Abstract
Bioactivity-guided fractionation of MeOH extract of the dried fruits of Amomum tsao-ko led to isolation of nine compounds (1 – 9). Their structures were elucidated by spectroscopic methods including extensive 1D and 2D-NMR, as alpinetin (1), naringenin-5-O-methyl ether (2), naringenin (3), hesperetin (4), 2′,4′,6′-trihydroxy-4-methoxy chalcone (5), tsaokoin (6), boesenbergin B (7), 4-hydroxyboesenbergin B (8), and tsaokoarylone (9). Of these, compound 8 was isolated from a natural source for the first time, which was previously reported as a synthetic product. The isolated compounds (1 – 9) were tested for their inhibitory effects on LPS-induced nitric oxide production in RAW 264.7 macrophages. Among them, three chalcone derivatives (compounds 5, 7, and 8) and a diarylheptanoid (compound 9) exhibited significant inhibitory activity on the NO production with IC50 values ranging from 10.9 to 22.5 µM.
Amomum tsao-ko Crevost et Lemaire (Zingiberaceae) is a medicinal plant that is widely used in Korea, China, and Japan, for the treatment of nausea, abdominal distension, diarrhea, and malaria.1 Previous phytochemical investigations on this plant reported the presence of monoterpenes, diarylheptanoids, flavonoids, and phenolic compounds.2345 In the course of a research program for the isolation of anti-inflammatory constituents from medicinal plants, a MeOH extract of the fruits of A. tsao-ko showed significant inhibitory effects on LPS-induced nitric oxide (NO) production (IC50 value of 23.5 µg/mL). Subsequently, the fruits of A. tsao-ko were extracted with MeOH, and the extract was suspended in water and then partitioned with n-hexane, CH2Cl2, EtOAc, and n-BuOH. Bioassay-guided fractionation of the CH2Cl2- and ethyl acetatesoluble fraction led to the isolation of 9 known compounds (1 – 9). The structures of isolates were determined by interpretation on their 1D and 2D NMR spectroscopic data as well as mass spectrometry, and by comparison of the spectroscopic data with literature. The isolated compounds (1 – 9) were tested for their inhibitory effects on LPS-induced nitric oxide production in RAW 264.7 macrophages. Herein, the isolation, structural determination, and the nitric oxide inhibitory effects of the isolated compounds are reported.
Optical rotations were measured on a JASCO DIP-1000 polarimeter. UV spectra were recorded on a JASCO UV-550 spectrophotometer, and IR spectra were measured on a JASCO FT-IR 4100 spectrometer. NMR spectra were recorded on a Bruker AVANCE 400 and 500MHz spectrometer. ESIMS and HRESIMS were obtained on LCQ Fleet and maXis 4G mass spectrometers, respectively. Column chromatography was performed on silica gel (Merck, 70 – 230 and 230 – 400 mesh) and Lichroprep RP-18 (Merck, 40 – 63 µm). MPLC was performed on a Biotage Isolera Prime chromatography system. Preparative HPLC was performed using Waters HPLC system equipped with two Waters 515 pumps with a 2996 photodiode-array detector using a YMC J'sphere ODS-H80 (4 µm, 150 × 20 mm, i.d., flow rate 6 mL/min). TLC was performed using precoated silica gel 60 F254 (0.25 mm, Merck) plates, and spots were detected by a 10% vanillin-H2SO4 in water spray reagent.
The dried fruits of A. tsao-ko (3 kg) were purchased from Kyungdong herbal market in Seoul, Korea, in May 2014. A voucher specimen (CBNU-2014-05-AT) was authenticated by B. Y. Hwang and deposited at the Herbarium of the College of Pharmacy, Chungbuk National University, Korea.
The dried fruits of A. tsao-ko (3 kg) were extracted with MeOH (3 × 5 L) at room temperature and filtered. The filtrates were evaporated under reduced pressure to give a MeOH extract (132 g), which was suspended in water and then successively partitioned with n-hexane, CH2Cl2, EtOAc, and n-BuOH. The CH2Cl2-soluble fraction (92 g) was chromatographed on a silica gel column (70 – 230 mesh), eluting with a gradient solvent system consisting of n-hexane/CH2Cl2/MeOH (4:1:0 to 0:1:1) yielded 8 fractions (ATC1 – ATC8). Fraction ATC 6 (31.9 g) was further separated on a silica gel column (230 – 400 mesh) with a gradient solvent system of CH2Cl2/MeOH (40:1 to 1:1) as the eluent to give six sub-fractions (ATC6-1 – ATC6-6). Fraction ATC 6-2 (2.7 g) was purified by preparative HPLC (MeCN-H2O, 30:70 to 60:40) to afford compound 1 (1.7 mg). Fraction ATC 6-4 (2.2 g) was further purified by preparative HPLC (MeCN-H2O, 20:80 to 50:50) to afford compound 2 (4.2 mg). The EtOAc-soluble fraction (21 g) was chromatographed on a silica gel column (230 – 400 mesh) and eluted with n-hexane/EtOAc gradient system (20:1 to 1:2) to afford 10 fractions (ATE1 – ATE10). Fraction ATE 2 (324 mg) was separated by MPLC with Lichroprep RP-18 column with MeOH-H2O gradient system (30:70 to 100:0) to give compound 7 (14.0 mg). Fraction ATE 7 (1.7 g) was separated on a Sephadex LH-20 column and eluted with MeOH-H2O (80:20) to give compound 8 (16.0 mg). Fraction ATE 8 (2.2 g) was further separated by MPLC with Lichroprep RP-18 column and eluted with MeOH-H2O (5:95 to 100:0) to give 12 fractions (ATE 8-1 – ATE 8-12). Fraction ATE 8-5 was purified by preparative HPLC (MeCN-H2O, 35:65, isocratic) to afford compound 6 (13.0 mg). Fraction ATE 8-7 was further purified by preparative HPLC (MeCN-H2O, 35:65, isocratic) to afford compounds 3 (1.2 mg) and 4 (0.8 mg). Fraction ATE 8-8 was further separated by preparative HPLC (MeCN-H2O, 40:60 to 45:55) to afford compounds 5 (2.2 mg) and 9 (1.5 mg).
