INTRODUCTION
METHODS
Study design and animals
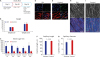 | Fig. 1Microvascular changes of the heart in diabetic pigs. (A) Study flow. (B) Weight of the pigs on day 0 and day 56. (C) The results of serum glucose tests of the pigs at certain time points. (D, E) Myocardial capillaries of the DM and control pigs. (F, G) Quantification of the capillaries between diabetic and control pigs. (H, I) Capillary endothelial junction (VE-cadherin) in the diabetic and control pigs.DM = diabetes mellitus, CAG = coronary angiography, OCT = optical coherence tomography, CCTA = coronary computed tomography angiography, CMR = cardiac magnet resonance, VE = vascular endothelial; DAPI = nucleus, α-SMA = alpha-smooth muscle actin, Lectin = BS-1 lectin, Merge = merged figure.
|
Coronary angiogram
OCT
CCTA
CMR imaging and perfusion
Immunofluorescence staining and confocal microscopic analysis
Immunofluorescence quantification of microvasculopathy in the myocardium
Patients
Statistical analysis
Ethics statement
RESULTS
Microvascular changes
Myocardial flow analysis
MR perfusion
 | Fig. 2Cardiac magnetic resonance perfusion in the diabetic pigs. (A) Representative figures measuring the myocardial perfusion in the 6 segments of the mid ventricle at rest and under adenosine stress. (B) Representative figures of the measured myocardial perfusion graphs. Red line = SI-time curve of the left ventricular cavity; cyan lines = SI-time curve of the 6 midventricular myocardial segments. (C) Mean values of the RRU, SRU, and MPRI of the 16 myocardial segments. (D) Estimated mean of RRU, SRU, and MPRI in diabetic and control pigs. P value by two-way analysis of variance. Error bars = standard deviation.BA = basal anterior, BAS = basal anteroseptal, BIS = basal inferoseptal, BI = basal inferior, BIL = basal inferolateral, BAL = basal anterolateral, MA = mid anterior, MAS = mid anteroseptal, MIS = mid inferoseptal, MI = mid inferior, MIL = mid inferolateral, MAL = mid anterolateral, AA = apical anterior, AS = apical septal, AI = apical inferior, AL = apical lateral, DM = diabetes mellitus, SI = signal intensity, RRU = relative upslope at rest, SRU = relative upslope at stress, MPRI = myocardial perfusion reserve index.
|
CT angiography
 | Fig. 3CT angiography. (A) Quantification of contrast-enhanced myocardial SI in the arterial phases of CT angiography. Regions of interest were drawn in the six segments of mid ventricle in the diabetic and control pigs. (B) Estimated mean of ratio of myocardial SI to aortic SI. P value by two-way analysis of variance.DM = diabetes mellitus, CT = computed tomography, SI = signal intensity.
|
Macrovascular changes
CAG and optical coherent tomography
 | Fig. 4Epicardial coronary artery. (A) Optical coherence tomography images of the coronary arteries of diabetic and control pigs at baseline, 4 weeks, and 8 weeks. (B) Vessel size, lumen diameter, and intima media thickness of the three segments of LAD at baseline, 4 weeks, and 8 weeks.DM = diabetes mellitus, LCX = left circumflex artery, LAD = left anterior descending artery.
|
Myocardial flow analysis in patients with MI
Table 1
Clinical characteristics and CMR data of patients with myocardial infarction

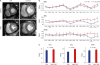 | Fig. 5Representative figures of cardiac MR perfusion of diabetic and control patients. (A) MR perfusion images under stress and at rest in diabetic and control patients (B) Mean values of the RRU, SRU, and MPRI of the 16 myocardial segments. (C) Estimated mean of RRU, SRU and MPRI in diabetic and control patients. P value by two-way analysis of variance. P value by Wilcoxon rank sum test. Error bars = standard deviation.MR = magnetic resonance, BA = basal anterior, BAS = basal anteroseptal, BIS = basal inferoseptal, BI = basal inferior, BIL = basal inferolateral, BAL = basal anterolateral, MA = mid anterior, MAS = mid anteroseptal, MIS = mid inferoseptal, MI = mid inferior, MIL = mid inferolateral, MAL = mid anterolateral, AA = apical anterior, AS = apical septal, AI = apical inferior, AL = apical lateral, RRU = relative upslope at rest, SRU = relative upslope at stress, MPRI = myocardial perfusion reserve index.
|
 | Fig. 6Blood glucose and HbA1c and clinical outcomes. (A) Diabetic patients have higher glucose and HbA1c levels. (B) There was no difference in MACE between patients with and without diabetes during a median follow-up of 3 years.DM = diabetes mellitus, HbA1c = hemoglobin A1c, MACE = major adverse cardiovascular outcome, MI = myocardial infarction.
|




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download