Abstract
Background
Behavioral and psychological symptoms of dementia (BPSD) are less well-defined aspects of Alzheimer's disease (AD). We designed this study to explore the followings: 1) the clinical profiles of BPSD 2) the clustered-groups domains of the Korean-Neuropsychiatric Inventory (K-NPI) assessment of BPSD 3) the clinical characteristics of the clustered-groups of BPSD in patients with drug-naïve probable AD.
Methods
Descriptive and cluster analyses of the 12 K-NPI domains were done in 220 patients with drug-naïve probable AD. After clustering these domains, characteristics of these positive symptoms clustered-group of patients were compared with the negative symptoms groups of patients.
Results
The mean Korean-Mini Mental Status Examination (K-MMSE), Clinical Dementia Rating (CDR) scale, and K-NPI scores were 15.0, 1.6, and 14.2, respectively. The CDR and K-MMSE scores correlated with total K-NPI scores, and depression was the most common symptom. According to cluster analysis, five major clusters were identified. Using the associated neuropsychological dysfunctions, characteristics of each group were defined.
Behavioral and psychological symptoms of dementia (BPSD) have been increasingly recognized as an important aspect of Alzheimer's disease (AD). They occur in 80-90% of patients with AD [1, 2], affect the quality of life of both the patients and the caregivers, and strongly determine the patient's lifestyle and management [3].
BPSD is a catch-all term that covers various symptoms, including psychotic, affective, and other symptoms. The BPSD substrate is probably related to anatomical dysfunctions, however, the neurological significances of these complex and bizarre symptoms have rarely studied thus far. In order to study these symptoms, an objective assessment tool is necessary. Among the assessment tools, the Neuropsychiatric Inventory (NPI) is the most widely used instrument for assessing BPSD in dementia patients [4].
However, it is unclear whether NPI domains can be regarded as a single entity, or not. There has not been enough evidence allowing for individualization of each domain. In recent years, several studies have been attempted to identify the grouping of these symptom domains [5-10]. The potential advantages of this approach are that these groups might be more homogenous, thus allow a coherent explanation of the pathogenesis, prognosis and treatment [11].
Most previous studies have been limited by the fact that they included a significant proportion of medicated patients. It means that they were exposed to various psychoactive medications including cholinesterase inhibitors, antipsychotics and antidepressants etc. These medications may possibly have selectively or non-selectively influenced the BPSD domains, and thereby confusing the research results.
AD is now recognized as a growing problem in Korea that has been brought about by the rapid growth of the elderly populations. During the last decade, Korean society has faced many of the same challenges that are related to cognitive and behavioral dysfunctions for patients with AD, as in western societies. Traditionally, family members in Korea assume total responsibility of care giving for the elderly. The long-standing Confucian virtue of filial piety emphasized the provision of personal care for one's parents, and Koreans are generous to one's parents even for aberrant behavior. Though, in recent years, this tradition has been drastically declining in Korea due to industrialization, but there is still a considerable portion of patients with mild to severe AD who visit the dementia clinics with never medicated status. Therefore, we can design a BPSD study in drug-naïve probable AD patients, which will thus overcome the psychoactive medication effects.
Using factor analysis, previous studies have mainly focused on reducing the NPI domains and thus re-defining the sub-syndromes of BPSD. They are less concerned with the neuropsychological characteristics of these groups and the possible implication as a neurological symptom. Reducing and extracting factors may be useful for explaining and understanding complex phenomena, but it may not be ideal for delineating the pathophysiology of these complex phenomena. To understand pathophysiology of BPSD, it is important to extract and re-classify a homogenous group from various BPSD symptom domains, and then find the characteristics of each group.
Although, cluster analysis and factor analysis may be similar, cluster analysis is designed to classify homogenous groups from various menus in a table. Whereas factor analysis try to detect underlying factors, and is often used in data reduction. Considering our aims to explore the homogenous BPSD domains and define the characteristics of these groups, cluster analysis may be more helpful.
The aims of this study were the followings; firstly, to examine the frequency and types of neuropsychiatric symptoms, secondly, to detect clustered-groups in the BPSD domains of the Korean-Neuropsychiatric Inventory (K-NPI), and thirdly, to explore the clinical characteristics of the clustered-group of BPSD in drug-naïve patients with AD. Finally, from these results, we suggested a hypothetical brain mapping for the BPSD sub-groups.
