Abstract
Sarcoidosis is a multisystem disease characterized by noncaseating granulomas. Cardiac involvement is known to have poor prognosis because it can manifest as a serious condition such as the conduction abnormality, heart failure, ventricular arrhythmia, or sudden cardiac death. Although early diagnosis and early treatment is critical to improve patient prognosis, the diagnosis of CS is challenging in most cases. Diagnosis usually relies on endomyocardial biopsy (EMB), but its diagnostic yield is low due to the incidence of patchy myocardial involvement. Guidelines for the diagnosis of CS recommend a combination of clinical, electrocardiographic, and imaging findings from various modalities, if EMB cannot confirm the diagnosis. Especially, the role of advanced imaging such as cardiac magnetic resonance (CMR) imaging and positron emission tomography (PET), has shown to be important not only for the diagnosis, but also for monitoring treatment response and prognostication. CMR can evaluate cardiac function and fibrotic scar with good specificity. Late gadolinium enhancement (LGE) in CMR shows a distinctive enhancement pattern for each disease, which may be useful for differential diagnosis of CS from other similar diseases. Effectively, T1 or T2 mapping techniques can be also used for early recognition of CS. In the meantime, PET can detect and quantify metabolic activity and can be used to monitor treatment response. Recently, the use of a hybrid CMR-PET has introduced to allow identify patients with active CS with excellent co-localization and better diagnostic accuracy than CMR or PET alone. However, CS may show various findings with a wide spectrum, therefore, radiologists should consider the possible differential diagnosis of CS including myocarditis, dilated cardiomyopathy (DCM), hypertrophic cardiomyopathy, amyloidosis, and arrhythmogenic right ventricular cardiomyopathy. Radiologists should recognize the differences in various diseases that show the characteristics of mimicking CS, and try to get an accurate diagnosis of CS.
References
1. Statement on sarcoidosis. Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. Am J Respir Crit Care Med. 1999; 160:736–755.
3. Silverman KJ, Hutchins GM, Bulkley BH. Cardiac sarcoid: a clinicopathologic study of 84 unselected patients with systemic sarcoidosis. Circulation. 1978; 58:1204–1211.

4. Lynch JP 3rd, Hwang J, Bradfield J, Fishbein M, Shivkumar K, Tung R. Cardiac involvement in sarcoidosis: evolving concepts in diagnosis and treatment. Semin Respir Crit Care Med. 2014; 35:372–390.

5. Yazaki Y, Isobe M, Hiroe M, et al. Prognostic determinants of longterm survival in Japanese patients with cardiac sarcoidosis treated with prednisone. Am J Cardiol. 2001; 88:1006–1010.

6. Ardehali H, Howard DL, Hariri A, et al. A positive endomyocardial biopsy result for sarcoid is associated with poor prognosis in patients with initially unexplained cardiomyopathy. Am Heart J. 2005; 150:459–463.

7. Hillerdal G, Nou E, Osterman K, Schmekel B. Sarcoidosis: epidemiology and prognosis. A 15-year European study. Am Rev Respir Dis. 1984; 130:29–32.
8. Morimoto T, Azuma A, Abe S, et al. Epidemiology of sarcoidosis in Japan. Eur Respir J. 2008; 31:372–379.

9. Varron L, Cottin V, Schott AM, Broussolle C, Seve P. Late-onset sarcoidosis: a comparative study. Medicine (Baltimore). 2012; 91:137–143.
10. Perry A, Vuitch F. Causes of death in patients with sarcoidosis. A morphologic study of 38 autopsies with clinicopathologic correlations. Arch Pathol Lab Med. 1995; 119:167–172.
11. Iwai K, Tachibana T, Takemura T, Matsui Y, Kitaichi M, Kawabata Y. Pathological studies on sarcoidosis autopsy. I. Epidemiological features of 320 cases in Japan. Acta Pathol Jpn. 1993; 43:372–376.

12. Mehta D, Lubitz SA, Frankel Z, et al. Cardiac involvement in patients with sarcoidosis: diagnostic and prognostic value of outpatient testing. Chest. 2008; 133:1426–1435.
14. Uemura A, Morimoto S, Hiramitsu S, Kato Y, Ito T, Hishida H. Histologic diagnostic rate of cardiac sarcoidosis: evaluation of endomyocardial biopsies. Am Heart J. 1999; 138:299–302.

15. Tavora F, Cresswell N, Li L, Ripple M, Solomon C, Burke A. Comparison of necropsy findings in patients with sarcoidosis dying suddenly from cardiac sarcoidosis versus dying suddenly from other causes. Am J Cardiol. 2009; 104:571–577.

16. Birnie DH, Nery PB, Ha AC, Beanlands RS. Cardiac sarcoidosis. J Am Coll Cardiol. 2016; 68:411–421.

17. Youssef G, Beanlands RS, Birnie DH, Nery PB. Cardiac sarcoidosis: applications of imaging in diagnosis and directing treatment. Heart. 2011; 97:2078–2087.

18. Sekhri V, Sanal S, Delorenzo LJ, Aronow WS, Maguire GP. Cardiac sarcoidosis: a comprehensive review. Arch Med Sci. 2011; 7:546–554.

19. Hiraga HHM, Iwai K. Guidelines for diagnosis of cardiac sarcoidosis: study report on diffuse pulmonary diseases (in Japanese). Tokyo: The Japanese Ministry of Health and Welfare. 1993. 23–24.
20. Soejima K, Yada H. The workup and management of patients with apparent or subclinical cardiac sarcoidosis: with emphasis on the associated heart rhythm abnormalities. J Cardiovasc Electrophysiol. 2009; 20:578–583.

