Abstract
Background
Methods
Results
Figures and Tables
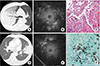 | Figure 1Consolidation. Case 1 was that of a 74-year-old man with dyspnea. (A) Chest computed tomography (CT) showing lobar consolidation with air bronchogram in the right upper lobe. (B) Radial probe endobronchial ultrasound (R-EBUS) in the posterior segmental bronchus of the right upper lobe revealing a homogenous, isoechoic tissue-like pattern around the probe and some hyperechoic dots away from the probe. (C) Histopathologic specimen from transbronchial lung biopsy (TBLB) showing chronic inflammation with organizing pneumonia pattern (H&E stain, ×200). His final diagnosis was organizing pneumonia secondary to bacterial infection. Case 2 was that of a 73-year-old man with febrile sensation. (D) Chest CT showing segmental consolidation in the lingular segment of the left upper lobe. (E) R-EBUS revealing a homogenous and isoechoic pattern around the probe. (F) Histopathologic specimen from TBLB showing round yeast-like fungi (Gomori's methenamine silver stain, ×400). His final diagnosis was pulmonary cryptococcosis. |
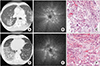 | Figure 2Ground-glass opacity (GGO). Case 3 was that of an 82-year-old man with dyspnea. (A) Chest computed tomography (CT) showing GGO in the posterior segment of the right upper lobe. (B) Radial probe endobronchial ultrasound (R-EBUS) revealing a mixed blizzard sign containing a heterogeneous acoustic shadow with hyperechoic dots, linear arcs, and vessels. (C) Histopathologic specimen from transbronchial lung biopsy (TBLB) showing organizing pneumonia pattern with focal atypical cells (H&E, ×200). His final diagnosis was cryptogenic organizing pneumonia. Case 4 was that of a 79-year-old man with febrile sensation. (D) Chest CT showing GGO in the right lower lobe. (E) R-EBUS in the anterobasal segmental bronchus of the right lower lobe, revealing a mixed blizzard sign. (F) Histopathologic specimen from TBLB showing organizing pneumonia (H&E stain, ×200). His final diagnosis was organizing pneumonia secondary to bacterial infection. |
Table 1
Baseline characteristics of the patients

Values are presented as median (range) or number (%).
*Statistically significant difference between the R-EBUS and blind TBLB groups at p<0.05 with the chi-square test. †Others: micronodules, fibrosis and alveolar interstitial thickening. ‡It is defined when emphysema is observed on chest CT irrespective of presence of respiratory symptoms, or abnormal pulmonary function test results. The reason for distinguishing emphysema from other comorbidities, is that it exerts a considerable influence on the incidence of pneumothorax. §It includes the following diseases: chronic obstructive pulmonary disease, bronchiectasis, post-tuberculous destroyed lung and pneumoconiosis.
R-EBUS: radial probe endobronchial ultrasound; TBLB: transbronchial lung biopsy; CT: computed tomography; GGO: ground-glass opacity.
Table 2
Comparison of the TBLB-related outcomes

Values are presented as median (range) or number (%).
*Statistically significant difference between the R-EBUS and blind TBLB groups at p<0.05 with the Mann-Whitney U test. †Biopsy was performed in two lobes in nine patients. ‡The size of the biopsy specimens was directly measured on a pathology slide in a pathology laboratory. TBLB: transbronchial lung biopsy; R-EBUS: radial probe endobronchial ultrasound; RUL: right upper lobe; RML: right middle lobe; RLL: right lower lobe; LUL: left upper lobe; LLL: left lower lobe.
Table 3
Comparison of the TBLB-related complications
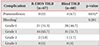
Values are presented as number (%).
*Statistically significant difference at p<0.05 with chi-square test between the R-EBUS and blind TBLB groups. †Bleeding severity is classified on an adapted scale: Grade 0, no bleeding; grade 1, bleeding requiring suction to clear but no other endoscopic procedures; grade 2, bleeding requiring endoscopic procedures (bronchial occlusion-collapse and/or instillation of ice-cold saline); grade 3, severe bleeding not controlled endoscopically, causing hemodynamic or respiratory instability, requiring surgical interventions or admission to the intensive care unit1617.
TBLB: transbronchial lung biopsy; R-EBUS: radial probe endobronchial ultrasound.
Table 4
Comparison of the TBLB results

Values are presented as number (%).
There were no statistically significant differences in the chi-square test between the R-EBUS and blind TBLB groups (p=0.385).
*Others: anthracosis one case, atypical cell one case, atypical gland one case.
TBLB: transbronchial lung biopsy; R-EBUS: radial probe endobronchial ultrasound.
Table 5
Comparison of the final diagnosis

Values are presented as number (%).
*The diagnostic yield was defined as the percentage of biopsy results that matched the final diagnosis (p=0.660). †Others: amyloidosis one case, pulmonary alveolar proteinosis one case.
R-EBUS: radial probe endobronchial ultrasound; TBLB: transbronchial lung biopsy; CTD: connective tissue disease; ILD: interstitial lung disease; NTM: non-tuberculous mycobacterium.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download