1. Fahy BG, Chau DF. The technology of processed electroencephalogram monitoring devices for assessment of depth of anesthesia. Anesth Analg. 2018; 126(1):111–117.

2. Checketts MR, Alladi R, Ferguson K, Gemmell L, Handy JM, Klein AA, et al. Recommendations for standards of monitoring during anaesthesia and recovery 2015: Association of Anaesthetists of Great Britain and Ireland. Anaesthesia. 2016; 71(1):85–93.
3. Johansen JW. Update on bispectral index monitoring. Best Pract Res Clin Anaesthesiol. 2006; 20(1):81–99.

4. Punjasawadwong Y, Phongchiewboon A, Bunchungmongkol N. Bispectral index for improving anaesthetic delivery and postoperative recovery. Cochrane Database Syst Rev. 2014; (6):CD003843.

5. Ku SW, Lee U, Noh GJ, Jun IG, Mashour GA. Preferential inhibition of frontal-to-parietal feedback connectivity is a neurophysiologic correlate of general anesthesia in surgical patients. PLoS One. 2011; 6(10):e25155.

6. Lee H, Noh GJ, Joo P, Choi BM, Silverstein BH, Kim M, et al. Diversity of functional connectivity patterns is reduced in propofol-induced unconsciousness. Hum Brain Mapp. 2017; 38(10):4980–4995.

7. Lioi G, Bell SL, Smith DC, Simpson DM. Measuring depth of anaesthesia using changes in directional connectivity: a comparison with auditory middle latency response and estimated bispectral index during propofol anaesthesia. Anaesthesia. 2019; 74(3):321–332.

8. Dahaba AA. Different conditions that could result in the bispectral index indicating an incorrect hypnotic state. Anesth Analg. 2005; 101(3):765–773.

9. Kim DH, Yoo JY, Kim JY, Ahn SH, Kim S, Min SK. Influence of electrocautery-induced electromagnetic interference on quantitative electroencephalographic monitoring of hypnosis during general anesthesia: comparison between the ADMS® and the BIS VISTATM. Korean J Anesthesiol. 2018; 71(5):368–373.
10. Kim YS, Chung D, Oh SK, Won YJ, Lee IO. Unusual elevation in Entropy but not in PSI during general anesthesia: a case report. BMC Anesthesiol. 2018; 18(1):22.

11. Chakrabarti D, Surve RM, Bs D, Masapu D. Intraoperative aberrant bispectral index values due to facial nerve monitoring. J Clin Anesth. 2017; 37:61–62.

12. Kim JH, Kim AN, Kim KW, Lee SI, Kim JY, Kim KT, et al. Interruption of bispectral index monitoring by nerve integrity monitoring during tympanoplasty: a case report. Korean J Anesthesiol. 2013; 64(2):161–163.

13. Lee HC, Jung CW. Vital Recorder-a free research tool for automatic recording of high-resolution time-synchronised physiological data from multiple anaesthesia devices. Sci Rep. 2018; 8(1):1527.

14. Kanoga S, Mitsukura Y. Eye blink artifact rejection in single-channel electroencephalographic signals by complete ensemble empirical mode decomposition and independent component analysis. Conf Proc IEEE Eng Med Biol Soc. 2015; 2015:121–124.

15. Miller A, Sleigh JW, Barnard J, Steyn-Ross DA. Does bispectral analysis of the electroencephalogram add anything but complexity? Br J Anaesth. 2004; 92(1):8–13.
16. Pilge S, Kreuzer M, Karatchiviev V, Kochs EF, Malcharek M, Schneider G. Differences between state entropy and bispectral index during analysis of identical electroencephalogram signals: a comparison with two randomised anaesthetic techniques. Eur J Anaesthesiol. 2015; 32(5):354–365.
17. Smith WD, Dutton RC, Smith NT. Measuring the performance of anesthetic depth indicators. Anesthesiology. 1996; 84(1):38–51.