21. Youssef G, Leung E, Mylonas I, et al. The use of 18F-FDG PET in the diagnosis of cardiac sarcoidosis: a systematic review and metaanalysis including the Ontario experience. J Nucl Med. 2012; 53:241–248.

22. Blankstein R, Osborne M, Naya M, et al. Cardiac positron emission tomography enhances prognostic assessments of patients with suspected cardiac sarcoidosis. J Am Coll Cardiol. 2014; 63:329–336.

23. Kouranos V, Wells AU, Sharma R, Underwood SR, Wechalekar K. Advances in radionuclide imaging of cardiac sarcoidosis. Br Med Bull. 2015; 115:151–163.

24. Birnie DH, Sauer WH, Bogun F, et al. HRS expert consensus statement on the diagnosis and management of arrhythmias associated with cardiac sarcoidosis. Heart Rhythm. 2014; 11:1305–1323.

25. Hulten E, Aslam S, Osborne M, Abbasi S, Bittencourt MS, Blankstein R. Cardiac sarcoidosis-state of the art review. Cardiovasc Diagn Ther. 2016; 6:50–63.
26. Jeudy J, Burke AP, White CS, Kramer GB, Frazier AA. Cardiac sarcoidosis: the challenge of radiologic-pathologic correlation: from the radiologic pathology archives. Radiographics. 2015; 35:657–679.
27. Puntmann VO, Isted A, Hinojar R, Foote L, Carr-White G, Nagel E. T1 and T2 mapping in recognition of early cardiac involvement in systemic sarcoidosis. Radiology. 2017; 285:63–72.

28. Kouranos V, Tzelepis GE, Rapti A, et al. Complementary role of CMR to conventional screening in the diagnosis and prognosis of cardiac sarcoidosis. JACC Cardiovasc Imaging. 2017; 10:1437–1447.

29. Patel MR, Cawley PJ, Heitner JF, et al. Detection of myocardial damage in patients with sarcoidosis. Circulation. 2009; 120:1969–1977.

30. Greulich S, Deluigi CC, Gloekler S, et al. CMR imaging predicts death and other adverse events in suspected cardiac sarcoidosis. JACC Cardiovasc Imaging. 2013; 6:501–511.

31. Ise T, Hasegawa T, Morita Y, et al. Extensive late gadolinium enhancement on cardiovascular magnetic resonance predicts adverse outcomes and lack of improvement in LV function after steroid therapy in cardiac sarcoidosis. Heart. 2014; 100:1165–1172.

32. Crouser ED, Ono C, Tran T, He X, Raman SV. Improved detection of cardiac sarcoidosis using magnetic resonance with myocardial T2 mapping. Am J Respir Crit Care Med. 2014; 189:109–112.
33. Perez IE, Garcia MJ, Taub CC. Multimodality imaging in cardiac sarcoidosis: is there a winner? Curr Cardiol Rev. 2016; 12:3–11.
34. Orii M, Imanishi T, Akasaka T. Assessment of cardiac sarcoidosis with advanced imaging modalities. Biomed Res Int. 2014; 2014:897956.

35. Roberts WC, McAllister HA Jr, Ferrans VJ. Sarcoidosis of the heart. A clinicopathologic study of 35 necropsy patients (group 1) and review of 78 previously described necropsy patients (group 11). Am J Med. 1977; 63:86–108.
36. Greulich S, Kitterer D, Latus J, et al. Comprehensive cardiovascular magnetic resonance assessment in patients with sarcoidosis and preserved left ventricular ejection fraction. Circ Cardiovasc Imaging. 2016; 9:e005022.

37. Ferreira VM, Piechnik SK. Seeing beyond the obvious: subclinical cardiac sarcoidosis revealed by cardiovascular magnetic resonance mapping. Circ Cardiovasc Imaging. 2016; 9:e005592.
38. Hinojar R, Foote L, Arroyo Ucar E, et al. Native T1 in discrimination of acute and convalescent stages in patients with clinical diagnosis of myocarditis: a proposed diagnostic algorithm using CMR. JACC Cardiovasc Imaging. 2015; 8:37–46.
39. Manoushagian SJ, Lakhter V, Patil PV. Multimodality imaging in the diagnosis and management of cardiac sarcoidosis. J Nucl Cardiol. 2017; 24:29–33.

40. Langah R, Spicer K, Gebregziabher M, Gordon L. Effectiveness of prolonged fasting 18f-FDG PET-CT in the detection of cardiac sarcoidosis. J Nucl Cardiol. 2009; 16:801–810.

41. Ishimaru S, Tsujino I, Takei T, et al. Focal uptake on 18F-fluoro-2-deoxyglucose positron emission tomography images indicates cardiac involvement of sarcoidosis. Eur Heart J. 2005; 26:1538–1543.

42. Harisankar CN, Mittal BR, Agrawal KL, Abrar ML, Bhattacharya A. Utility of high fat and low carbohydrate diet in suppressing myocardial FDG uptake. J Nucl Cardiol. 2011; 18:926–936.

43. Betensky BP, Tschabrunn CM, Zado ES, et al. Longterm follow-up of patients with cardiac sarcoidosis and implantable cardioverter-defibrillators. Heart Rhythm. 2012; 9:884–891.