An initial 1,092 patients with dementia were screened from March 2001 to February 2010 at the Hyoja Geriatric Hospital (Fig. 1). Among 1,092 patients with dementia, the subjects of this study were 220 patients with probable AD, who were newly diagnosed and not medicated before visiting hospital. All patients included in this study met National Institute of Neurological and Communicative Disroders and Sroke-Alzheimer's Disease and Related Disorders Association (NINCDS-ADRDA) criteria for probable AD [12]. The patients were drug-naive, except for episodic hypnotics that were taken for sleep disturbances. Patients who were taking psychoactive drugs, including antipsychotics, antidepressants, anticonvulsants, benzodiazepines, and cholinesterase inhibitors, were excluded in this study.
The diagnostic evaluation included complete medical history, physical and neurological evaluation, comprehensive neuropsychological tests, a routine laboratory tests, and brain magnetic resonance imaging (MRI) or computed tomography (CT) scans. The age at onset of the dementia was considered the time of onset of memory disturbances that exceeded the episodic forgetfulness.
The Korean version of the NPI (K-NPI) [13], which is a retrospective informant-based rating scale for the behavioral and psychological symptoms in patients with dementia, was used for the evaluation of the neuropsychiatric symptoms. This scale addresses 12 specific behavioral and psychological symptoms; delusion, hallucination, agitation-aggression, depression, anxiety, euphoria, apathy, disinhibition, irritability, aberrant motor behavior, sleep disturbances, and eating abnormalities. The K-NPI gives a composite score for each domain that is the product of the frequency by severity sub-scores. The total K-NPI score was calculated by adding the 12 composite scores. The test-retest reliability and internal consistency of the K-NPI was assessed in the previous study [13].
In order to assess global dementia severity, the Korean version of the MMSE (K-MMSE) [14], CDR scale [15], and the Clinical Dementia Rating Scale-Sum of Boxes (CDR-SB) were used. A Barthel index [16] for the activities of daily living (ADL) evaluation, and a Geriatric Depression Scale (GDS) [17] for depression were also assessed. Neuropsychological tests representing specific cognitive functions were selected for defining the clinical characteristics of clustered-group. The Seoul verbal learning test (SVLT; Korean version of HPVLT) [18] immediate recall to assess attention, SVLT delayed recall to assess temporal function, semantic word fluency for Controlled Oral Word Association Task (COWAT) to assess frontal lobe function, Stroop Color Naming Test to assess mesial frontal function, calculation for left parietal function, Rey-Osterrieth Complex Figure Test (RCFT) copy for right parietal function were adopted.
First, baseline characteristics of the study subjects were assessed. Second, the frequency and mean total score of the K-NPI domains according to CDR stage were assessed. Third, to identify homogenous K-NPI domains, a hierarchical cluster analysis was performed on all 12 K-NPI domains. Calculation of distance (0-25) based on the measure of similarity (Yule's Q) and the cluster algorithm (average linkage), thus dendrogramm was presented.
Finally, to find the clinical characteristics of these groups, independent T-test was used to compare each group with positive symptoms to that without positive symptoms. Statistical analyses were performed with the SPSS version 18.0 (SPSS, Inc, Chicago, IL, USA).
After complete description of the study was given to the subjects and the caregivers, written informed consent was obtained from the patients or caregivers.
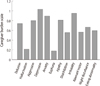
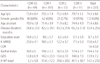
The study included 87 men (40%) and 133 women (60%). The mean age was 74.5±8.0 years. The average K-MMSE and CDR scores were 15.0±6.7 and 1.6±0.8 respectively, indicating that subjects with mild to moderate AD were mainly recruited (Table 1). Depression was the most common symptom for the patients and the most troublesome symptom for the caregivers, being present in 53.6% of the patients (Table 2). Disinhibition and irritability were also very common, occurring in about 40% of the patients. The rarest symptoms were euphoria and hallucinations (7%) (Table 2, Fig. 2). The total K-NPI scores increased significantly according to CDR stage (Fig. 3).
A hierarchical cluster analysis was performed to further delineate the structure of the variable K-NPI domains. Scores for each of the K-NPI domains for the 220 patients were plotted in a geometric space according to their average linkage. The hierarchy results from clustering the data of the 220 patients are shown in Fig. 4. On this dendrogram, closely inter-correlated K-NPI domains were joined at an early stage in the analysis (at the left of the dendrogram), and less closely inter-correlated domains were joined at later stages (at the right of the dendrogram). Five major clusters were found in the analysis, which were detected by a marked discontinuity or a "step" in the hierarchy.
