See the letter "Letter: Thyroid-Stimulating Hormone Reference Ranges in the First Trimester of Pregnancy in an Iodine-Sufficient Country (Endocrinol Metab 2018;33:466-72, Carmen Castillo et al.)" on page 93.
Difference in diagnostic criteria to define functional thyroid disease is a real and urgent problem today [1]. There is a general agreement in order that the best reference is the locally defined using the calculated 2.5th to 97.5th percentile as the thyroid-stimulating hormone (TSH) normal range.
The need to use local populations arises when recognizing that TSH levels can be affected by genetics or race, but there are other important factors, like environmental exposure to thyroid disruptors, obesity and iodine intake that could affects local thyroid parameters. In relationship to the last factors, National Academy of Clinical Biochemistry (NACB) and American Thyroid Association (ATA) guidelines [23] only include the recommendation to perform the studies in “normal iodinated population” but iodine status is variable in different countries; the use of iodised salt is not widespread worldwide; therefore, when we are trying to establish local TSH levels, it is necessary to know the past iodine history of the population.
TSH levels and hypothyroidism prevalence in Chile are higher than described elsewhere as Dr Kwon notes. It is possible that chronic high iodine intake exposition and high levels of obesity in our country could have a great role in these findings.
In 2016, we reported that body mass index (BMI) influenced pregnancy thyroid hormone reference ranges in Chilean women, we found that TSH distribution was shifted towards higher values and free thyroxine towards lower values in obese pregnant women compared to normal weight pregnant women [4], now we are reporting the influence of iodine [5].
The reason why urinary iodine (UI) levels were not measured in all participants was only because it is expensive and we do not have enough founding; however, we performed a randomization to have a representative sample of women with UI.
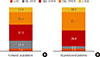
Exploring the relationship between hypothyroidism and iodine status, as Dr Kwon requires, our results showed that hypothyroid patients had a higher percentage of UI above the requirements or in excess than the population without hypothyroid patients as shown in Fig. 1.
It is possible that the differences in thyroid disease prevalence between countries could be related to prevalence of obesity or differences in iodine intake in populations or both. In the view of the new data, additional efforts are needed in order to reach an agreement regarding the definition of reference TSH values and how to include iodine exposition and BMI in the reference population definition.
Figures and Tables
References
1. Dong AC, Stagnaro-Green A. Differences in diagnostic criteria mask the true prevalence of thyroid disease in pregnancy: a systematic review and meta-analysis. Thyroid. 2019; 29:278–289.

2. The National Academy of Clinical Biochemistry: Standards of Laboratory Practice. Laboratory Support for the Diagnosis & Monitoring of Thyroid Disease [Internet]. Washington DC: AACC;2002. cited 2019 Apr 2. Available from: https://www.aacc.org/science-and-practice/practice-guidelines/thyroid-disease.
3. Alexander EK, Pearce EN, Brent GA, Brown RS, Chen H, Dosiou C, et al. 2017 Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and the postpartum. Thyroid. 2017; 27:315–389.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download