44. Ahmadian A, Brogan A, Berman J, et al. Quantitative interpretation of FDG PET/CT with myocardial perfusion imaging increases diagnostic information in the evaluation of cardiac sarcoidosis. J Nucl Cardiol. 2014; 21:925–939.

45. Gormsen LC, Haraldsen A, Kramer S, Dias AH, Kim WY, Borghammer P. A dual tracer (68)Ga-DOTANOC PET/CT and (18)F-FDG PET/CT pilot study for detection of cardiac sarcoidosis. EJNMMI Res. 2016; 6:52.

46. Manabe O, Hirata K, Shozo O, et al. (18)F-fluoromisonidazole (FMISO) PET may have the potential to detect cardiac sarcoidosis. J Nucl Cardiol. 2017; 24:329–331.

47. Ohira H, Tsujino I, Ishimaru S, et al. Myocardial imaging with 18F-fluoro-2-deoxyglucose positron emission tomography and magnetic resonance imaging in sarcoidosis. Eur J Nucl Med Mol Imaging. 2008; 35:933–941.

48. Dweck MR, Abgral R, Trivieri MG, et al. Hybrid magnetic resonance imaging and positron emission tomography with fluorodeoxyglucose to diagnose active cardiac sarcoidosis. JACC Cardiovasc Imaging. 2018; 11:94–107.
49. Wicks EC, Menezes LJ, Barnes A, et al. Diagnostic accuracy and prognostic value of simultaneous hybrid 18F-fluorodeoxyglucose positron emission tomography/magnetic resonance imaging in cardiac sarcoidosis. Eur Heart J Cardiovasc Imaging. 2018; 19:757–767.

50. Ishida Y, Yoshinaga K, Miyagawa M, et al. Recommendations for (18)F-fluorodeoxyglucose positron emission tomography imaging for cardiac sarcoidosis: Japanese Society of Nuclear Cardiology recommendations. Ann Nucl Med. 2014; 28:393–403.

51. Friedrich MG, Sechtem U, Schulz-Menger J, et al. Cardiovascular magnetic resonance in myocarditis: a JACC white paper. J Am Coll Cardiol. 2009; 53:1475–1487.

52. Yilmaz A, Ferreira V, Klingel K, Kandolf R, Neubauer S, Sechtem U. Role of cardiovascular magnetic resonance imaging (CMR) in the diagnosis of acute and chronic myocarditis. Heart Fail Rev. 2013; 18:747–760.

53. Yazaki Y, Isobe M, Hiramitsu S, et al. Comparison of clinical features and prognosis of cardiac sarcoidosis and idiopathic dilated cardiomyopathy. Am J Cardiol. 1998; 82:537–540.

54. Vignaux O. Cardiac sarcoidosis: spectrum of MRI features. AJR Am J Roentgenol. 2005; 184:249–254.

55. Moraes GL, Higgins CB, Ordovas KG. Delayed enhancement magnetic resonance imaging in nonischemic myocardial disease. J Thorac Imaging. 2013; 28:84–92. quiz 93–95.

56. Marcus FI, McKenna WJ, Sherrill D, et al. Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: proposed modification of the task force criteria. Circulation. 2010; 121:1533–1541.
57. Steckman DA, Schneider PM, Schuller JL, et al. Utility of cardiac magnetic resonance imaging to differentiate cardiac sarcoidosis from arrhythmogenic right ventricular cardiomyopathy. Am J Cardiol. 2012; 110:575–579.

