Abstract
There are a variety of different causes of parkinsonism including PD, secondary parkinsonism, and the parkinsonism plus syndromes. Secondary parkinsonism is caused by structural, toxic, metabolic, or infectious mechanisms. Among structural causes, intracranial neoplasms are a rare cause of secondary parkinsonism. Moreover, there are almost never case reports with intracranial space-occupying lesions resulting in parkinsonism associated with rapid cognitive impairment. Therefore, we report herein a 37-year-old woman diagnosed with papillary meningioma who presented with parkinsonism associated with rapidly progressive cognitive impairment mimicking diffuse Lewy body disease.
Parkinson's disease (PD) is a progressive neurological disorder characterized by tremor, rigidity, and slowness of movements, and is associated with progressive neuronal loss of the substantia nigra and other brain structures [1]. Accurate diagnosis of PD, however, continues to be challenging and previous clinicopathological studies have shown that clinicians diagnose the disease incorrectly in approximately 25% of patients [2]. There are a variety of different causes of parkinsonism including PD, secondary parkinsonism, and the parkinsonism plus syndromes. Secondary parkinsonism is caused by structural, toxic, metabolic, or infectious mechanisms [3]. Among structural causes, intracranial neoplasms are a rare cause of secondary parkinsonism [4, 5]. Meningioma is the second most common intracranial tumor and has been shown to demonstrate heterogeneous clinical features according to tumor size and location. Most meningiomas are slowly growing and result in benign clinical outcomes, but only 20% exhibit clinically aggressive features [6]. Although it is rare for intracranial space-occupying lesions to present with a parkinsonism, few cases of parkinsonism due to meningioma have been reported in patients. However, to the best of our knowledge, there are no case reports with intracranial space-occupying lesions resulting in parkinsonism associated with rapid cognitive impairment.
We report herein a patient diagnosed with papillary meningioma who presented with parkinsonism associated with rapidly progressive dementia mimicking diffuse Lewy body disease (DLB).
A 37-year-old woman was admitted with bradykinesia and progressive cognitive impairment. The patient presented with mild slowness of movement on the left side, loss of dexterity in the left hand, and shuffling gait of the left side approximately 18 months in duration. The patient's motor symptoms gradually worsened, and she consequently complained of severe bradykinesia, gait disturbance with falling tendencies and rigidity of the left upper arm on hospital admission. Moreover, the patient noticed cognitive impairment including memory disturbance, visuospatial dysfunction and intermittent visual hallucination for the prior 12 months. The patient reported getting lost on the way home, misidentifying people and talking to herself. She had become restless and wandered at night about 10 days prior to admission. The patient and caregiver complained of progressive and insidious cognitive impairments, such as memory disturbance, visuospatial dysfunction and visual hallucinations, as well as motor dysfunction on admission. The patient also complained of a progressive moderate headache prior to admission approximately 20 days in duration. She had no systemic diseases and no history of head trauma, and was not taking any medication. She had never been exposed to either chemical materials or heavy metals. There was no family history of neurological disorders.
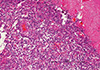
On physical and neurologic examination, she was hypomimic and displayed bilateral bradykinesia and cog-wheel rigidity that were more prominent on left side. However, tremor, myoclonus and ataxia were not found. Her gait was slow and shuffling, associated with short steps without bilateral arm swing during walking. Laboratory tests included a complete blood count, blood chemistry, thyroid function testing, vasculitis markers, HIV antibody, and vitamin B12 and folate levels; all were within normal limits. On detailed neuropsychological testing, the patient scored 23 out of 30 on the Mini-Mental State Examination with a marked impairment in executive function and visuospatial ability (Table 1). Magnetic resonance imaging (MRI) of the brain with gadolinium revealed a large extra-axial cystic tumor in right fronto-temporal region. It showed homogeneous enhancement of the tumor itself and along the margin of the cystic component in T1-weighted images. There was mass effect with compressing lateral ventricle and basal ganglia causing a midline shift to the left (Fig. 1). Single photon emission computed tomography (SPECT) showed increased perfusion of the right frontal region including mass lesion and decreased perfusion of the right basal frontal region and both basal ganglia (Fig. 2). In subsequent biopsy of the lesion, histologic examination revealed that the tumor was composed of sheets of cells arranged in a predominantly papillary pattern with perivascular pseudo-rosettes (Fig. 3). The microscopic findings showed positivity for epithelial membrane antigen (EMA) and vimentin, but negativity for glial fibrillary acidic protein (GFAP). The Ki-67 labeling index was 10.3%. These histologic findings were consistent with papillary meningioma. On the basis of the above-mentioned results, images and pathology, the patient was transferred to the neurosurgery department for operative removal of the papillary meningioma.
