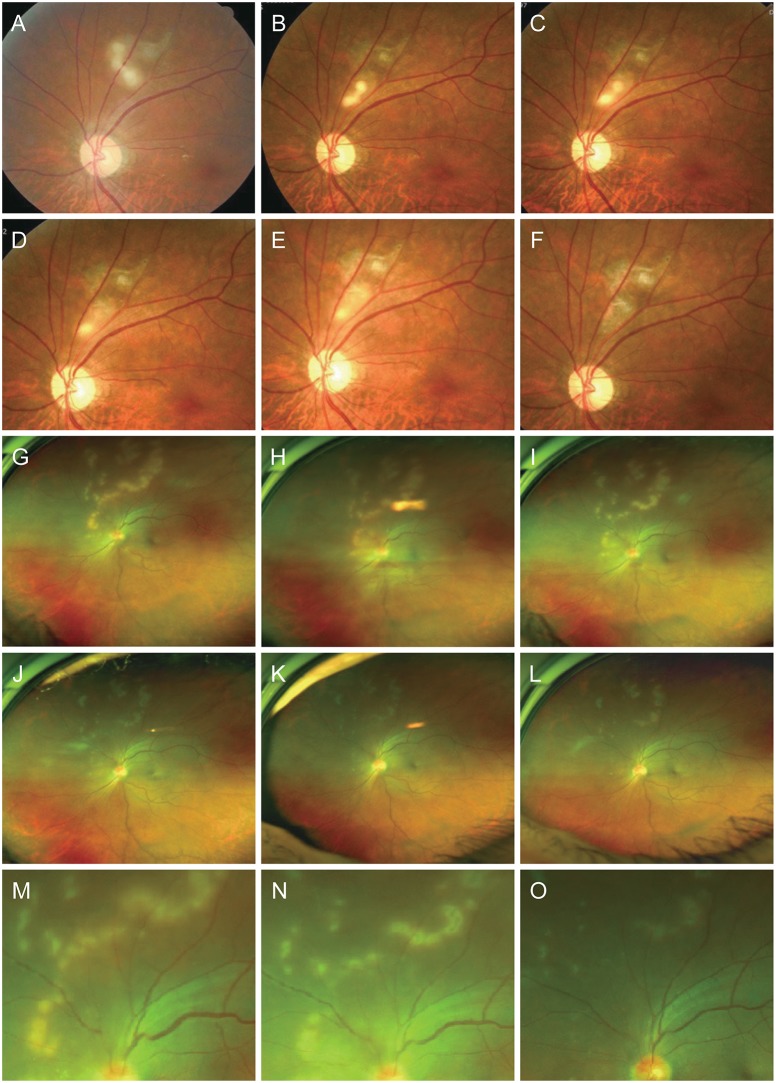
 | Fig. 1Initial and follow-up fundus photography. (A–F) Case 1. (A) Initial visit on February 22, 2012; follow-up visits on (B) May 30, 2012, (C) June 8, 2012, (D) June 22, 2012, (E) June 29, 2012, and (F) September 5, 2012. (G–O) Case 2. Initial visit on (G) December 12, 2016; follow-up visits on (H) December 19, 2016, (I) December 23, 2016, (J) December 30, 2016, (K) January 5, 2017, and (L) January 19, 2017; (M) enlarged image on December 12, 2016; (N) enlarged image on December 23, 2016; (O) enlarged image on January 19, 2017. |
Dear Editor,
Toxocara canis is a common helminth parasite of dogs that causes ocular toxocariasis (OT) in infected humans. OT occurs when Toxocara larvae migrate into the eye through blood circulation [1]. OT is clinically diagnosed based on its typical clinical ocular presentation, positive serologic test results, and exclusion of other possible causes of ocular granulomatous disease [2]. Thus, clinical ocular findings in OT are important factors in differential diagnosis. We report two cases of OT confirmed by serologic testing showing multiple retinal lesions that could have been overlooked on initial diagnosis.
Case 1. A 55-year-old man without relevant medical history presented to our hospital with a 7-day history of pain and blurry vision in the left eye. His best-corrected visual acuity (BCVA) was 20 / 25 in the affected eye. Funduscopic examination showed several focal yellowish-white retinal lesions comprising one quarter to one third of the optic disc diameter. The lesions' appearance was similar to a linear superimposed bead pattern or elongated, segmented shapes accompanied by a distal serpiginous retinal scar (Fig. 1A–1F).
Case 2. A previously healthy 59-year-old man presented with a 1-week history of blurred vision in his left eye. He had raised a dog indoors and had close contact with the animal. The BCVA of the affected eye was 20 / 25. On funduscopic examination, multiple yellowish-white retinal lesions comprising one quarter to one third of the optic disc diameter were distributed from the superior portion to the mid periphery of the optic disc. Some lesions were present independently as foci while others overlapped and appeared as elongated, segmented lesions that formed meandering lines (Fig. 1G–1O).
Neither of these cases had specific findings in the anterior segments except mild chamber reactions; vitreous cells were not apparent. No serological or radiologic evidence of toxoplasma, rubella, cytomegalovirus, herpes simplex virus, syphilis or rheumatologic disease was observed. The only major finding was a positive enzyme-linked immunosorbent assay result for Toxocara canis immunoglobulin G antibody. Enzyme-linked immunosorbent assay has sufficient specificity to be a reliable indirect test for diagnosis of this infection [13]. Considering serologic test results and by ruling out other potential retinal inflammatory diseases, these cases were diagnosed with OT. They were treated with albendazole (400 mg twice a day) for 10 days, and with methylprednisolone for 4 weeks (16 mg/day for 1 week, followed by 8 mg/day for 1 week, and finally 4 mg/day for 2 weeks) beginning after 1 week of albendazole treatment. Both patients responded to the combination treatment. The BCVA of the affected eye improved to 20 / 20 and retinal lesions gradually subsided in response to treatment.