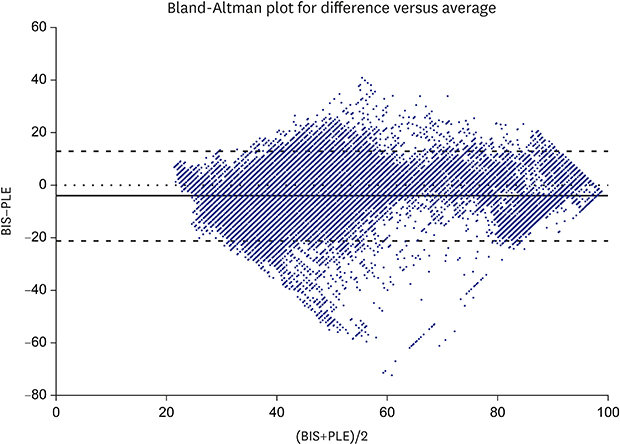
18. Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986; 1(8476):307–310.

19. Voss L, Sleigh J. Monitoring consciousness: the current status of EEG-based depth of anaesthesia monitors. Best Pract Res Clin Anaesthesiol. 2007; 21(3):313–325.

20. Nicolaou N, Hourris S, Alexandrou P, Georgiou J. EEG-based automatic classification of ‘awake’ versus ‘anesthetized’ state in general anesthesia using Granger causality. PLoS One. 2012; 7(3):e33869.

21. Barakat AR, Sutcliffe N, Schwab M. Effect site concentration during propofol TCI sedation: a comparison of sedation score with two pharmacokinetic models. Anaesthesia. 2007; 62(7):661–666.

22. Irwin MG, Hui TW, Milne SE, Kenny GN. Propofol effective concentration 50 and its relationship to bispectral index. Anaesthesia. 2002; 57(3):242–248.

23. Milne SE, Troy A, Irwin MG, Kenny GN. Relationship between bispectral index, auditory evoked potential index and effect-site EC50 for propofol at two clinical end-points. Br J Anaesth. 2003; 90(2):127–131.
24. Iannuzzi M, Iannuzzi E, Rossi F, Berrino L, Chiefari M. Relationship between Bispectral Index, electroencephalographic state entropy and effect-site EC50 for propofol at different clinical endpoints. Br J Anaesth. 2005; 94(4):492–495.
25. Kaskinoro K, Maksimow A, Långsjö J, Aantaa R, Jääskeläinen S, Kaisti K, et al. Wide inter-individual variability of bispectral index and spectral entropy at loss of consciousness during increasing concentrations of dexmedetomidine, propofol, and sevoflurane. Br J Anaesth. 2011; 107(4):573–580.

26. Kang MH, In CB, Kim MH, Lim KJ, Park EY, Lee HM, et al. Inappropriate elevation of bispectral index values in robot assisted thyroidectomy with electromyographic endotracheal tube: a case report. Korean J Anesthesiol. 2011; 61(6):511–514.

27. Sloan PA. Interference of bispectral index monitoring with intraoperative use of the electromyograph endotracheal tube. Can J Anaesth. 2007; 54(12):1028–1029.

28. Schuller PJ, Newell S, Strickland PA, Barry JJ. Response of bispectral index to neuromuscular block in awake volunteers. Br J Anaesth. 2015; 115:Suppl 1. i95–i103.

29. Chen X, Tang J, White PF, Wender RH, Ma H, Sloninsky A, et al. A comparison of patient state index and bispectral index values during the perioperative period. Anesth Analg. 2002; 95(6):1669–1674.

30. Liu PP, Wu C, Wu JZ, Zhang MZ, Zheng JJ, Shen Y, et al. The prediction probabilities for emergence from sevoflurane anesthesia in children: a comparison of the perfusion index and the bispectral index. Paediatr Anaesth. 2018; 28(3):281–286.










 PDF
PDF Citation
Citation Print
Print






 XML Download
XML Download