58. Bartlett ML, Bacharach SL, Voipio-Pulkki LM, Dilsizian V. Artifactual inhomogeneities in myocardial PET and SPECT scans in normal subjects. J Nucl Med. 1995; 36:188–195.
Fig. 1.
Acute cardiac sarcoidosis (CS) in a 54-year-old man who presented as an acute atypical chest pain. Short-axis late gadolinium enhanced (LGE) MR images (a) show multifocal patchy enhancement in the anteroseptal and inferoseptal wall of basal LV level (upper row) and inferoseptal wall of midventricuar LV level (lower row). Notice the enhancement is located in the noncoronary vascular territory and preserved subendocardial layer. Short-axis T2-weighted MR images (b) show high signal intensity (arrows) in the corresponding regions with the enhancement area, which is suggested of the myocardial edema.
Fig. 2.
Chronic CS in a 74-year-old woman who complained dyspnea. Short-axis SSFP MR cine image (a) shows decreased wall motion and decreased wall thickness of inferolateral wall (arrows) at the basal LV level. Short-axis LGE MR image (b) shows transmural enhancement at thinned inferolateral wall (arrows) and multifocal patchy enhancement in the inferoseptal wall (arrowheads).
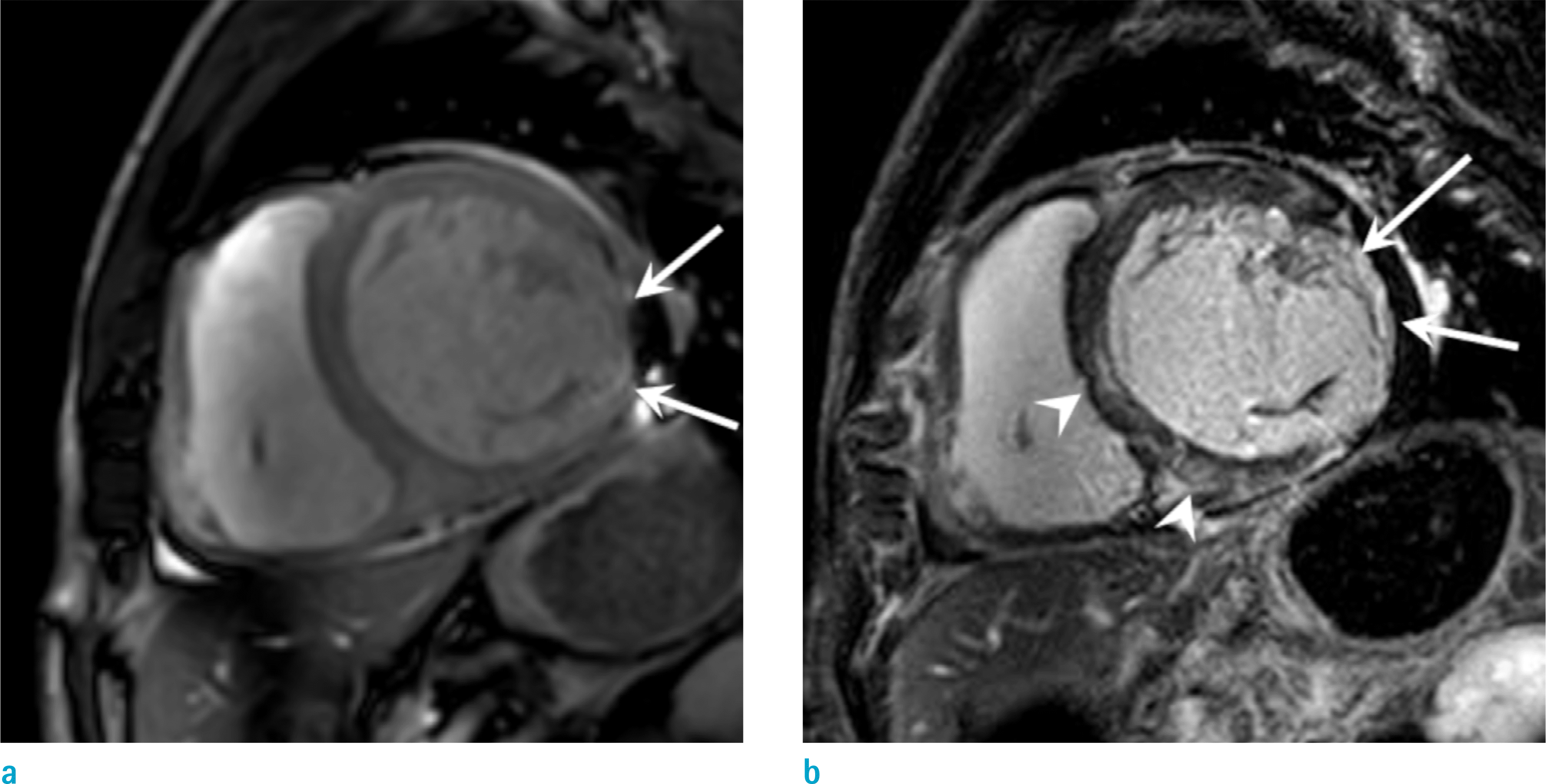
Fig. 3.
Suspected CS in a 36-year-old man who complained orthopnea. Short-axis LGE (a) shows subtle enhancement at midventricular inferoseptal wall and anterolateral wall (arrows). Pre-contrast T1 map (b) and post-contrast T1 map (c) shows increased native T1 value (1320 msec) and increased extracellular volume (35.3%) than normal control value, which is helpful for the early diagnosis of a cardiac involvement of sarcoidosis.
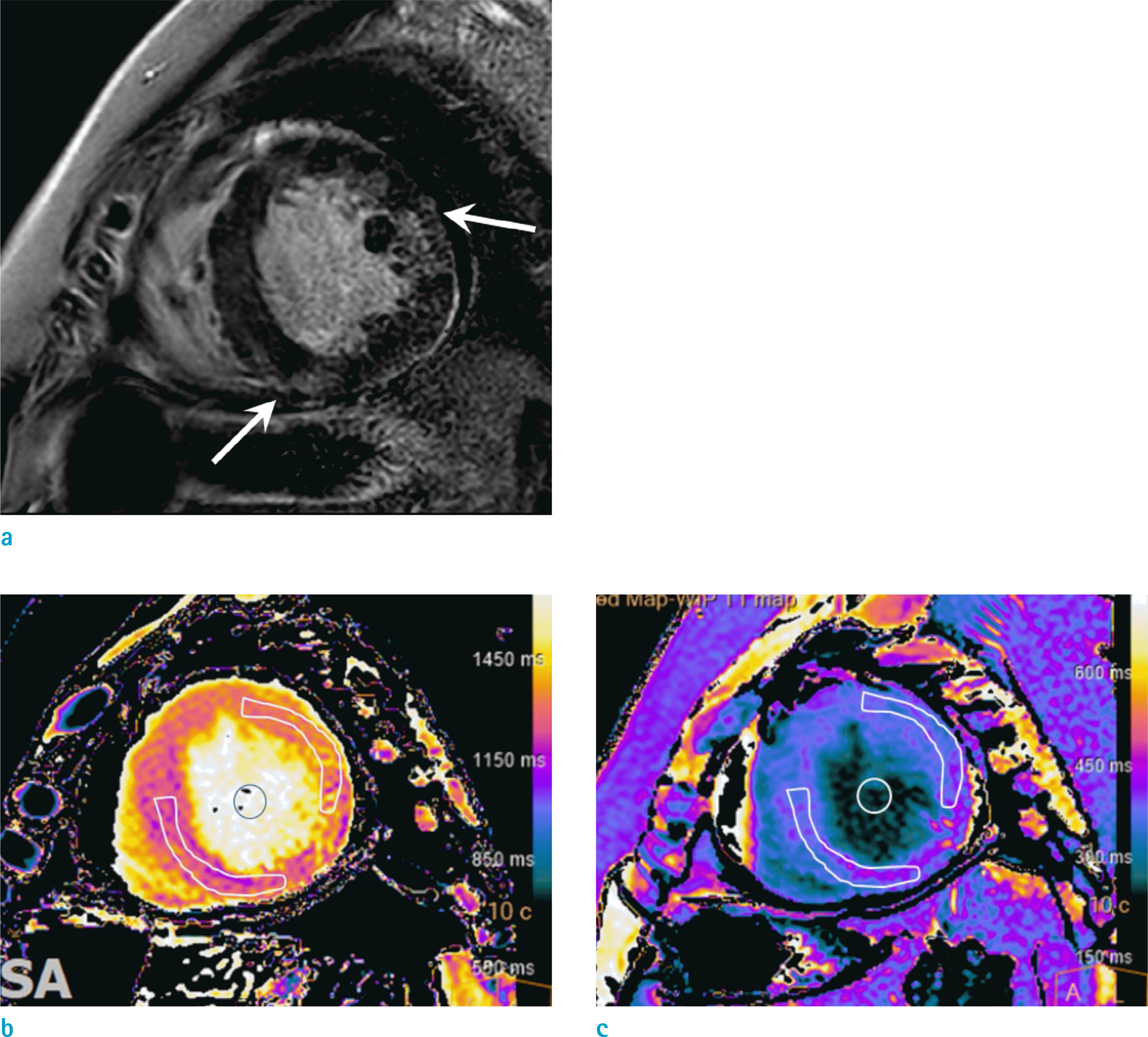
Fig. 4.
Multimodality imaging in a 59-year-old woman with systemic sarcoidosis. Non-ECG gated chest CT images with mediastinal window setting (a) show enlarged lymph nodes along the mediastinum (arrowheads) and lung window setting (b) show scattered several lung nodules in both lung (arrows). Four-chamber LGE MR image (c) shows subepicardial enhancement (arrowheads) at the basal septal wall and midventricular to basal lateral wall. The FDG PET images (d, e) show increased uptake in mediastinal lymph nodes (arrowheads in d) and septal and lateral wall of the LV (arrowheads in e) which are at a corresponding region with enhancement on the LGE MR image (arrowheads in c).

