INTRODUCTION
Cowden's disease is a rare autosomal dominant, multiple hamartoma syndrome
1 characterized by multiple hamartoma of all three embryonic origins. It carries a high risk of breast, thyroid, and other cancers
2 as well as benign hamartomatous overgrowth of tissues (skin, colon, thyroid, etc.)
3. Several sporadic cases have also been reported. The common mucocutaneous findings include facial trichilemmomas, oral papillomas, and acral and palmoplantar keratosis
4. The original report in 1963 by Lloyd and Dennis first named the syndrome after their patient Rachel Cowden
5. Cowden's disease is the most common of the phosphatase and tensin homolog (PTEN) hamartomatous tumor syndromes, which includes other syndromes such as Bannayan-Railey-Ruvalcaba syndrome, Proteus syndrome, and Proteus-like syndrome
67.
This article presents a case of Cowden's disease with special emphasis on the clinical and pathologic features of the skin lesions on both hands and feet, and oral manifestations. We report this rare condition along with a brief review of the disease.
Go to :

CASE REPORT
A 21-year-old male patient presented with diffuse hyperkeratotic lesions with 1 to 2 mm tiny punctations on both his hands and feet. The patient reported that the lesions had slightly increased in number over the last 10 years without symptoms. He also presented with multiple localized erythematous papules on his forehead that had occurred about two years prior. He first visited the department of internal medicine due to repeated melena and hematochezia for about a month. While he underwent several examinations due to these symptoms, he also visited the department of dermatology for his cutaneous symptoms. There was no history of any other systemic disease or trauma. No other family member had similar lesions. Only his sister had a history of thyroid cancer.
On physical examination, multiple mucocutaneous lesions were observed. Multiple punctate hyperkeratotic macules were observed on both palms and soles (
Fig. 1A, B). The facial skin showed multiple skin-colored papules on his nose (
Fig. 1C). On his tongue and oral mucosa, several erythematous papules were noted, with appearance slightly like cobblestones or fibroma-like polyps (
Fig. 1D). We received the patient's consent form about publishing all photographic materials.
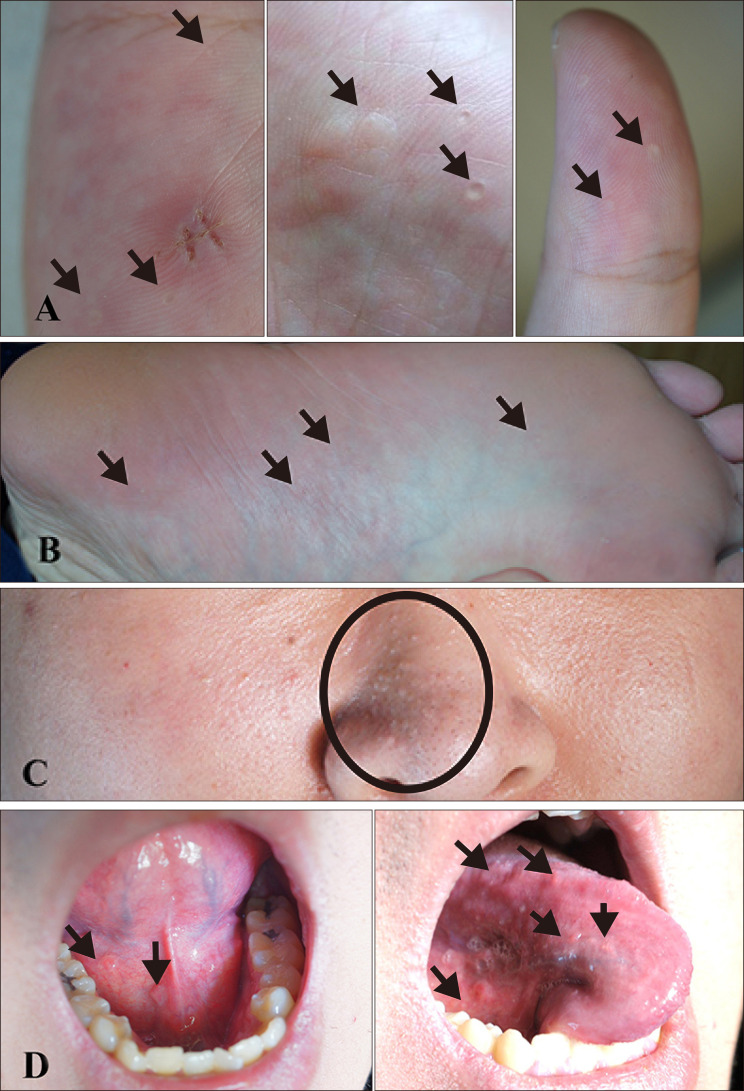 | Fig. 1Clinical images of the 21-year-old patient. Multiple punctate hyperkeratotic macules are localized on both (A) palms and both (B) soles (black arrows). Multiple skincolored papules are observed on his (C) nose (black circle). On his (D) tongue and oral mucosa, several erythematous papules are found resembling cobblestones or fibroma-like polyps (black arrows).
