Abstract
Although many imaging modalities can play some roles in the diagnosis of vertebral artery dissection (VAD), digital subtraction angiography (DSA) remains the gold standard method, with the highest detection rate and ability to assist in planning for endovascular treatment. However, this tool is often avoided in children because its invasive nature and it exposes them to radiation. High resolution magnetic resonance imaging (HR-MRI) have been suggested to be a reliable and non-invasive alternative, but it has never been discussed in children in whom vertebral artery dissection is a rare condition. In this report, we evaluate a case of a 2-year-old child who initially presented with cerebellar symptoms, and was early diagnosed with vertebral artery dissection using HR-MRI and was successfully treated.
Vertebral artery dissection (VAD) is a rare cause of posterior circulation stroke in children. VAD in children was first reported in 1970 by Ouvrier and Hopkins and the number of cases has risen ever since, though the incidence of VAD in this age group is still unreported (12). The predisposing factors for VAD often include trauma, cervical anomaly, or systemic connective tissue diseases such as fibromuscular dysplasia. Spontaneous VAD in children is also, and it accounts for about 26% of the cause (1). VAD presents diverse symptoms including visual disturbances, hemiparesis, ataxia, headache, nausea and vomiting. Non-specific symptoms make early diagnosis difficult, causing an inappropriate delay in treatment.
Conventionally, catheter angiography is considered the gold standard in the diagnosis of vertebral artery dissection. Severe stenotic changes with slow flow, vessel wall irregularity, and the presence of an intimal flap are known as the angiographic findings of VAD, intimal flap being the most definitive sign (3). But catheter angiography does not always ensure definite diagnosis of dissection, due to its inability to evaluate the arterial wall (4). Furthermore, its invasiveness and the inevitable exposure to radiation also makes its application to children questionable. With the advances of non-invasive neuroimaging techniques, such as magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA), a recent trend of non-invasive evaluation is noted in the adult population. Moreover, high-resolution MR imaging (HR-MRI) provides an excellent efficacy in showing definitive features of intracranial vertebral dissection such as dissection flap and mural hematoma (4). However, the utilization of HR-MRI in VAD of children, has not yet been discussed.
In this case report, we report on the case of a child who initially presented cerebellar symptoms and was successfully diagnosed as vertebral artery dissection using HR-MRI.
A healthy 2-year-old boy was admitted to the emergency department with a sudden onset of irritability, recurrent vomiting and unwillingness to walk. He had no history of systemic illness or cervical trauma. The family history was unremarkable. The patient was fully conscious, and the vital signs were normal. On neurologic examination, his muscle tone and senses were normal, but he could not sit nor stand independently due to truncal ataxia. The laboratory findings including inflammatory markers and coagulation profile, were within the normal range.
Computed tomography (CT) scan was initially performed for brain evaluation. The focal low density was observed at cerebellar vermis on pre-contrast CT scan. The brain MRI with MRA was additionally acquired for further evaluation. On diffusion-weighted image (DWI), bright signal foci were noted at cerebellar vermis and bilateral posterior inferior cerebellar artery (PICA) territories including the cerebellar tonsil (Fig. 1). These lesions showed diffusion restriction on ADC map and high signal intensity on T2 weighted image. These findings were compatible with an acute infarction. There was no evidence of hemorrhage at the brain parenchyma or subarachnoid space. On MR angiography, the left vertebral artery flow was not seen and sharp distal edge was observed (Fig. 1). This finding strongly suggested the possibility of vertebral artery dissection. Afterwards, we performed a HR-MRI, instead of the conventional catheter angiography, to confirm the vertebral artery dissection, considering the patient's young age. HR-MRI was performed 5 days later after the onset of the symptoms with the following parameters: 1) PDWI (repetition time/echo time, 2000/31 ms; field of view, 10 cm; matrix size, 200 × 200; slice thickness, 2 mm without inter-slice gap; 22 slices; number of excitations, 2); 2) T2WI (2000/100 ms; 10 cm; 200 × 200; 2 mm; 22 slices; 4); and 3) T1WI (1000/7.9 ms; 10 cm; 200 × 200; 2 mm; 22 slices; 2), 4) Time of flight (TOF) imaging (25/3.45 ms; flip angle, 20°; 20 × 25 cm; 880 × 267; 1.2 mm; 160 slices). Contrast enhanced (CE)-T1WI was obtained using 0.1 mmol/kg gadobutrol (Gadovist; Bayer Pharma AG, Berlin, Germany) and T1-weighted parameters as described above. The black-blood technique with pre-regional saturation pulses of 80 mm thickness to saturate incoming arterial flow was used for all MRI scans. The total image acquisition times for HR-MRI and TOF MRA were approximately 35–40 min. The HR-MRI revealed the dilatation of the outer wall of the occluded left vertebral artery segment seen on MRA. We could also observe the dissection flap on T2WI as well as mural hematoma on T1WI of HR-MRI (Fig. 2, Supplementary Figs. 1, 2). The unruptured left VAD was diagnosed on the basis of HR-MR images and anticoagulation therapy was started as soon as the diagnosis was confirmed. After 3 days of treatment, the symptom of irritability was relieved and a week later, the patient was able to walk independently again. Two weeks later, the patient was discharged from the hospital without any other neurologic symptoms or signs.