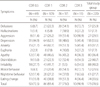
The results of the cluster analysis of the total group of patients are shown in Table 3. Group 1 included a wide range of symptom domains (aggression, aberrant motor behavior, delusion, irritability, disinhibition, and hallucination), contrasting Group 2 (sleep disturbance) and Group 4 (eating abnormality) which included a single symptom domain.
All clustered-groups were related to global cognitive dysfunctions, and specific cognitive dysfunctions were found in each group. Group 1 showed significantly lower scores in SVLT immediate recall, SVLT delayed recall, and COWAT tests. Group 2 showered significantly lower scores in RCFT copy. Group 3 showed significantly lower scores in COWAT test. Group 4 showed significantly lower scores in SVLT immediate recall, RCFT copy, and Group 5 showed significantly lower scores in COWAT and RCFT copy (Table 4).
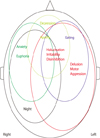
If additionally selected neuropsychological test dysfunctions for a specific clustered group means that a selected anatomical disturbance represents this dysfunction, hypothetical mapping for each group can be drawn (Fig. 5). The same color represents the same group, the size of the circle means the degree of global dysfunction, and the text location indicates right, left, or anterior of the brain.
Since development of NPI, the lack of consistent or weak relationships between cognitive and neuropsychiatric abnormalities was suggested as evidence that neuropsychiatric abnormalities are not reactive to cognitive impairment [19-21], but rather, are epiphenomena of AD. However, a recent study using untreated patients with AD showed that disease severity was correlated with neuropsychiatric abnormalities except the affective syndromes [10]. Moreover, in our study, K-NPI scores were very closely related to general and specific cognitive functions in all groups, including the depression and apathy group (e.g. affective symptom group). These finding suggested that, if we exclude medication effects, cognitive dysfunction and BPSD in AD are not separate phenomena but may be connected in some way.
To our knowledge, this is the first study to explore the clustered groups of NPI domains in all stages of patients with drug-naïve AD and to define the clinical characteristics of the clustered-groups. Previous studies have mainly focused on reducing NPI domain factors and thus defining sub-syndromes of AD [5-10]. However, our study focused on extracting homogenous groups from numerous NPI domains. Using this, we tried to define the characteristic of each group. A recent study, that recruited only untreated AD patients as we did, also showed five major groups, but they were somewhat dissimilar to our study [10]. This raises the question of why there was a discrepancy concerning the group domains between this study and ours. Both studies were free from the psychoactive medications, so medication effects can be excluded. The difference may have originated from ethnic difference or inclusion differences because our study included more severe cases and educated patients. Differences in the adopted statistical methods also may be responsible for this. Ethnically, in Asia, depression is the most common symptoms whereas apathy is more frequently experienced in Western countries [9, 22]. Whether these ethnical differences influence neuropsychiatric symptom architecture is controversial.
Cluster analysis revealed five major groups that comprised a wide range of symptom complexes (Group 1) to single symptom domain clusters (Group 2 and Group 4). Delusions, aggression, aberrant motor behavior and, depression and apathy were highly inter-correlated (homogenous). Anxiety and euphoria were moderately inter-correlated and were separated from other symptom domains (Fig. 4).
These clusters are somewhat different compared to the previous studies which usually sub-grouped as psychotic, affective and other symptom domains [5-10]. One possible explanation for these differences is that most previous studies included a significant portion of medicated patients. If the side effects of a certain medication are sleep disturbance and appetite loss, these subjects showed significantly higher prevalence of these symptoms, and they would be masked as a homogenous cluster. Numerous psychoactive drugs are prone to affect a wide range of neuropsychiatric symptoms. Therefore, to explicitly define NPI symptoms, observations in drug-naïve patients are essential.
Another reason for the difference in BPSD clustering may be the selection bias. A hospital-based study may include patients with more severe symptoms and, in contrast, a study of drug-naïve AD patients may be prone to the recruitment of patients with more mild dementia. Therefore, our study included subjects with mixed and complex stages of dementia.