The onset of a secondary parkinsonism in patients with intracranial tumor is decidedly rare [4, 5]. However, the possibility of a purely incidental relationship must be taken into consideration in patients with tumor-induced parkinsonism because the removal of an intracranial tumor can lead to cessation of parkinsonian symptoms. Therefore, neuroimaging is an important tool to exclude structural lesions as a possible cause of parkinsonism, even in cases that fulfill necessary clinical criteria for idiopathic PD [3]. Supratentorial tumors located in frontal, parietal, and temporal areas may present with secondary parkinsonism. Pathogenetic mechanisms for those clinical features have been suggested including compression of the basal ganglia and nigrostriatal pathway, and infiltration of the striatum [4, 7].
In the case of our patient, bilateral basal ganglia dysfunction evidenced by the findings of brain perfusion SPECT can explain the associated motor symptoms of parkinsonism. Bradykinesia and rigidity developed on the left side first and then spread to right side. This clinical evolution may result from the mass lesion. The mass compressed the right side basal ganglia first and sequentially compressed the left side as the tumor grew. Therefore, mechanical compression and distortion of the basal ganglia and nigrostriatal pathway, tumor infiltration and impaired tissue perfusion due to tumor edema could be proposed as causative mechanisms [7].
The main presenting symptoms of our young patient consisted of rapidly progressive cognitive decline and parkinsonism. Although dementia usually affects elderly people and results in slowly progressive deterioration, the present patient was young and experienced a rapidly progressive clinical course of cognitive deterioration composed of progressive memory impairment, visual hallucinations, restlessness and agitation, visuospatial dysfunction and deficits in attention. She demonstrated a rapid and severe deterioration of personal daily living capacity. More interestingly, the dementia pattern in the present patient was compatible with probable DLB according to revised criteria for diagnosis of DLB by McKeith et al. [8]. Namely, our patient showed central features (progressive dementia including deficits of attention, executive function and visuospatial ability) plus intermittent visual hallucinations and parkinsonism. To our knowledge, there are no clinical reports about supratentorial tumors presenting as DLB to date.
A basal frontal meningioma can be associated with visual hallucinations, psychiatric symptoms, and other signs of frontal dysfunction such as executive dysfunction [9]. Therefore, we could assume that the present patient also had intermittent visual hallucinations and executive dysfunction due to the fronto-temporal meningioma lesions and associated edema.
Brain disorders with rapidly progressive cognitive impairment include neurodegenerative, infectious, toxic, metabolic, autoimmune, endocrine and neoplastic causes. According to previous reports, neoplastic causes included in the differential diagnosis of rapidly progressive dementia are primary CNS lymphoma, intravascular lymphoma, lymphomatoid granulomatosis, gliomatosis cerebri, and CNS metastasis [3, 7]. On the other hand, the neuropathology of the present patient was papillary meningioma. However, we suspected that the malignant nature of papillary meningioma and associated rapid growth may have contributed the rapid time course of dementia in our patient.
In conclusion, brain tumors are rare causes of secondary parkinsonism associated with cognitive impairment and there are no prior reports of this phenomenon, to our knowledge. We described herein a rare case of a young patient diagnosed with papillary meningioma presenting as parkinsonism and cognitive impairment, similar to DLB. Therefore, we assert that neuroimaging has a place in the assessment of patients with parkinsonism, particularly if symptoms include atypical neurological signs such as cognitive impairment, since early recognition of an intracranial tumor as a cause of parkinsonism
is important to prevent further neurological deficits.
Figures and Tables
Fig. 1
Brain magnetic resonance images show a large mass (size 8×7.2×7 cm) with gadolinium enhancement and cystic change in right frontotemopral region, which compresses the right basal ganglia and lateral ventricle and causes mid-line shift to the left side by mass effect.
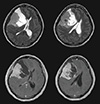
Fig. 2
Brain perfusion SPECT also indicates increased perfusion of the right frontal region including mass lesion and decreased perfusion of right frontal and parieto-occipital regions, and both basal ganglia.
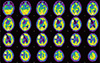
Fig. 3
It shows papillary meningioma with perivascular pseudorosettes, cellular dehiscence, and variable quantities of eosinophilic cytoplasm attached to the dura (×100). The submitted specimen is a dural-based tumor with a bosselated surface. On sections, it is yellow tan with necrosis on the cut surface. On microscopic findings, it has poor cellular cohesion, perivascular pseudorosettes, foci of geographic necrosis, and prominent nucleoli with positive for EMA and vimentine, negative for GFAP, and high Ki-67 index (10.3%). The diagnosis is papillary meningioma, grade III by WHO.
References
1. Hughes AJ, Daniel SE, Lees AJ. Improved accuracy of clinical diagnosis of lewy body parkinson's disease. Neurology. 2001; 57:1497–1499.

2. Tolosa E, Wenning G, Poewe W. The diagnosis of parkinson's disease. Lancet Neurol. 2006; 5:75–86.

3. Bostantjopoulou S, Katsarou Z, Petridis A. Relapsing hemiparkinsonism due to recurrent meningioma. Parkinsonism Relat Disord. 2007; 13:372–374.

4. Krauss JK, Paduch T, Mundinger F, Seeger W. Parkinsonism and rest tremor secondary to supratentorial tumours sparing the basal ganglia. Acta Neurochir (Wien). 1995; 133:22–29.

5. Salvati M, Frati A, Ferrari P, Verrelli C, Artizzu S, Letizia C. Parkinsonian syndrome in a patient with a pterional meningioma: case report and review of the literature. Clin Neurol Neurosurg. 2000; 102:243–245.

7. Adhiyaman V, Meara J. Meningioma presenting as bilateral parkinsonism. Age Ageing. 2003; 32:456–458.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download