The follow up HR-MRI and MRA were performed after three months. The vascular contour and the flow of previously dissected left vertebral artery were found to be normalized on follow-up images. The dissection flap and hematoma were no longer visible. The left PICA that was previously unseen, was also visualized on follow-up MRA and HR-MRI, revealing that it was the dominant supplier of bilateral cerebellum. This could be the reason why the initial infarction was seen in both sides of the cerebellum (Fig. 3).
Our report diagnosed spontaneous VAD causing ischemic stroke and presenting cerebellar symptoms in a 2-year-old boy with HR-MRI. Although VAD is a rare condition with nonspecific symptoms, it should be suspected in children with acute cerebellar symptoms. However, its diagnosis has been limited in many ways. Conventional catheter angiography, due to its radiation exposure and invasive nature, has setbacks in its utility for children. Kaufmann et al. reported 2.63% of neurologic complications and 0.14% of stroke with permanent disability in diagnostic cerebral angiography (5). But no other modality has shown a corresponding efficacy in the diagnosis until recently.
The HR-MRI presents a new possibility in the diagnosis of VAD. Han et al. reported that HR-MRI taken within 7 days after onset of symptoms showed excellent results in identifying definitive signs of VAD, such as dissection flap detected in 91.4% in CE-T1WI and 68.6% in T2WI. Mural hematoma was also detected in 54.3% of the cases in T1WI and CE-T1WI (4). The VAD is considered a dynamic pathology, thus the natural course with serial geometric changes can be reflected on HR-MRI findings, confusing the diagnosis in the later stages of the disease (67). A recent study showed significantly lower detection rates of key VAD findings including intimal flap, double lumen, aneurysmal dilatation, and intramural hematoma when comparing the chronic stage (> 60 days) to earlier stages (8). Additionally, intramural hematoma indicated the highest signal intensity in acute (0–3 days) to subacute stage (3–60 days) (7). Therefore, HR-MRI should ideally be performed at least before going to the chronic stage. In our case, we performed HR-MRI 5 days after the onset of the symptoms, and successfully detected the dissection flap in T2WI and mural hematoma in T1WI.
The HR-MRI can be a good follow-up modality as well. During the repairing process of the vessel wall, intramural hematoma is resorbed, replaced by the granulation tissue. The formation of the intima can reinforce the disruption defects of the internal elastic lamina, and eventually, it can be reflected on the HR-MRI as the disappearance of previous intimal flap, aneurysmal dilatation, and intramural hematoma (9). These major geometric healing changes are reported to take place between 2 weeks and 2 months in 63.6 to 83.9% of un-ruptured intracranial artery dissection patients (89). Therefore, 2 to 3 months after the onset can be suggested as an adequate follow-up interval. The vertebral artery dissection can progress to multiple delayed complications including pseudo aneurysm, thrombosis, and recurrent stroke (10). 81.5% of the recurrent stroke was reported to occur in first 22 days (8). Therefore, careful evaluation should be performed in the acute to subacute phase for complication signs. In our case, HR-MRI follow-up after 3 months revealed the disappearance of previous dissection flap and mural hematoma, with restoration of the left vertebral artery and PICA contour. Although further study is warranted, we can easily expect the role of HR-MRI in the confirmation of vessel healing and detection of delayed complications.
Some limitations of the conventional diagnostic modality still remain with HR-MRI in that sedation of the patient is in most cases inevitable with this age group. Further study regarding the usage of HR-MRI in the diagnosis of VAD in children should be conducted.
In summary, our case report introduces HR-MRI as a reliable and non-invasive diagnostic tool not only for confirming the diagnosis of vertebral artery dissection but also for follow up in children.
Figures and Tables
Fig. 1
A 2-year-old boy underwent CT and MRI due to sudden onset of irritability, vomiting and gait disturbance. Focal low density (arrows) is noted at cerebellar vermis on the initial CT scan (a) and diffusion restriction foci (dashed arrows) are observed at the corresponding cerebellar vermis and additionally at posterior inferior cerebellar artery (PICA) territory on DWI (b). On the MRA, the left vertebral artery flow is not seen and sharp distal edge is observed (c, arrowheads)
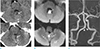
Fig. 2
High resolution MRI (HR-MRI) was performed 5 days later after the onset of the symptoms to confirm the vertebral artery dissection. The dilatation of the outer wall (dashed line) at occluded left vertebral artery segment on MRA is seen on T2WI (a) of HR-MRI. The dissection flap (arrows points to both ends of flap on each slice) is also evident on serial T2WIs (from left to right in figure show cranio-caudal slices). The mural hematoma (arrowheads) in early subacute stage is detectable as high signal intensity lesion on not only T1WI (b) but also contrast enhanced (CE)-T1WI (c) at medial side of double lumen. The dissection flap is also enhanced linearly (arrow) on CE-T1WI.