Fig. 5.
Conjoined imaging of PET and MR in a 59-year-old woman with cardiac sarcoidosis. Short-axis SSFP cine image (a) shows hypokinesia at the region of the midventricular inferoseptal wall (arrow), with decreased EF as 48%. Short-axis T2-weighted image (b) shows high signal intensity at inferoseptal wall of the LV (arrow), suggesting edema. The LGE MR image (c) shows subepicardial enhancement at concordant area, indicating fibrosis. Conjoined PET-MR image (d) reveals increased uptake (arrow) at the enhanced area of the inferoseptal wall in LGE MR image, which is well presented in the active inflammatory phase of CS (c and d images are LGE MR image and PET image matching the same level).

Fig. 6.
Acute myocarditis in a 28-year-old man who presented with the syncope and elevated cardiac enzyme. Short-axis LGE images (a) show multifocal patchy enhancement at the subepicardial layer of anterolateral and inferoseptal wall with preserved subendocardial layer. T2-weighted images (b) show high signal intensity in the same regions (arrows) with enhanced area. The condition resembles CS, therefore clinical presentation and other blood tests should be carefully considered.
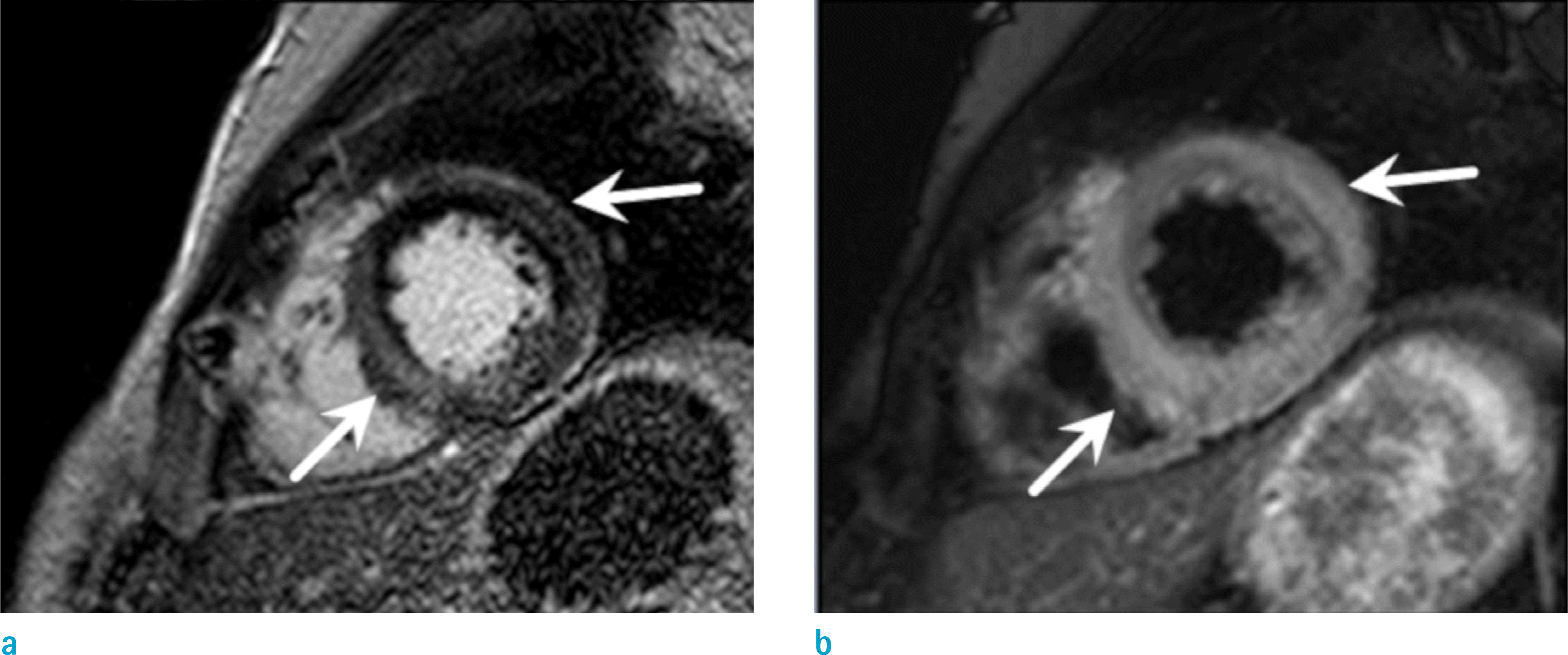
Fig. 7.
Dilated cardiomyopathy in a 54-year-old woman who complained of dyspnea and a progressed heart failure. Short-axis LGE MR image shows dilated LV chamber and linear enhancement at the midwall region of the interventricular septum (arrows).
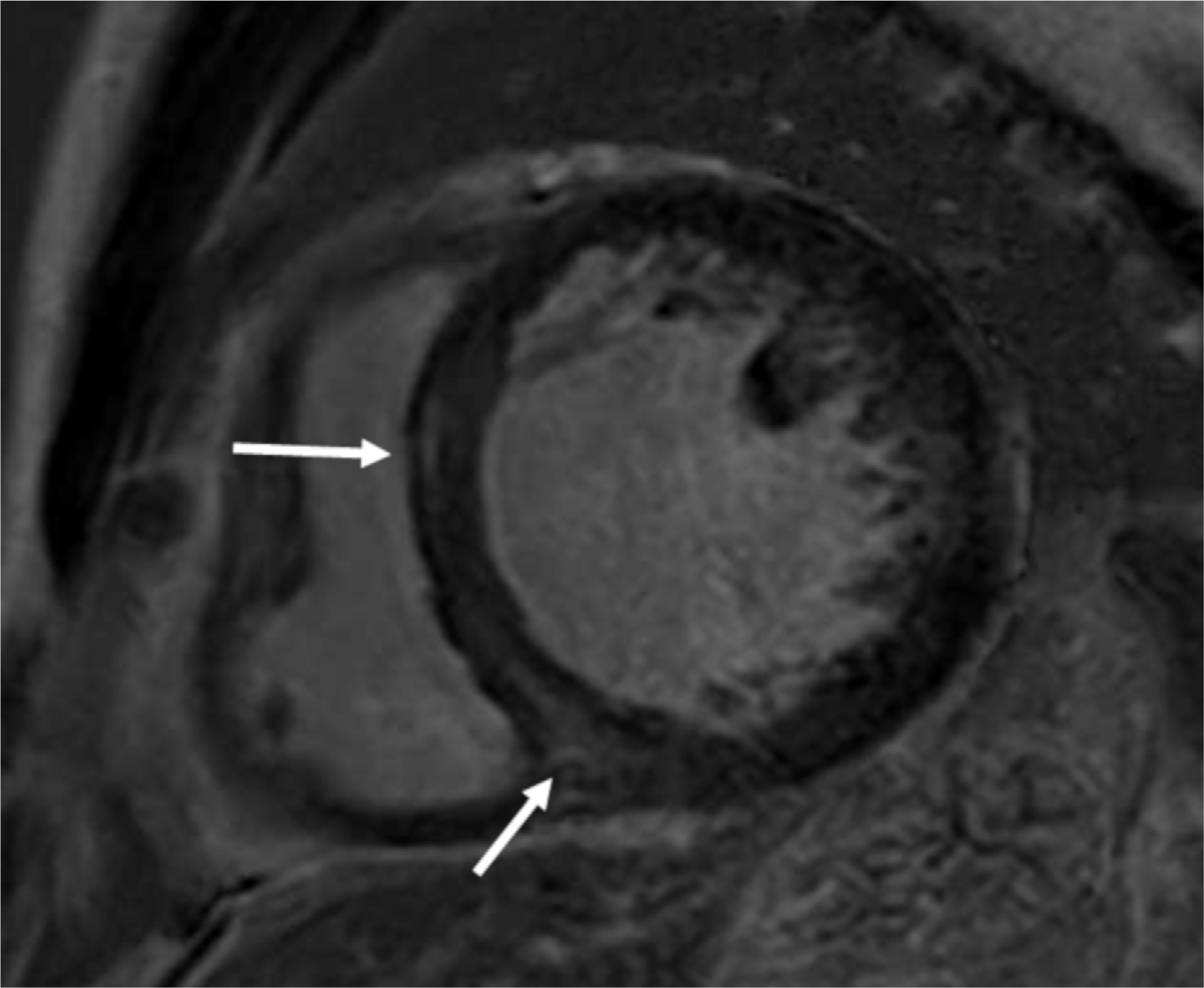
Fig. 8.
Hypertrophic cardiomyopathy in a 58-year-old woman with recurrent ventricular tachycardia. Short-axis LGE MR image (a) shows multifocal patchy enhancement (arrows) at hypertrophied anteroseptal wall, around the anterior RV insertion sites. However, the T2-weighted image (b) show no evidence of abnormally increased signal intensity.
