This article has been
cited by other articles in ScienceCentral.
Dear Editor:
Pseudomyogenic hemangioendothelioma, also known as epithelioid sarcoma-like hemangioendothelioma or fibroma-like variant of epithelioid sarcoma, is a rare soft tissue tumor with indolent course accompanied with frequent local recurrence and low metastatic risk. It has recently been characterized molecularly by t(7,19)(q22,q13) translocation resulting in
SERPINE1-FOSB fusion. It predominantly affects young to middle aged male around 20 to 50 years old with propensity to involve distal extremities especially lower limb in the same anatomic site as well as less frequently involves head and neck, chest, abdomen, and genital area. Clinically, Pseudomyogenic hemangioendothelioma can present as erythematous to skin colored papule, plaque and subcutaneous nodule with multifocal lesions and multiple tissue plane involvement, such as subcutaneous tissue, muscle, and bone
12345.
A 30-year-old male developed several painful erythematous papules with progression to erythematous indurated plaque with overlying crust and central necrosis around right forearm (
Fig. 1A) for 4 months. Multiple painful erythematous subcutaneous nodules over periungal area of right 3rd finger developed subsequently (
Fig. 1B). Pathological findings revealed ill-defined intradermal and subcutaneous tumor with plump spindled cell proliferation arranged in interlacing fascicles. These tumor cells showed vesicular nuclei, small nucleoli, and pale to eosinophilic cytoplasm in the background with brisk inflammatory cell infiltration. Inconspicuous pleomorphism and mitotic activity (<5/10 high power field) are found (
Fig. 2C). Immunohistochemistry staining demonstrates diffuse positive staining for AE1/AE3, CD31, friend leukemia integration 1 transcription factor (FLI-1), erythroblastosis virus E26 transforming sequence-related gene (ERG), integrase interactor 1 (INI-1) whereas negative staining for CD34 (
Fig. 2D~I). Computed tomography with contrast for right forearm demonstrates several enhanced nodules in right flexor digitorum superficialis muscle, flexor digitorum profundus muscle and in subcutaneous region of the ulnar site (
Fig. 1C). Thus far, he was diagnosed with pseudomyogenic hemangioendothelioma. Wide local excision with a margin of 2 cm and adjuvant chemotherapy with MAID (mesna, adriamycin, ifosfamide, dacarbazine) were arranged and no recurrence was found after one year follow-up. We received the pateint's consent form about publishing all photographic materials.
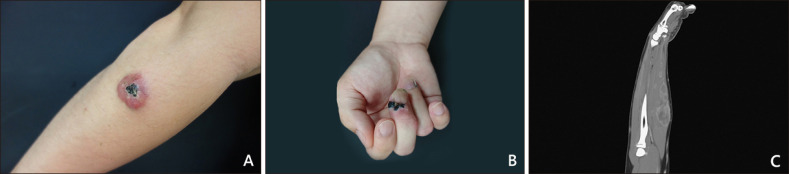 | Fig. 1(A) Solitary painful erythematous indurated plaque with overlying crust and ulcer on right radial side of forearm. (B) Multiple tender erythematous subcutaneous nodules with overlying crust over periungal region over 3rd finger. (C) Computed tomography of right forearm shows several enhanced nodule up to 3.4 cm in flexor digitorum superficialis muscle, flexor digitorum profundus muscle, flexor carpi ulnaris muscle, extensor carpi radialis brevis muscle and in subcutaneous region over ulnar side.
