Abbreviations
2-DG
BMDM
ChIP
ECAR
GLUT1
IRAK1
IRF5
M1
M2
M-MDSC
MDSC
OCR
OVA
OXPHOS
Peli1-mKO
Pellino-1-mKO
PMN-MDSC
TAM
TME
Tyk2
Ub
WT
INTRODUCTION
MATERIALS AND METHODS
Mouse experiments
Reagent
Preparation and polarization of macrophages
Extracellular acidification rate (ECAR) and oxygen consumption rate (OCR) assays
Immunoprecipitation and immunoblotting
Chromatin immunoprecipitation (ChIP)
Quantitative RT-PCR
Flow cytometry
Statistical analysis
RESULTS
Pellino-1 negatively regulates IL-10-induced M2c polarization of macrophages
Figure 1
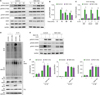
Pellino-1 regulates M1 and M2c, but not M2a and M2b macrophage polarization. Peritoneal macrophages from control or Peli1-mKO mice were polarized to M1 via treatment of LPS and IFN-γ (A), M2a via IL-4 and IL-13 treatment (B) and M2b via LPS and immune complexes (C) for indicated time. Expressions of each polarization markers were measured by quantitative RT-PCR. (D) Peritoneal macrophages from control or Peli1-mKO mice were treated with IL-10 for indicated time, and expressions of M2c-polarized macrophage-specific markers including Il10, Socs3, and Bcl3 were measured. (E) BMDMs from control or Peli1-mKO mice were prepared and treated with IL-10 for indicated time. IL-10-mediated gene expression of Il10, Socs3, and Bcl3 was evaluated by quantitative RT-PCR. All data were representative of 3 independent experiments and presented as means±SEMs (n=4).

Pellino-1-deficient macrophages exhibit a defect in mitochondria respiration, but enhancement of glycolysis during M2c polarization
Figure 2
Pellino-1-deficient macrophages show increase of glycolysis during M2c polarization. Control or Peli1-mKO BMDMs (1.5×105 cells/well) were treated with IL-10 for 6 h for M2c macrophage polarization. (A) OCR in control BMDMs (left panel) and WT versus Peli1-mKO BMDMs (right panel) in the presence or absence of IL-10. Dashed lines indicate treatment of oligomycin (left), carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone (FCCP, middle), and antimycin A (right). (B) Maximal respiratory capacity of BMDMs was measured by estimating AUC of OCR graphs and analyzed statistically. (C) ECAR in control BMDMs (left panel) and WT versus Peli1-mKO BMDMs (right panel) in the presence or absence of IL-10. Dashed lines indicate treatment of glucose (left), oligomycin (middle), and 2-DG (right). (D) Glycolysis of BMDMs was measured by estimating AUC of ECAR graphs and analyzed statistically. (E) ECAR (glycolysis) and OCR (maximal respiratory capacity) values were plotted. Arrows indicate metabolic changes from unstimulated BMDMs to M2c-polarized BMDMs. (F) To induce M2c polarization, BMDMs were treated with IL-10 for 6 h after pretreatment of 2-DG (1 mM) for 1 h. The expression of M2c polarization markers was measured. All data were representative of 2 independent experiments (A-E) and 3 independent experiments (F) and presented as means±SEMs (n=6 in A-E and n=4 in F).

Upon IL-10 stimulation, Pellino-1 promotes STAT1 phosphorylation via K63-linked ubiquitination of IRAK1 in macrophages
Figure 3
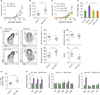
Pellino-1 regulates phosphorylation of STAT1 by K63-linked ubiquitination of IRAK1, during M2c macrophage polarization. (A) Control or Peli1-mKO BMDMs were treated with IL-10 for indicated time periods. Phosphorylation of STAT1 and STAT3 were measured by blotting. (B) Quantitative analysis of tyrosine phosphorylation of STAT1. (C) Chromatin immunoprecipitation assay was performed using control or Peli1-mKO BMDMs treated with IL-10 for 6 h. The promoter regions of M2c polarization markers including Il10, Socs3, and Bcl3 were estimated in immunoprecipitated STAT1. (D) Lysates from control or Peli1-mKO BMDMs treated with IL-10 for 10 min were immunoprecipitated using anti-IRAK1 Ab, and then blotted with Ab specific for Ub, K63-specific Ub, PELI1 and IRAK1. (E) Control or Peli1-mKO BMDMs were pre-treated with IRAK inhibitor for 1 h and followed by IL-10 treatment for 30 min. Blotting was performed to detect phosphorylation of STAT1 and STAT3. (F) Control or Peli1-mKO BMDMs were pre-treated with IRAK inhibitor for 1 h, followed by IL-10 treatment for 6 h. The expression of M2c markers was measured by quantitative RT-PCR. All data were representative of 3 independent experiments and presented as means±SEMs (n=3 in B and C, n=4 in F).
Pellino-1-mKO mice increase tumor grwoth via regulating M2c polarization in tumor microenvironment compared with WT mice
Figure 4
Peli1-mKO mice show enhancement of tumor growth. (A-B) Control or Peli1-mKO mice were subcutaneously injected with B16F10 cells and tumor growth was measured every 2–3 days (A). Sixteen days after tumor inoculation, tumor was excised and weighed (B). Control of Peli1-mKO mice were injected with either isotype control IgG or anti-CSF1 Ab. One day after Ab injection, B16F10 melanoma cells were injected subcutaneously, and tumor size (C) and tumor weight were measured (D). (E-H) Tumor-associated macrophages (TAM, CD45+ CD11b+ F4/80+), M1 TAM (CD45+ CD11b+ F4/80+ MHC-IIhigh CD206low), M2 TAM (CD45+ CD11b+ F4/80+ MHC-IIlow CD206high) populations in tumors from control or Peli1-mKO mice were analyzed by flow cytometry (E) and statistically analyzed (F). (G) IL-10-producing TAMs were analyzed by intracellular staining of IL-10. (H) M1 markers (middle), and M2a markers (right) were measured in sorted TAM by quantitative RT-PCR. All data were representative of 3 independent experiments and presented as means±SEMs (n=8).




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download