Abstract
Background
Methods
Results
Acknowledgements
Notes
Authors' Disclosures of Potential Conflicts of Interest: The authors declare that they have no conflict of interests.
Author Contributions:
Research conception and design: Ji Hun Jeong, Yiel Hea Seo, Jeong Yeal Ahn, Kyung Hee Kim, Ja Young Seo, Pil Whan Park.
Data acquisition: Ji Hun Jeong.
Review of patients' clinical information: Ka Yeong Chun, Yong Su Lim.
Data analysis and interpretation: Ji Hun Jeong, Pil Whan Park.
Statistical analysis: Ji Hun Jeong.
Drafting of the manuscript: Ji Hun Jeong, Pil Whan Park.
Critical revision of the manuscript: Ji Hun Jeong, Pil Whan Park.
Approval of final manuscript: all authors.
References
Fig. 1
ROC curves for CK-MB, TnI, and copeptin according to the time of chest pain onset in AMI patients. The ROC curve plots sensitivity on the Y-axis and (1-specificity) on the X-axis. The area under the curve (AUC) of each marker is presented in each graph. Copeptin as a single marker was superior to CK-MB within six hours [(A)–(C)] and to TnI within two hours [(A) & (B)] in the STEMI group (P<0.05). Copeptin showed better diagnostic performance than CK-MB within two hours [(G) & (H)] and TnI within one hour in the AMI group (G) (P<0.05).
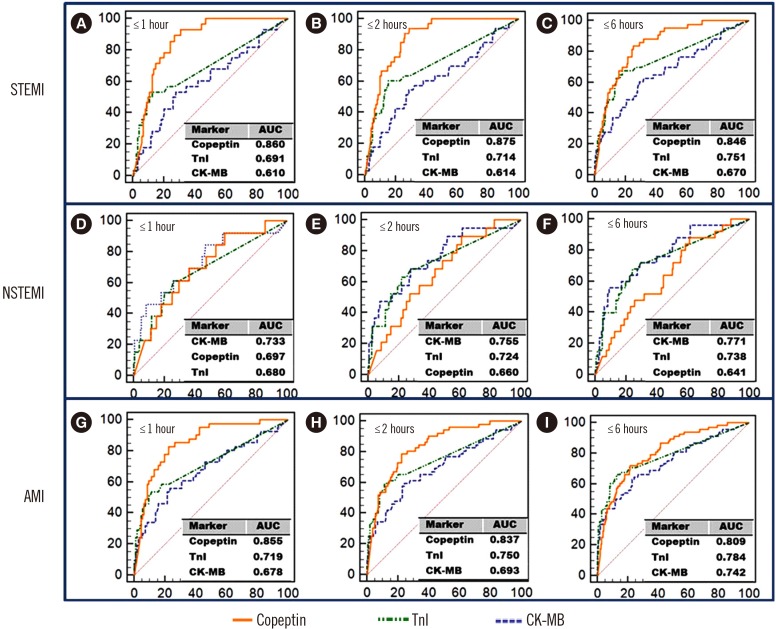
Fig. 2
ROC curves for combined markers according to the time of chest pain onset in AMI patients. A combination of the three markers showed the highest ROC curves in all patient groups. Copeptin plus TnI showed better diagnostic performance than TnI plus CK-MB in the STEMI [(A)–(C)] and AMI [(G)–(I)] groups (P<0.05).
