This article has been
cited by other articles in ScienceCentral.
Abstract
Solitary fibrous tumors (SFTs) are a unique group of soft tissue tumors originating from fibroblastic or myofibroblastic tissue. SFTs rarely occur in the liver, with less than 30 cases reported in the literature. We report a case of SFT of the liver presenting unique imaging features on CT and MRI. A 52-year-old woman presented with an incidentally detected hepatic mass that showed homogeneous strong enhancement, starting from the arterial phase until the delayed phase images on CT. On the T2-weighted MR image, the mass was heterogeneously hyperintense with multifocal hypointense dots and bands within the mass. The mass was microscopically characterized by bland-looking, ovoid to spindle shaped cells with stromal and perivascular hyalinization. Immunohistochemical studies showed reactivity for CD34.
초록
고립성 섬유종은 섬유모세포 혹은 근섬유모세포 조직으로부터 기원한 연조직 종양이다. 고립성 섬유종이 간에 발생하는 경우는 흔하지 않으며, 30예 이하의 증례가 보고된 바 있다. 본 연구에서는 컴퓨터단층촬영과 자기공명영상에서 독특한 소견을 보이는 간의 고립성 섬유종 증례를 보고하고자 한다. 52세 여자 환자가 우연히 발견된 간 종괴를 주소로 내원하였다. 종양은 컴퓨터단층촬영에서 동맥기부터 지연기까지 균질하고 강한 조영증강을 보였고, 자기공명영상에서는 T2 강조 영상에서 불균질한 높은 신호 강도를 보이며 내부 여러 군데에 신호강도가 낮은 점과 띠를 포함하고 있었다. 병리조직학적 검사에서 종양세포들은 타원형 혹은 방추형이었으며, 세포질 및 혈관주위 유리질화가 관찰되었다. 또한 면역조직화학 검사에서 CD34에 양성을 보였다.
Keywords: Solitary Fibrous Tumor, Liver, Abdomen
INTRODUCTION
Solitary fibrous tumor (SFT) is a mesenchymal tumor of fibroblastic or myofibroblastic cell origin in adults (
1). It was considered to occur primarily in the pleural cavity (
2). However, SFT may develop in virtually any site of the body, including the head and neck and the abdomen and pelvis (
12345). Although it seldom involves the liver, SFTs of the liver are usually seen as large well-enhancing tumors with variable signal intensities according to major histological components on MR imaging (
235). Given the rare incidence and non-specific clinical scenario, SFTs are not routinely included in the differential diagnoses in patients with hypervascular masses in the liver. In this report, we describe a case of a patient with SFT of the liver, which shows unique imaging features at CT and MRI.
CASE REPORT
A 52-year-old woman visited our hospital owing to a hepatic mass that was incidentally detected during a screening health checkup. She was a non-drinker and had no history of hepatitis or any previous abdominal surgery or trauma. On physical examination, the abdomen was soft and flat, without a palpable mass in the epigastric area. The initial routine laboratory test results, including those for full blood count and liver function test including alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, and total bilirubin, were within normal limits. Levels of tumor markers (alpha-fetoprotein and carcinoembryonic antigen) were also unremarkable.
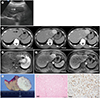
On ultrasonogram (
Fig. 1A), a relatively homogeneous hypoechoic mass was noted in the left lateral segment of the liver. CT images (
Fig. 1B) showed a well-marginated mass in the left hepatic lobe, which was vividly enhanced on the arterial and portal venous phase images. The mass also demonstrated persistent enhancement on the delayed phase CT image. MR imaging demonstrated that the signal intensity was homogeneously hypointense and heterogeneously hyperintense as compared to the hepatic parenchyma on the T1- and T2-weighted images, respectively. Further, the fat-saturated T2-weighted image depicted multifocal hypointense dots and bands within the mass (
Fig. 1C). Gadoxetic acid-enhanced MR imaging (
Fig. 1D) showed that the mass was homogeneously hyperintense and hypointense on the arterial phase (30 sec) and hepatobiliary phase (15 min) images, respectively. The patient underwent left lateral sectionectomy of the liver through the laparoscopic approach with a presumptive diagnosis of hepatic adenoma. Macroscopic analysis revealed a well-defined exophytic mass with a whitish cut surface (
Fig. 1E). The mass was microscopically characterized by bland-looking, ovoid to spindle shaped cells with stromal and perivascular hyalinization (
Fig. 1F). There was no mitotic activity. Immunohistochemical studies showed reactivity for CD34 (
Fig. 1F). However, the mass was negative for C-kit protein, S-100, desmin, and neurofilament. The pathological diagnosis was SFT of the liver.