Although, hallucinations are classified in the same group as delusions, inter-correlation was looser than with other symptoms (aberrant motor behavior and aggression, Fig. 4). Considering aberrant motor behavior and aggression are highly correlated with delusions, these symptoms may be interpreted as being flip side of delusions, and the management of these symptoms may be in line with delusion treatment.
Patients with Group 1 scored significantly lower in almost neuropsychological tests, and this means that this group had complex and multiple substrate pathophysiologies. Patients in Group 2 scored significantly lower in RCFT copy, suggesting right hemispheric dysfunction with general cognitive dysfunction. Patients with depression and apathy (Group 3) scored significantly lower in SVLT immediate recall and COWAT, indicating attention and frontal dysfunction. Although CDR-SB was also significantly lower in this group, other general cognitive tests, e.g. K-MMSE, CDR, were not significantly low compared to negative symptom patients. This means that this group was somewhat loosely related with general cognitive functions compared to other clustered-groups, and frontal dysfunction may be associated. Other point is that apathy and depression are very closely related to each other. Some studies using the NPI have not been able to distinguish two syndromes of depression and apathy [23, 24], and other studies separate apathy from depression, implying an independent factor [9, 20, 25, 26]. The question of whether depression and apathy are distinct syndromes has been the topic of considerable discussion, however, it remains still controversial. Considering that co-prevalence and specific focal cognitive functions were hampered in both symptoms, these symptoms may not be an independent entity, but may be related to each other in some ways. Although we could not ascertain whether this relation was primary or secondary, it may represent frontal dysfunction in drug-naïve patients with AD.
The patients with eating abnormalities (Group 4) scored low in SVLT immediate recall and RCFT copy score and the patients with anxiety and euphoria (Group 5) scored lower in COWAT and RCFT copy. Group 4 and 5 suggested right hemispherical dysfunction. Co-prevalence of euphoria and anxiety may be somewhat embarrassing. However, considering that manic episodes and anxiety were commonly associated in epidemiological study, and these systems share common norepinephrine, dopamine and serotonin systems, the pathophysiology of these overlapping symptomatology in AD may be similar to bipolar disorders [27].
At least two hypotheses can be drawn to explain the co-occurrence of behavioral symptoms. First, some behavioral symptoms might be secondary to others. Alternatively, behavioral symptoms might share common anatomical and pathophysiological substrates. In the same way, the co-occurrence of behavioral symptoms and specific cognitive dysfunctions can be explained.
If we hypothesize that clustered-groups showing a significant deficit in a neuropsychological test representing a specific lobar function may share common anatomy, we can hypothetically (and roughly) map each cluster of BPSD. Using this hypothesis, we may understand the fuzzy and unclear symptomatology of BPSD related to focal neurologically relevant symptomatology.
This study had some limitations. First, the sample size is relatively small for the number of factors analyzed. Second, although our study included more severe patients than previous studies, still they were mostly in the mild to moderate stages of dementia, and so this study was mostly biased towards mild cases. Third, attempts in hypothetical mapping may have some limitations and have less clinical relevance, because the anatomical localization of BPSD symptoms accompanying region-specific neuropsychological test dysfunction may not always result from the same anatomical disruption. Finally, our study is a hospital-based study, and so it may not represent a real community.
In conclusion, this is the first study to identify the cluster group for NPI domains and explore the possible anatomical substrates for these groups in drug-naïve AD patients. Although this study has some limitations, these results may clarify the complex and bizarre neuropsychiatric symptoms as more neuropsychologically relevant symptoms. In a future study, larger sample size of drug-naïve patients and more specific tests that represent specific anatomical substrates may provide a better understanding of the behavioral and psychiatric symptoms of dementia.
Figures and Tables
 | Fig. 3Mean K-NPI total score according to CDR staging. Except for the difference between CDR 2 and 3 (p=0.18), all groups are significantly different (p<0.01). K-NPI, Korean Neuropsyhicatirc Inventory; CDR, Clinical Dementia Rating Scale. |
 | Fig. 4Dendrogram for clustered variable of K-NPI domains. Closely intercorrelated variables are joined at an early stage in the analysis (left of the dendrogram) and less closely intercorrelated variables are joined at a later stage in the analysis (right of the dendrogram). Fs, frequency x severity; K-NPI, Korean Neuropsyhicatirc Inventory. |
Table 4
Clinical characteristics of clustered group