Fig. 3
Follow up HR-MRI was performed 3-months later. Normalized vascular flow (arrows) and contour (thick dashed lines) of left vertebral artery are seen on MRA (a), HR-T2WI (b) and T1WI (c). The dissection flap and hematoma are no longer visible. The left PICA artery flow (dashed arrows) and vascular contour (thin dashed line, thin dashed circle indicates the origin of left PICA from vertebral artery) are also newly observed on follow up images.

References
1. Hasan I, Wapnick S, Tenner MS, Couldwell W. Vertebral artery dissection in children: a comprehensive review. Pediatr Neurosurg. 2002; 37:168–177.

2. Cheon JE, Kim IO, Kim WS, Hwang YS, Wang KC, Yeon KM. MR diagnosis of cerebellar infarction due to vertebral artery dissection in children. Pediatr Radiol. 2001; 31:163–166.

3. Rafay MF, Armstrong D, Deveber G, Domi T, Chan A, MacGregor DL. Craniocervical arterial dissection in children: clinical and radiographic presentation and outcome. J Child Neurol. 2006; 21:8–16.

4. Han M, Rim NJ, Lee JS, Kim SY, Choi JW. Feasibility of high-resolution MR imaging for the diagnosis of intracranial vertebrobasilar artery dissection. Eur Radiol. 2014; 24:3017–3024.

5. Kaufmann TJ, Huston J 3rd, Mandrekar JN, Schleck CD, Thielen KR, Kallmes DF. Complications of diagnostic cerebral angiography: evaluation of 19,826 consecutive patients. Radiology. 2007; 243:812–819.
6. Choi YJ, Jung SC, Lee DH. Vessel wall imaging of the intracranial and cervical carotid arteries. J Stroke. 2015; 17:238–255.

7. Habs M, Pfefferkorn T, Cyran CC, et al. Age determination of vessel wall hematoma in spontaneous cervical artery dissection: a multi-sequence 3T cardiovascular magnetic resonance study. J Cardiovasc Magn Reson. 2011; 13:76.

8. Park KJ, Jung SC, Kim HS, et al. Multi-contrast high-resolution magnetic resonance findings of spontaneous and unruptured intracranial vertebral artery dissection: qualitative and quantitative analysis according to stages. Cerebrovasc Dis. 2016; 42:23–31.

Supplementary Materials
Supplementary Figure 1. Magnified HR-MRI of T2WI (a) and CE-T1WI (b) for dissecting left vertebral artery segment. The dissection flap (arrows points to both ends of flap on each slice) is seen on not only serial T2WIs but also CET1WI (from left to right in figure show cranio-caudal slices). Mural hematoma (arrowheads) in early subacute stage is detectable as high signal intensity lesion on CE-T1WI at medial side of double lumen. The lower line figures are added annotations to upper line figures for easily identifying the outer line of vessel walls (dashed lines), dissection flap (arrows) and hematomas (arrowheads).(https://doi.org/10.13104/imri.2019.23.3.259)
Supplementary Figure 2. Cine images comprised of HRT2WI. Dissection flaps crossing the lumen of left vertebral artery is seen as a high signal line.(https://doi.org/10.13104/imri.2019.23.3.259)
Supplementary Fig. 1
Magnified HR-MRI of T2WI (a) and CE-T1WI (b) for dissecting left vertebral artery segment. The dissection flap (arrows points to both ends of flap on each slice) is seen on not only serial T2WIs but also CET1WI (from left to right in figure show cranio-caudal slices). Mural hematoma (arrowheads) in early subacute stage is detectable as high signal intensity lesion on CE-T1WI at medial side of double lumen. The lower line figures are added annotations to upper line figures for easily identifying the outer line of vessel walls (dashed lines), dissection flap (arrows) and hematomas (arrowheads).




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download