DISCUSSION
SFTs are a unique group of soft tissue tumors originating from fibroblastic or myofibroblastic tissue (
1). Since its first description in 1931, SFTs have been reported to occur in various anatomical sites including thorax, head and neck, abdomino-pelvic cavity, and extremities (
367). SFTs may rarely occur in the liver, with less than 30 cases reported to date in the literature (
2). In the liver, the tumor often presents as a slow-growing, asymptomatic mass in middle-aged women (
2).
SFTs generally appear as well-marginated hypervascular tumors (
12345). Histologically, they may comprise a wide spectrum of microscopical features, from predominantly fibrous lesions to highly cellular tumors (
2). While the fibrous variant shows large collagenized zones, thick-walled hyalinized vessels, and predominant CD34 reactivity, the characteristics of the cellular variant include moderate to high cellular density, little fibrosis, thin-walled branching vessels, and weak CD34 expression (
8).
Regarding imaging findings, SFTs were reported to typically show strong enhancement owing to hypervascularity of the tumor regardless of the involved sites (
12). Further, given the presence of abundant fibrotic tissues within the tumors, early arterial enhancement may persist into the portal venous and delayed phases (
25). The tumor may sometimes also show central necrosis (
1). While SFTs in the pelvis tend to have a homogeneous enhancement pattern, the tumors occurring in the liver were reported to show heterogeneous enhancement, with serpentine feeding vessels in the periphery of the mass (
25). However, in our case, the tumor showed homogeneous strong enhancement starting from the arterial phase until the delayed phase. This might be related to high cellular density and hypervascularity of the mass, which was accompanied by scattered fibrotic tissues within the tumor. On MR imaging, T2-weighted images can show variable signal intensities based on the main components of the tumors (
2). Areas of low T2 signal intensity may imply the presence of collagenization and fibrosis. In contrast, high T2 signal intensity correlates with hypercellularity, little collagenous matrix, and myxoid change. In our case, the tumor showing strong enhancement starting from the arterial phase is believed to be related to high cellular component and hypervascularity of the tumor. And due to some fibrous portion within the tumor, the tumor maintained persistent and homogeneous enhancement on delayed phase.
SFT of the liver should be differentiated from other hypervascular hepatic tumors including adenoma, focal nodular hyperplasia, hemangioma, and hepatocellular carcinoma.
Histologically, most SFTs are benign. However, malignant features are found in up to 20% of cases (
2). Thus, the mass should be managed with complete surgical resection and careful long-term follow-up (
12). Further, given the hypervascular nature of the mass, embolization of feeding vessels of SFTs before surgical resection or biopsy would be beneficial (
1).
In summary, we present a case of SFT of the liver in which the mass showed homogeneous strong arterial enhancement that persisted on portal and delayed phase images.
Acknowledgments
This work was supported by the funds from the National Research Foundation of Korea (2018R1D-1A3B07043473) and the Chonnam National University Hospital Research Institute of Clinical Medicine in South Korea (CRI18091-2).
References
1. Fernandez A, Conrad M, Gill RM, Choi WT, Kumar V, Behr S. Solitary fibrous tumor in the abdomen and pelvis: a case series with radiological findings and treatment recommendations. Clin Imaging. 2018; 48:48–54.
2. Shanbhogue AK, Prasad SR, Takahashi N, Vikram R, Zaheer A, Sandrasegaran K. Somatic and visceral solitary fibrous tumors in the abdomen and pelvis: cross-sectional imaging spectrum. Radiographics. 2011; 31:393–408.
3. Tian TT, Wu JT, Hu XH, Yang GM, Sun J, Chen WX, et al. Imaging findings of solitary fibrous tumor in the abdomen and pelvis. Abdom Imaging. 2014; 39:1323–1329.
4. Shinde RS, Gupta A, Goel M, Patkar S. Solitary fibrous tumor of the liver - an unusual entity: a case report and review of literature. Ann Hepatobiliary Pancreat Surg. 2018; 22:156–158.
5. Soussan M, Felden A, Cyrta J, Morère JF, Douard R, Wind P. Case 198: solitary fibrous tumor of the liver. Radiology. 2013; 269:304–308.
6. Hwang JD, Kim JW, Chang JC. Imaging findings of a solitary fibrous tumor in pancreas: a case report. J Korean Soc Radiol. 2014; 70:53–57.
7. Kim TK, Han HY, Kim JH. Dynamic contrast-enhanced CT findings of a extrapleural solitary fibrous tumor in the spleen: a case report and literature review. J Korean Soc Radiol. 2018; 78:404–407.
8. Gengler C, Guillou L. Solitary fibrous tumor and haemangiopericytoma: evolution of a concept. Histopathology. 2006; 48:63–74.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download