|
His esophagogastroduodenoscopy and colonofibroscopy were abnormal, with numerous hamartomas papillomatosis over the entire lumen surfaces of stomach, duodenum, and esophagus (
Fig. 2A). Biopsy of a gastric lesion and polypectomy showed histopathology of a gastric polyp with hyperplasty and an inflammatory polyp with prominent reactive lymphoid follicle (
Fig. 2B). Colonoscopy showed several small polyps (
Fig. 2C). Histopathology of a colorectal polyp showed that it was inflammatory (
Fig. 2D). In laboratory findings including a complete blood count, chemistry, and urinalysis, results were within normal range. The chest x-ray and electrocardiography showed no abnormal results.
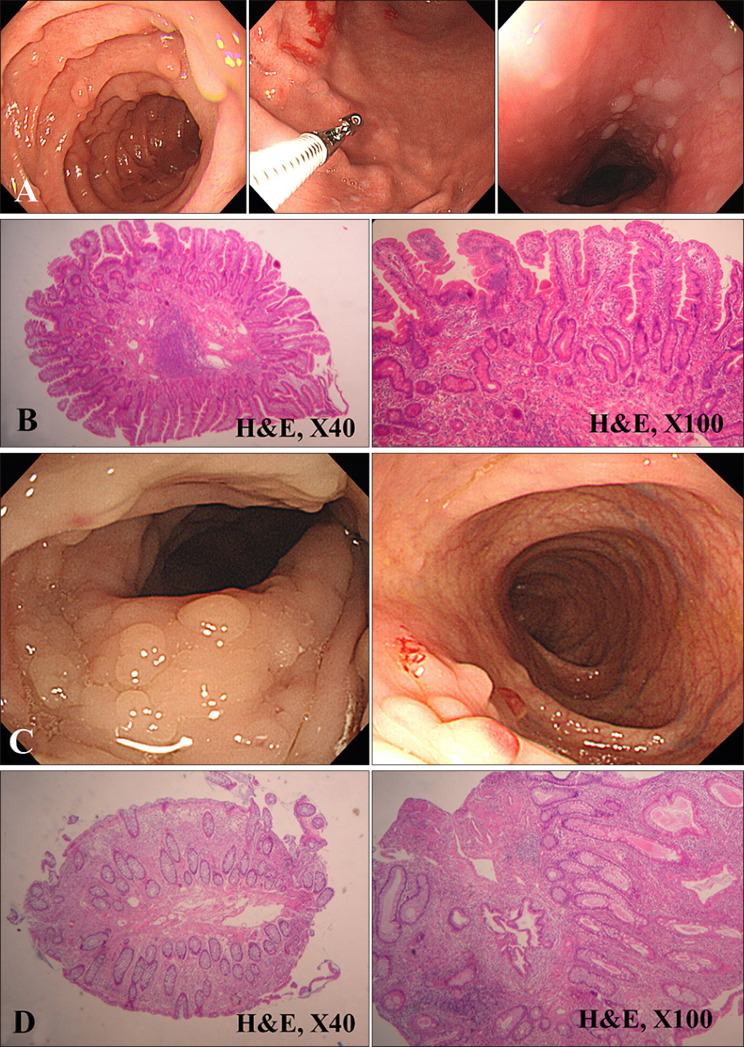 | Fig. 2(A) Endoscopic photograph showing multiple hamartomas papillomatosis in the duodenum and stomach, and esophageal keratosis like nodular lesions in the esophagus. (B) By biopsy of the gastric lesion and polypectomy, histopathology of a gastric polyp reveals a hyperplastic and inflammatory polyp with prominent reactive lymphoid follicle (H&E; ×40, ×100). (C) Colonoscopy shows several small polyps. (D) Histopathology of a colorectal polyp shows it to be inflammatory (H&E; ×40, ×100).