|
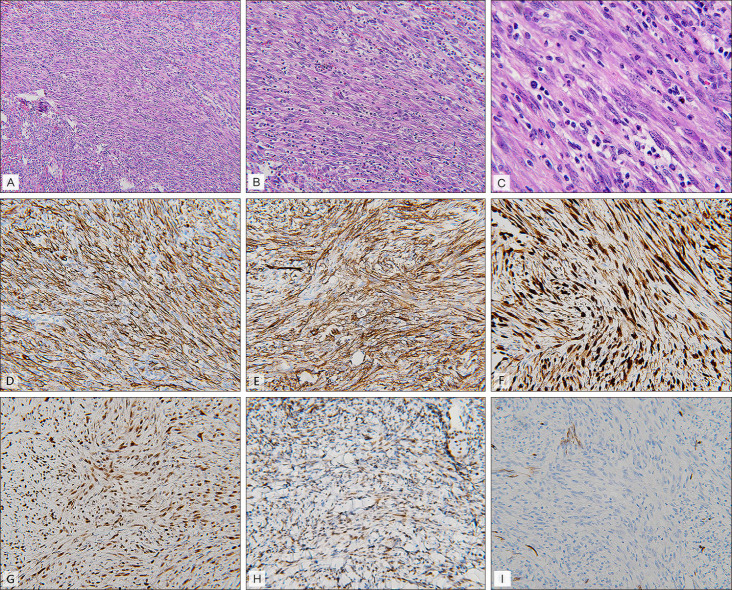 | Fig. 2(A) Infiltrative intradermal to subcutical tumor mass with proliferative spindle cells and inflammatory cell infiltration (haematoxylin and eosin [H&E], ×100). (B) Fasicles of interlacing spindle cells and brisk inflammatory cell scattered among the tumor cell. Red blood cell extravasation is also seen (H&E, ×200). (C) Plumped spindle cell with vesicular nucleus, prominent nucleoli and ample eosinophilic cytoplasm. Mitotic figure and pleomorphism are inconspicuous (H&E, ×400). (D) Immunohistochemistry staining showed diffuse positive staining for AE1/AE3 (×200), (E) erythroblastosis virus E26 transforming sequencerelated gene (×200), (F) CD31 (×200), (G) friend leukemia integration 1 transcription factor (×200), (H) integrase interactor 1 (×200), and (I) negative staining for CD34 (×200).
|
Immunohistochemical staining for pseudomyogenic hemangioendothelioma showed positive result for AE1/AE3, CD31, ERG, FLI-1, INI-1 and negative result for CD34 and epithelial membrane antigen. Pathological differential diagnosis includes epithelioid sarcoma which revealed naked granuloma with central necrosis and immunohistochemical staining showed negative result for CD31, INI-1, and ERG. Epithelioid hemangioendothelioma which showed vasculocentric cord of epithelioid cells in myxohyaline stroma and epithelioid angiosarcoma which showed sheets of plump spindle cells with marked atypia, which both of them show positive staining for CD34 and focally rather than diffusely staining for AE1/AE3 compared to pseudomyogenic hemangioendothelioma
5.
Based on currently available literature, the treatment options for pseudomyogenic hemangioendothelioma mainly focus on surgical excision, while on account of multifocal lesion with easily local recurrence and occasionally unresectable or high risk of fracture, systemic management with chemotherapy or target therapy has been raised in some case report
12345. Currently, there is no consensus about which chemotherapy regimen is more suitable for pseudomyogenic hemangioendothelioma. SERPINE1-FOSB fusion will lead to truncated FOSB protein formation, and it will bind to Jun family protein which results in activating protein-1 (AP-1) formation. AP-1 complex will promote downstream signal transduction and cause proliferation, differentiation and transformation of the tumor cells. As for target therapy, multiple tyrosine kinase inhibitor such as telatinib, mainly blocked the upstream surface receptor vascular endothelial growth factor/platelet-derived growth factor receptor, which further impeded SERPINE1-FOSB downstream pathway. It have been reported with promising prognosis when inadequate response with chemotherapy was found in some cases
34.
In conclusion, pseudomyogenic hemangioendothelioma is an easily neglected diagnosis which requires familarity with its pathological features and an aid in immuno histochemical staining. Appropriate treatment followed by regular follow-up for local recurrence and distant metastasis are necessary.






 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download