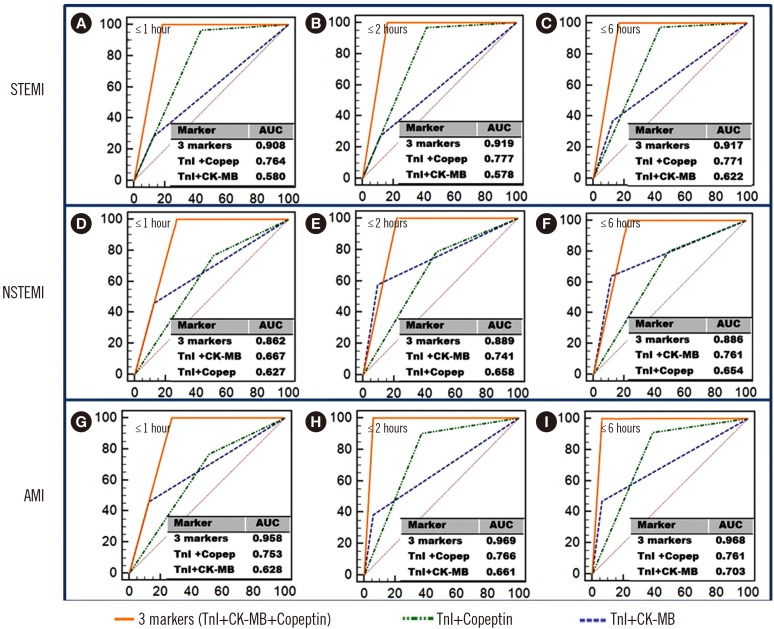
Table 1
Baseline characteristics of the 271 patients by disease group
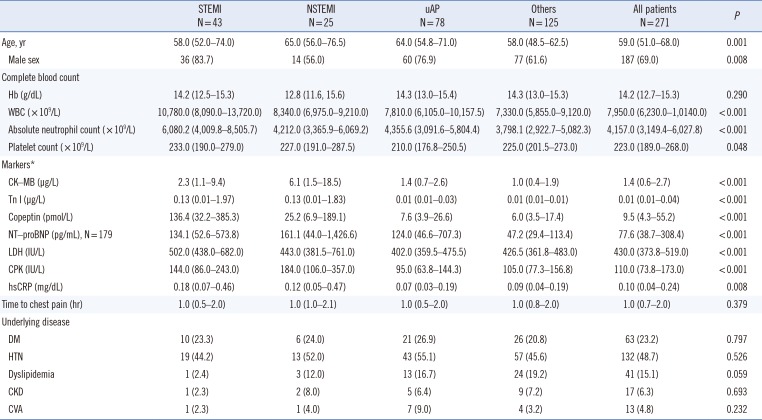
Continuous variables are summarized as median (IQR), and categorical values are summarized as number (%).
Characteristics were analyzed using the Kruskal-Wallis test for continuous variables or Pearson's chi square test for categorical variables.
*Reference ranges of markers: CK-MB, 0.0–5.0 µg/L; Tn I, 0.0–0.78 µg/L; copeptin, 0.0–10.0 pmol/L; NT-proBNP has different reference ranges by age; LDH, 200–485 IU/L; CPK, 32–294 IU/L in males and 33–211 IU/L in females; hsCRP, 0.0–0.5 mg/dL.
Abbreviations: STEMI, ST elevation myocardial infarction; NSTEMI, non-ST elevation myocardial infarction; uAP, unstable angina pectoris; CK-MB, creatine kinase myocardial band; IQR, interquartile range; WBC, white blood cell; ANC, absolute neutrophil count; CK-MB, creatine kinase myocardial band; TnI, Troponin I; NT-proBNP, N-terminal probrain natriuretic peptide; LDH, lactate dehydrogenase; CPK, creatine phosphokinase; hsCRP, high sensitivity C-reactive protein; DM, diabetes mellitus; HTN, hypertension; CKD, chronic kidney disease; CVA, cerebrovascular accident.
Table 2
Sensitivity, specificity, PPV, and NPV for CK-MB, TnI, and copeptin according to the time of chest pain onset in AMI patients
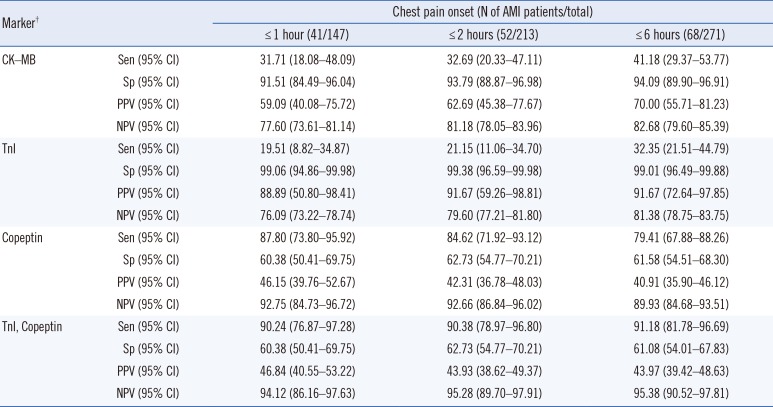
Table 3
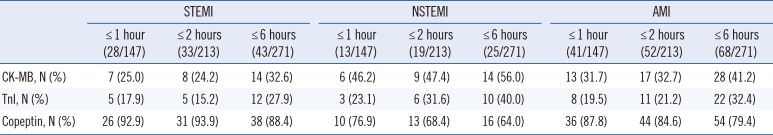
Elevation of cardiac markers according to the time of chest pain onset in AMI patients




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download