|
Skin biopsy was performed on the palm and forehead. Biopsy of a punctate lesion on the palm revealed palmoplantar keratosis with a large hyperkeratotic mount and thickened granular layer. Typical histologic backgrounds of palmoplantar keratoderma, including marked hyperkeratosis and epidermal proliferation, were noted. Slightly dilated blood vessels in the papillary dermis and a sparse infiltrate of lymphocytes were noted (
Fig. 3A, B). Biopsy of small erythematous papules from the forehead showed a plugging of the hair follicle by keratin with typical histologic backgrounds of folliculitis including perifollicular lymphocyte infiltration (
Fig. 3C, D).
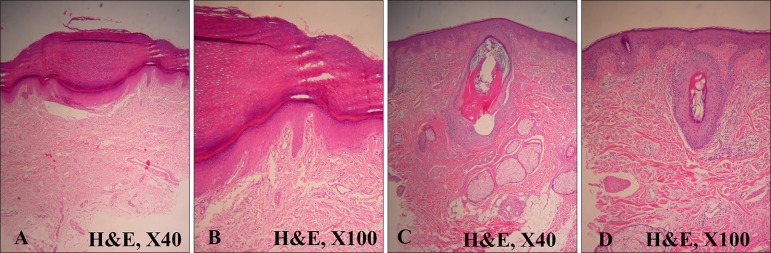 | Fig. 3(A) Hematoxylin and eosin (H&E) stained specimen from palm shows a large hyperkeratotic mount with thickened granular layer. Typical histologic backgrounds of palmoplantar keratosis, including marked hyperkeratosis and epidermal proliferation, are noted (H&E, ×40). (B) Epidermal hyperplasia with hypergranulosis, a thin layer of parakeratosis, and overlying orthokeratosis can be observed. Slightly dilated blood vessels in the papillary dermis and a sparse infiltrate of lymphocytes are noted (H&E, ×100). (C, D) A specimen from forehead shows plugging of the hair follicle by keratin with typical histologic backgrounds of acne including perifollicular lymphocyte infiltration (H&E; C: ×40, D: ×100).
|
The small bowel series showed a filling defect lesion, which suggested a localized mass or polyp, on the junction of duodenum 2nd and 3rd portion (2 cm long) (
Fig. 4A). No other specific filling defect was observed in the small bowel loops, especially on the terminal ileum (
Fig. 4B). On thyroid ultrasonography, several probable benign nodules were noted in the right thyroid gland.
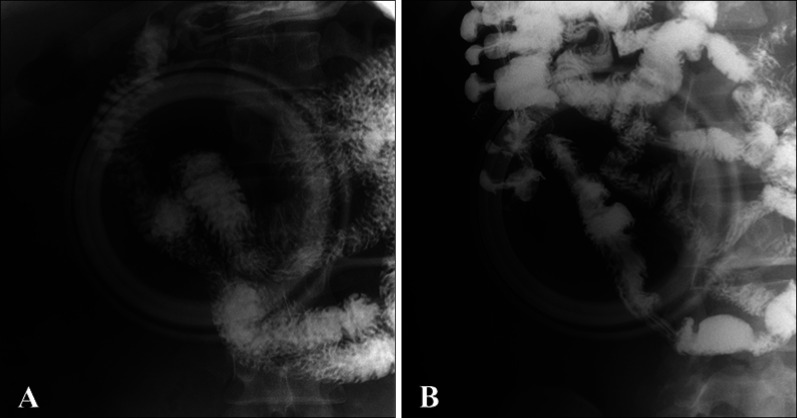 | Fig. 4The small bowel series shows (A) Filling defect lesion on the junction of duodenum 2nd and 3rd portion (2 cm long). (B) No other specific filling defect can be observed in the small bowel loops, especially on the terminal ileum.
|
Genetic analysis by polymerase chain reaction and direct sequencing of all nine exons of the
PTEN gene on chromosome 10q23 was performed. Sequence variation was described according to the recommendations in Gene Reviews (
www.genetests.org) and Genecard, using an mRNA reference sequence (NM.000314.4). Sequence analysis revealed a heterozygous mutation, c.388C>T (p.Arg130
*), in exon 5.