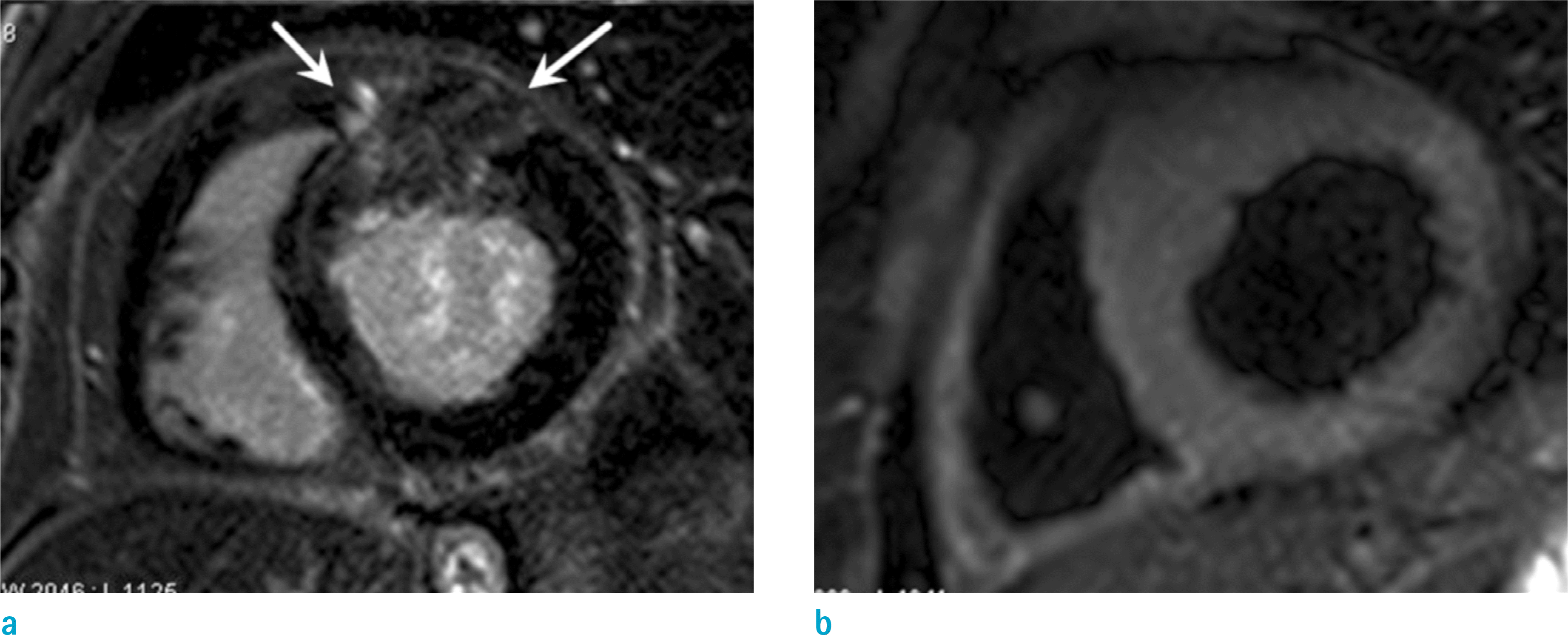
Fig. 9.
Cardiac amyloidosis in a 68-year-old man with exertional dyspnea. Short-axis LGE image shows global subendocardial enhancement (arrows) at whole LV wall. Subtle enhancement is also noted in RV free wall (arrowheads).
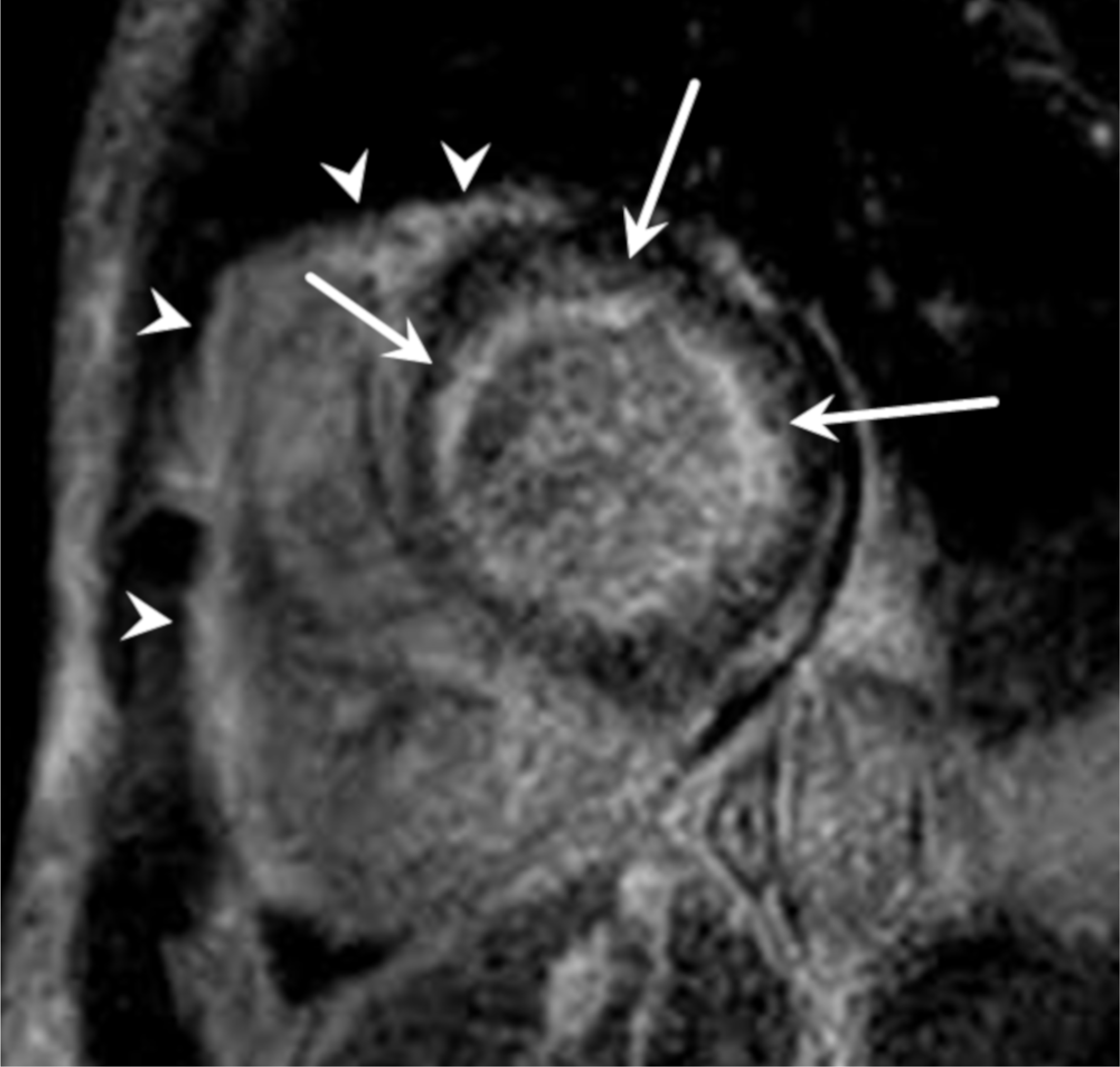
Table 1.
JMHW 2006 Revised Guidelines for Diagnosis of CS
| Histologic diagnosis group |
| CS is confirmed when EMB specimens show noncaseating epithelioid cell granulomas with histologic clinical diagnosis of extracardiac sarcoidosis |
| Clinical diagnosis group |
| Although EMB specimens do not show noncaseating epithelioid cell granulomas, extracardiac sarcoidosis was diagnosed histologically or clinically and satisfies the following criteria* |
| Major criteria |
| Advanced AV block |
| Basal thinning of the interventricular septum |
| Positive gallium uptake in the heart |
| Depressed LVEF (< 50%) |
| Minor criteria |
| Abnormal ECG findings: ventricular arrhythmias (VT, multifocal or frequent PVC), complete RBBB, axis deviation, or abnormal Q wave |
| Abnormal echocardiographic findings: regional abnormal wall motion or morphologic abnormality (ventricular aneurysm, wall thickening) |
| Nuclear medicine findings: perfusion defect detected with thallium 201 (201Th) or technetium 99m (99mTc) myocardial scintigraphy |
| MR imaging findings: delayed (late) gadolinium-induced enhancement of myocardium EMB findings: interstitial fibrosis or monocyte infiltration of more than moderate grade |
Table 2.
