Abstract
Purpose
Early detection of Mycoplasma pneumoniae is important for appropriate antimicrobial therapy in children with pneumonia. This study aimed to evaluate the diagnostic value of a rapid antigen test kit in detecting M. pneumoniae from respiratory specimens in children with lower respiratory tract infection (LRTI).
Methods
A total of 215 nasopharyngeal aspirates (NPAs) were selected from a pool of NPAs that had been obtained from children admitted for LRTI from August 2010 to August 2018. The specimens had been tested for M. pneumoniae by culture and stored at −70°C until use. Tests with Ribotest Mycoplasma® were performed and interpreted independently by two investigators who were blinded to the culture results.
Results
Among the 215 NPAs, 119 were culture positive for M. pneumoniae and 96 were culture negative. Of the culture-positive specimens, 74 (62.2%) were positive for M. pneumoniae by Ribotest Mycoplasma®, and 92 of the 96 (95.8%) culture-negative specimens were negative for M. pneumoniae by Ribotest Mycoplasma®. When culture was used as the standard test, the sensitivity and specificity of Ribotest Mycoplasma® were 62.2% and 95.8%, respectively. Additionally, the positive predictive value, negative predictive value, and overall agreement rates with Ribotest Mycoplasma® were 94.9%, 67.2%, and 77.2%, respectively.
Go to : 
References
1. Principi N, Esposito S. Emerging role of Mycoplasma pneumoniae and Chlamydia pneumoniae in paediatric respiratory-tract infections. Lancet Infect Dis. 2001; 1:334–44.
2. Han MS, Yun KW, Lee HJ, Park JY, Rhie K, Lee JK, et al. Contribution of co-detected respiratory viruses and patient age to the clinical manifestations of Mycoplasma pneumoniae pneumonia in children. Pediatr Infect Dis J. 2018; 37:531–6.
3. Chalker V, Stocki T, Litt D, Bermingham A, Watson J, Fleming D, et al. Increased detection of Mycoplasma pneumoniae infection in children in England and Wales, October 2011 to January 2012. Euro Surveill. 2012; 17:20081. PUBMED.

4. Centers for Disease Control and Prevention. Outbreak of community-acquired pneumonia caused by Mycoplasma pneumoniae–Colorado, 2000. JAMA. 2001; 285:2073–4.
5. Eibach D, Casalegno JS, Escuret V, Billaud G, Mekki Y, Frobert E, et al. Increased detection of Mycoplasma pneumoniae infection in children, Lyon, France, 2010 to 2011. Euro Surveill. 2012; 17:20094. PUBMED.

6. Waites KB, Talkington DF. Mycoplasma pneumoniae and its role as a human pathogen. Clin Microbiol Rev. 2004; 17:697–728.
7. Okazaki N, Narita M, Yamada S, Izumikawa K, Umetsu M, Kenri T, et al. Characteristics of macrolide-resistant Mycoplasma pneumoniae strains isolated from patients and induced with erythromycin in vitro. Microbiol Immunol. 2001; 45:617–20.
8. Hong KB, Choi EH, Lee HJ, Lee SY, Cho EY, Choi JH, et al. Macrolide resistance of Mycoplasma pneumoniae, South Korea, 2000–2011. Emerg Infect Dis. 2013; 19:1281–4.
9. Cao B, Zhao CJ, Yin YD, Zhao F, Song SF, Bai L, et al. High prevalence of macrolide resistance in Mycoplasma pneumoniae isolates from adult and adolescent patients with respiratory tract infection in China. Clin Infect Dis. 2010; 51:189–94.
10. Kawai Y, Miyashita N, Kubo M, Akaike H, Kato A, Nishizawa Y, et al. Nationwide surveillance of macrolide-resistant Mycoplasma pneumoniae infection in pediatric patients. Antimicrob Agents Chemother. 2013; 57:4046–9.
11. Zhou Z, Li X, Chen X, Luo F, Pan C, Zheng X, et al. Macrolide-resistant Mycoplasma pneumoniae in adults in Zhejiang, China. Antimicrob Agents Chemother. 2015; 59:1048–51.
12. Meyer Sauteur PM, Unger WW, Nadal D, Berger C, Vink C, van Rossum AM. Infection with and carriage of Mycoplasma pneumoniae in children. Front Microbiol. 2016; 7:329.
13. Kohno S, Ishida T, Izumikawa K, Iwata S, Kadota J, Tanaka H, et al. Guiding principles for treating Mycoplasma pneumoniae pneumonia [Internet]. Tokyo: The Committee of Japanese Society of Mycoplasmology;2014. [cited 2019 Mar 10]. Available from:. http://square.umin.ac.jp/jsm/Eng. %20shisin.pdf.
14. Quanquin NM, Cherry JD. Mycoplasma and ureaplasma infection. Cherry JD, Harrison GJ, Kaplan SL, Steinbach WJ, Hotez PJ, editors. editors.Feigin and Cherry's textbook of pediatric infectious diseases. 8th Ed.Philadelphia: Elsevier, Inc.;2019. p. 1976–2003.
15. Miyashita N, Kawai Y, Tanaka T, Akaike H, Teranishi H, Wakabayashi T, et al. Diagnostic sensitivity of a rapid antigen test for the detection of Mycoplasma pneumoniae: comparison with real-time PCR. J Infect Chemother. 2015; 21:473–5.
16. She RC, Thurber A, Hymas WC, Stevenson J, Langer J, Litwin CM, et al. Limited utility of culture for Mycoplasma pneumoniae and Chlamydophila pneumoniae for diagnosis of respiratory tract infections. J Clin Microbiol. 2010; 48:3380–2.
17. Yamazaki T, Kuroki H, Itagaki T, Iwata S, Tateda K. Evaluation of a rapid antigen detection kit targeting L7/L12 ribosomal protein for Mycoplasma pneumoniae. Kansenshogaku Zasshi. 2015; 89:394–9.
18. Miyashita N, Kawai Y, Kato T, Tanaka T, Akaike H, Teranishi H, et al. Rapid diagnostic method for the identification of Mycoplasma pneumoniae respiratory tract infection. J Infect Chemother. 2016; 22:327–30.
19. Stewart EH, Davis B, Clemans-Taylor BL, Littenberg B, Estrada CA, Centor RM. Rapid antigen group A streptococcus test to diagnose pharyngitis: a systematic review and metaanalysis. PLoS One. 2014; 9:e111727.