Go to :

DISCUSSION
The diagnostic criteria for Cowden's disease were proposed by Salem and Steck
8, who divided the diagnostic features into major criteria, minor criteria, and family history. Our patient was suitable for operational diagnosis of Cowden's disease by biopsy-proven multiple palmoplantar keratosis according to the criteria of the International Cowden Consortium (Operational Diagnostic Criteria)
9. These criteria are based on the presence of pathognomonic mucocutaneous lesions with multiple facial trichilemmomas, acral keratosis, mucous lesions, and papillomatous papules. Major and minor features included hamartomas (lipomas, fibromas, or adenomas) of other organs such as breast, thyroid, central nervous system, and gastrointestinal tract
9.
Mucocutaneous lesions are present in 90% to 100% of Cowden's disease cases so they are a prominent feature of the disease
1. Most often, the characteristic skin signs develop in the 2nd and 3rd decades, but onset age may range from 4 to 75 years
10. Cutaneous signs are usually the first presenting lesions of the disease
7. Our patient noticed the first skin signs after the age of about 10. Facial papules are the most frequent lesions, can be found in up to 86% of cases, and are preferentially localized in periorificial regions
10. A histologic review of facial lesions associated with Cowden's disease was done by Brownstein et al.
11 Many of the patients who underwent histologic examination of facial lesions showed facial trichilemmomas and it was diagnostic
10. The results indicate that most facial lesions belong to a spectrum of follicular abnormalities that include trichilemmomas and tumors of the follicular infundibulum. Our case showed histologic features of folliculitis, a follicular abnormality
10. Oral mucosal papillomatosis were seen in 83% of the patients reviewed
8. The histology of oral lesions was that of benign fibromas
8 or inflammatory fibro-epithelial hyperplasia
1. In Salem and Steck's study
8, palmoplantar keratosis was present in 42% of the reported cases and represented translucent, hard papules on the palms and soles that may show a central depression. The palmoplantar lesions were present in half of the patients reported in the literature, and our patient also showed palmoplantar keratosis confirmed by skin biopsy.
Further associated cutaneous findings include lipomas, hemangiomas, xanthomas, vitiligo, neuromas, café au lait spots, periorificial and acral lentigines, and acanthosis nigricans
10.
Including cutaneous symptoms, Cowden's disease can involve any organ, but especially breast, thyroid, and gastrointestinal tract
12. In 71% of cases, gastrointestinal polyps accompanied lesions anywhere in the gastrointestinal tract. The pathologic spectrum of the gastrointestinal polyps reported in Cowden's disease includes hamartomatous, juvenile, lipomatous, lymphomatous, inflammatory, hyperplastic, and occasionally adenomatous
1. Also, in our patient, multiple papillomatous lesions in the gastrointestinal tract were confirmed by endoscopic biopsy. If the breast or thyroid is affected, the condition can easily progress to malignancy, so cautious observation is needed
6. Our patient showed several probable benign nodules in the right thyroid gland on thyroid ultrasonography. It is known that the thyroid is affected in about 67% of cases. In this case, there was a family history of thyroid cancer (his sibling). Although the lesions are usually benign in nature, further evaluation is recommended
4.
The cause of Cowden's disease is not known. It is a familial hereditary, autosomal dominant condition with incomplete penetrance
4. In 1996, Nelen et al.
13 mapped Cowden's disease locus on the human chromosome.
PTEN, a tumor-suppressor gene, is frequently deleted at chromosome 10q23. PTEN appears to be a frequent mutation target in human tumors associated with genomic instability
1415. PTEN mutations were found to associate with phenotypes characteristic of hamartoma tumor syndromes
1617. Mutations in exons 5, 7, and 8 are likely to alter the phosphatase activity of the protein or result in a truncated protein
1819. Furthermore, biochemical analyses from previous studies have shown that most of the missense mutations found in the phosphatase domain, dramatically decrease or inactivate phosphatase activity
18.
Herein, we report a rare case of sporadic Cowden's disease, in which the patient had positive typical cutaneous features with cutaneous facial papules, oral mucosal papillomatosis, palmoplantar keratosis, and hamartomas of the gastrointestinal tract and thyroid gland. Further regular laboratory and image studies will be planned for our patient, as well as his family members.
Go to :








 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download