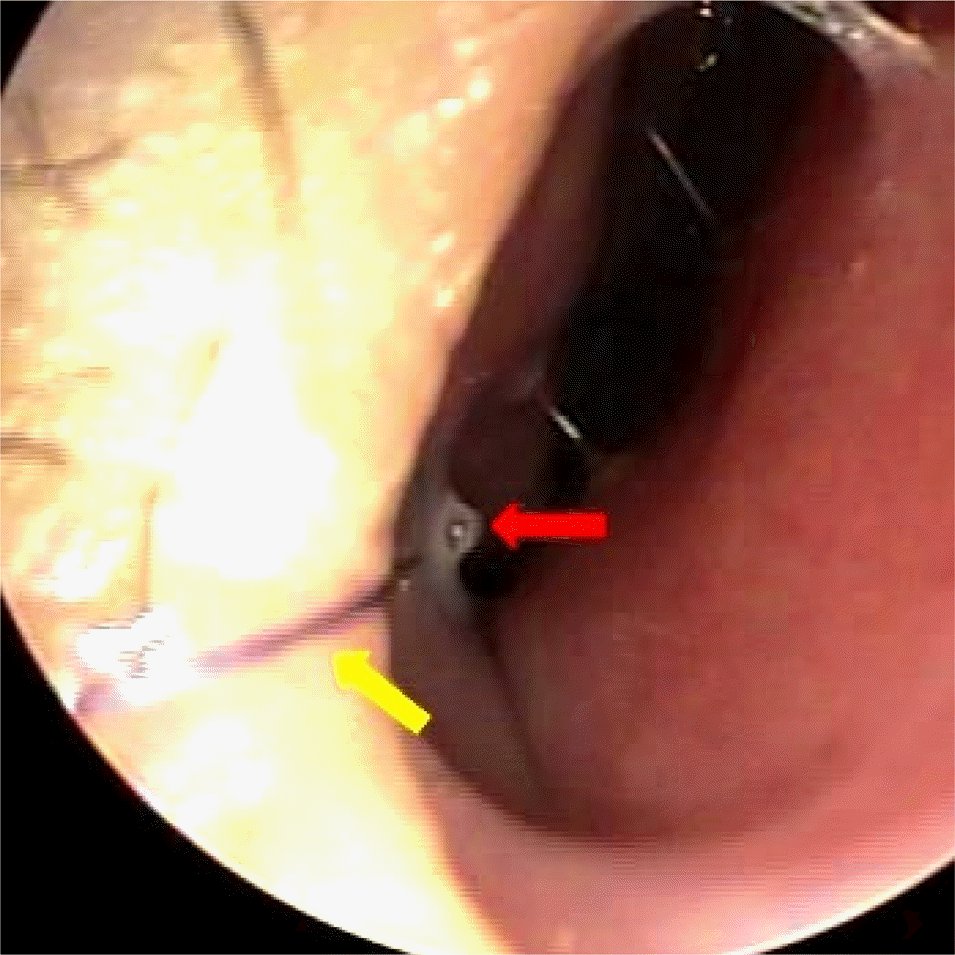
Table 1.
Patient characteristics
| Characteristic | Non-fixed group (eye = 36) | Nasal-fixed group (eye = 35) | p-value |
|---|---|---|---|
| Sex | |||
| Male | 20 | 19 | 0.914* |
| Female | 16 | 16 | |
| Age (years) | 1.8 ± 1.53 | 2.4 ± 1.59 | 0.004† |
| Side | |||
| Right | 20 | 18 | 0.727* |
| Left | 16 | 17 |
Table 2.
Tube retention time and success rate
| Variable | Non-fixed group (eye = 36) | Nasal-fixed group (eye = 35) | p-value |
|---|---|---|---|
| Tube retention time (months | |||
| Normal removal | 3.0 ± 0.71 (15) | 2.2 ± 0.35 (16) | 0.001* |
| Early displacement | 0.8 ± 0.75 (21) | 1.4 ± 0.37 (19) | 0.001* |
| 0–4 weeks | 13 | 0 | 0.000† |
| 4–16 weeks | 8 | 19 | –† |
| Total | 21 | 19 | 0.731† |
| Success rate (%) | 97 (35/36) | 100 (35/35) | 0.321† |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download