20. Lee CS, Lee JH, Kim CH. Time-dependent sensitivity of a rapid antigen test in patients with 2009 H1N1 influenza. J Clin Microbiol. 2011; 49:1702.

21. Furr PM, Taylor-Robinson D. Long-term viability of stored mycoplasmas and ureaplasmas. J Med Microbiol. 1990; 31:203–6.

22. Atkinson TP, Balish MF, Waites KB. Epidemiology, clinical manifestations, pathogenesis and laboratory detection of Mycoplasma pneumoniae infections. FEMS Microbiol Rev. 2008; 32:956–73.
Go to : 
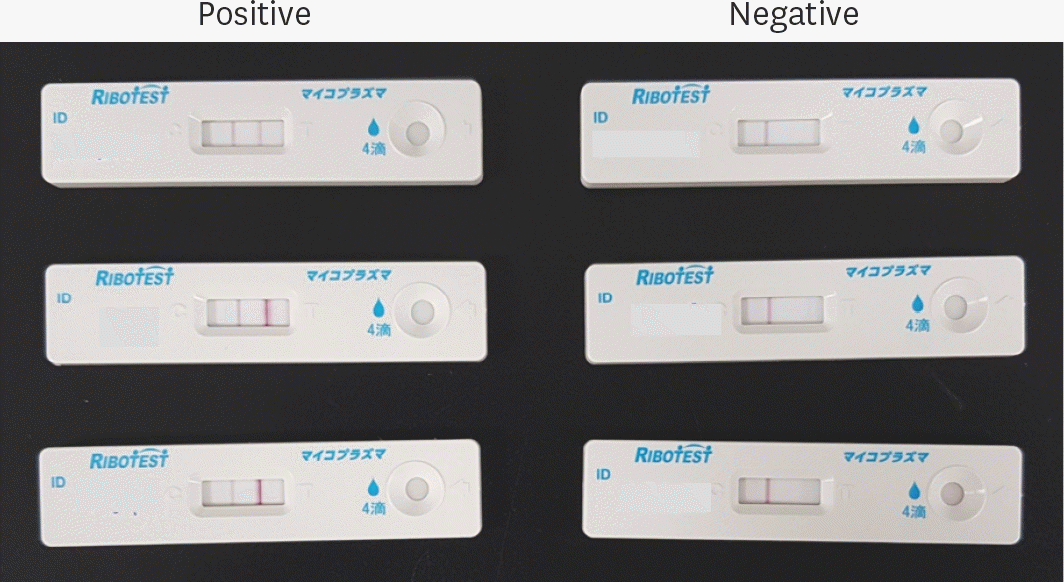 | Fig. 1.Pictures showing test plates that are positive and negative results of Ribotest Mycoplasma® test for detection of M. pneumoniae from nasal aspirates of children with low respiratory traction infections. |
Table 1.
Results of Ribotest Mycoplasma® test compared to cell culture method
| Results of culture test | No. (%) of samples with Ribotest® | ||
|---|---|---|---|
| Positive | Negative | Total | |
| Positive | 74 (94.9) | 45 (32.8) | 119 (55.3) |
| Negative | 4 (5.1) | 92 (67.2) | 96 (44.7) |
| Total | 78 (100) | 137 (100) | 215 (100) |
Table 2.
Diagnostic sensitivity, specificity, positive and negative predictive values, and agreement of Ribotest Mycoplasma® test compared to cell culture method




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download