In these two cases, retinal lesions did not appear as typical granulomas and the number and distribution patterns varied from typical OT, which occurs in one of three forms: as a peripheral granuloma, which is a focal, elevated, white nodule, with or without fibrocellular bands running from a peripheral inflammatory mass to the more posterior retina or the optic nerve; as a posterior pole granuloma, usually <1 disc diameter with pigmentation; or as chronic endophtalmitis [4]. Our cases had multiple foci or elongated, segmented retinal lesions. Case 1 showed continuous larva migration [2], a unique feature of OT compared with other inflammatory retinal diseases [5]. Meanwhile, case 2 had a greater number of widely spread retinal lesions, which were arranged linearly around the optic disc and became more amorphous toward the periphery. Active lesions subsided after a relatively short clinical course of 5 weeks without definite larva migration. The appearance was similar to the multifocal yellow lesions seen in the early stage of diffuse unilateral subacute neuroretinits. However, the disease course of case 2 was not recurrent, and neither evanescent episodes nor larva in the surrounding area were observed.
In summary, we experienced serologically diagnosed OT cases with multiple atypical retinal lesions. The patients responded to combination treatment with albendazole and methylprednisolone. In a situation where biopsy is practically impossible, a positive serologic result and response to treatment may be an important basis for diagnosis; clinical presentation can serve as a useful clue in such cases. We anticipate that this case report will provide insight into atypical fundus presentations of OT that could easily be overlooked in early differential diagnosis.
References
1. Despommier D. Toxocariasis: clinical aspects, epidemiology, medical ecology, and molecular aspects. Clin Microbiol Rev. 2003; 16:265–272. PMID: 12692098.

2. Ahn SJ, Woo SJ, Jin Y, et al. Clinical features and course of ocular toxocariasis in adults. PLoS Negl Trop Dis. 2014; 8:e2938. PMID: 24922534.

3. Bae KW, Ahn SJ, Park KH, Woo SJ. Diagnostic value of the serum anti-Toxocara IgG tilter for ocular toxocariasis in patients with uveitis at a tertiary hospital in Korea. Korean J Ophthalmol. 2016; 30:258–264. PMID: 27478352.
4. Woodhall D, Starr MC, Montgomery SP, et al. Ocular toxocariasis: epidemiologic, anatomic, and therapeutic variations based on a survey of ophthalmic subspecialists. Ophthalmology. 2012; 119:1211–1217. PMID: 22336630.

5. Ahn SJ, Ryoo NK, Woo SJ. Ocular toxocariasis: clinical features, diagnosis, treatment, and prevention. Asia Pac Allergy. 2014; 4:134–141. PMID: 25097848.

Go to : 




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download