Heart Rhythm Society (HRS) Expert Consensus Recommendations on Criteria for Diagnosis of CS*
Table 3.
The Reported Common Distribution Patterns of Late Gadolinium Enhancement of Myocardium in Patients with Cardiac Sarcoidosis
Table 4.
Differential Diagnosis of CS
| Diagnosis | Clinical characteristics | MRI findings |
|---|---|---|
| CS | Multisystem disease, conduction abnormality, | Subepicardial and mid wall LGE at basal septum or inferolateral wall |
| mediastinal lymphadenopathy | ||
| Myocarditis | Recent history of symptoms from viral infection | Subepicardial LGE in noncoronary pattern, edema in acute setting, |
| such as fever or chills, chest pain mimicking MI, | commonly involves lateral wall* | |
| elevated cardiac enzyme | ||
| Idiopathic | Heart failure, dyspnea on exertion, palpitations, | Dilated LV or both ventricles and reduced systolic function, no LGE or |
| DCM | asymptomatic cardiomegaly | linear mid wall LGE in interventricular septum* |
| HCM | Wide spectrum from asymptomatic to sudden | Increased LV wall thickness >15 mm, LGE at hypertrophied segment, |
| cardiac death | frequently involve at interventricular septum, adjacent RV insertion site* | |
| Amyloidosis | Multisystem disease, heart failure and dyspnea | Global subendocardial LGE |
| Thickening of LV wall, valve leaflets, and papillary muscles | ||
| Interatrial septal thickening and other chamber involvement* | ||
| ARVC | Family history, syncope or tachycardia | RV dilatation and focal dyskinesia or akinesia, fibrofatty infiltration of |
| Frequently RV involve (LV involvement in about | RV, especially in the outflow tract and anterobasal area* | |
| 10% of cases) |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download