Each positive group was compared with each negative group, i.e. apathy and depression-positive group was compared to apathy and depression negative-group. If one symptom was positive and other symptom was negative, this data was not compared. Statistical significance means that positive symptom groups had a lower score than the negative symptom groups.
*p value<0.05; †p value<0.01 (One-Way ANOVA test was used).
MMSE, Korean Mini-Mental State Examination; CDR, Clinical Dementia Rating Scale; CDR-SB, Clinical Dementia Rating Scale-Sum of Boxes; GDS, Geriatric Depression Scale.
References
1. Aalten P, de Vugt ME, Lousberg R, Korten E, Jaspers N, Senden B, et al. Behavioral problems in dementia: a factor analysis of the neuropsychiatric inventory. Dement Geriatr Cogn Disord. 2003. 15:99–105.

2. Steinberg M, Tschanz JT, Corcoran C, Steffens DC, Norton MC, Lyketsos CG, et al. The persistence of neuropsychiatric symptoms in dementia: the Cache County Study. Int J Geriatr Psychiatry. 2004. 19:19–26.

3. Matsui T, Nakaaki S, Murata Y, Sato J, Shinagawa Y, Tatsumi H, et al. Determinants of the quality of life in Alzheimer's disease patients as assessed by the Japanese version of the Quality of Life-Alzheimer's disease scale. Dement Geriatr Cogn Disord. 2006. 21:182–191.

4. Cummings JL, Mega M, Gray K, Rosenberg-Thompson S, Carusi DA, Gornbein J. The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology. 1994. 44:2308–2314.

5. Frisoni GB, Rozzini L, Gozzetti A, Binetti G, Zanetti O, Bianchetti A, et al. Behavioral syndromes in Alzheimer's disease: description and correlates. Dement Geriatr Cogn Disord. 1999. 10:130–138.

6. Lyketsos CG, Breitner JC, Rabins PV. An evidence-based proposal for the classification of neuropsychiatric disturbance in Alzheimer's disease. Int J Geriatr Psychiatry. 2001. 16:1037–1342.

7. Mirakhur A, Craig D, Hart DJ, McIlroy SP, Passmore AP. Behavioural and psychological syndromes in Alzheimer's disease. Int J Geriatr Psychiatry. 2004. 19:1035–1039.

8. Aalten P, Verhey F, Boziki M, Bullock R, Byrne EJ, Camus V, et al. Neuropsychiatric syndromes in dementia: results from the European Alzheimer Disease Consortium (EADC). Part 1. Dement Geriatr Cogn Disord. 2007. 24:457–463.
9. Kang HS, Ahn IS, Kim JH, Kim DK. Neuropsychiatric symptoms in Korean patients with Alzheimer's disease: exploratory factor analysis and confirmatory factor analysis of the neuropsychiatric inventory. Dement Geriatr Cogn Disord. 2010. 29:82–87.

10. Spalletta G, Musicco M, Padovani A, Rozzini L, Perri R, Fadda L, et al. Neuropsychiatric symptoms and syndromes in a large cohort of newly diagnosed, untreated patients with Alzheimer disease. Am J Geriatr Psychiatry. 2010. 18:1026–1035.

11. Robert PH, Verhey FR, Byrne EJ, Hurt C, De Deyn PP, Nobili F, et al. Grouping for behavioral and psychological symptoms in dementia: clinical and biological aspects. Consensus paper of the European Alzheimer disease consortium. Eur Psychiatry. 2005. 20:490–496.

12. McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM. Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease. Neurology. 1984. 34:939–944.

13. Choi SH, Na DL, Kwon HM, Yoon SJ, Jeong JH, Ha CK. The Korean version of the Neuropsychiatric Inventory: a scoring tool for neuropsychiatric disturbance in dementia patients. J Korean Med Sci. 2000. 15:609–615.

14. Kang Y, Na DL, Hahn S. A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J Korean Neurol Assoc. 1997. 15:300–308.
15. Hughes CP, Berg L, Danziger WL, Coben LA, Martin RL. A new clinical scale for the staging of dementia. Br J Psychiatry. 1982. 140:566–572.

16. Mahoney FT, Barthel DW. Functional evaluation: Barthel index. Md State Med J. 1965. 14:61–65.
17. Jung IK, Kwak DI, Shin DK, Lee MS, Lee HS, Kim JY. A Reliability and Validity Study of Geriatric Depression Scale. J Korean Neuropsychiatr Assoc. 1997. 36:103–111.
18. Kang Y. Samsung Neuropsychological Screening Battery. Current research in dementia. 1998. Seoul: The Korean Dementia Association;99–107.
19. Cummings JL. The Neuropsychiatric Inventory: assessing psychopathology in dementia patients. Neurology. 1997. 48:S10–S16.

20. Spalletta G, Baldinetti F, Buccione I, Fadda L, Perri R, Scalmana S, et al. Cognition and behaviour are independent and heterogeneous dimensions in Alzheimer's disease. J Neurol. 2004. 251:688–695.

21. McKeith I, Cummings J. Behavioural changes and psychological symptoms in dementia disorders. Lancet Neurol. 2005. 4:735–742.

22. Fuh JL, Lam L, Hirono N, Senanarong V, Cummings JL. Neuropsychiatric inventory workshop: behavioral and psychologic symptoms of dementia in Asia. Alzheimer Dis Assoc Disord. 2006. 20:314–317.

23. Frisoni GB, Rozzini L, Gozzetti A, Binetti G, Zanetti O, Bianchetti A, et al. Behavioral syndromes in Alzheimer's disease: description and correlates. Dement Geriatr Cogn Disord. 1999. 10:130–138.

24. Mirakhur A, Craig D, Hart DJ, McLlroy SP, Passmore AP. Behavioural and psychological syndromes in Alzheimer's disease. Int J Geriatr Psychiatry. 2004. 19:1035–1039.

25. Marin RS, Firinciogullari S, Biedrzycki RC. The sources of convergence between measures of apathy and depression. J Affect Disord. 1993. 28:117–124.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download