Colorless needles, [α]D25: −3.1 (c=0.1 MeOH); UV (MeOH) λmax nm: 209, 286, 320; IR (KBr) νmax cm−1: 3520, 3200, 3075, 1636, 1610, 1580, 1474, 1315, 1274, 1110; ESIMS m/z: 271.17 [M+H]+; 1H NMR (DMSO-d6, 500 MHz): δ 7.36~7.5 (5H, m, H-2′, 3′, 4′, 5′, 6′), 6.07 (1H, d, J = 2.0 Hz, H-6), 6.00 (1H, d, J = 2.0 Hz, H-8), 5.5 (1H, dd, J = 3.0, 12.5 Hz, H-2), 3.74 (3H, s, OCH3), 3.01 (1H, dd, J = 12.5, 16.5 Hz, H-3α), 2.65 (1H, dd, J = 3.0, 16.5 Hz, H-3β); 13C NMR (DMSO-d6, 125 MHz): δ 187.7 (C-4), 165.1 (C-7), 164.5 (C-9), 162.6 (C-5), 139.7 (C-1′), 129.0 (C-3′, 5′), 128.8 (C-4′), 126.9 (C-2′, 6′), 104.8 (C-10), 96.2 (C-6), 93.9 (C-8), 78.4 (C-2), 56.1 (OCH3), 45.3 (C-3).
Colorless oil, [α]D25: −14.0 (c=0.01, CHCl3); UV (MeOH) λmax nm: 226, 283; IR (KBr) νmax cm−1: 3366, 1642; ESIMS m/z: 287.17 [M+H]+; 1H NMR (DMSO-d6, 400 MHz): δ 7.3 (2H, d, J = 8.8 Hz, H-2′, 6′), 6.79 (2H, d, J = 8.8 Hz, H-3′, 5′), 6.04 (1H, d, J = 2.0 Hz, H-6), 5.93 (1H, d, J = 2.0 Hz, H-8), 5.34 (1H, dd, J = 2.8, 12.4 Hz, H-2), 3.73 (3H, s, OCH3), 2.98 (1H, dd, J = 12.4, 16.3 Hz, H-3α), 2.48 (1H, dd, J = 3.1, 16.4 Hz, H-3β); 13C NMR (DMSO-d6, 100 MHz): δ 188.2 (C-4), 164.9 (C-5), 164.7 (C-7), 162.7 (C-9), 158.0 (C-4′), 131.0 (C-1′), 128.6 (C-2′, 6′), 115.6 (C-3′, 5′), 104.9 (C-10), 96.1 (C-6), 93.7 (C-8), 78.5 (C-2), 56.1 (OCH3), 45.2 (C-3).
Colorless gum, [α]D25: −17.5 (c=0.1 , MeOH); UV (MeOH) λmax nm: 248, 293; IR (KBr) νmax cm−1: 3112, 1600, 1517, 1493, 1459, 1246, 1178, 1154; ESIMS m/z: 273.10 [M+H]+; 1H NMR (CD3OD, 500 MHz): δ 7.32 (2H, d, J = 8.6 Hz, H-2′, 6′), 6.82 (2H, d, J = 8.6 Hz, H-3′, 5′), 5.89 (1H, d, J = 2.1 Hz, H-6), 5.88 (1H, d, J = 2.1 Hz, H-8), 5.36 (1H, dd, J = 3.0, 13.0 Hz, H-2), 3.14 (1H, dd, J = 13.0, 17.0 Hz, H-3α), 2.71 (1H, dd, J = 3.0, 17.0 Hz, H-3β); 13C NMR (CD3OD, 125 MHz): δ 197.8 (C-4), 168.6 (C-7), 165.5 (C-5), 164.9 (C-9), 159.1 (C-4′), 131.1 (C-1′), 129.0 (C-2′, 6′), 116.3 (C-3′, 5′), 103.3 (C-10), 97.1 (C-6), 96.2 (C-8), 80.5 (C-2), 44.1 (C-3).
Colorless gum, [α]D25: −10.6 (c=0.1, MeOH); UV (MeOH) λmax nm: 288, 325; IR (KBr) νmax cm−1: 3499, 3433, 1635, 1586, 1514, 1473, 1442; ESIMS m/z: 303.1 [M+H]+; 1H NMR (CD3OD, 500 MHz): δ 7.08 (1H, d, J = 2.0 Hz, H-2′), 6.93 (1H, dd, J = 2.0, 8.2 Hz, H-6′), 6.82 (1H, d, J = 8.2 Hz, H-5′), 5.91 (1H, d, J = 2.1 Hz, H-6), 5.88 (1H, d, J = 2.1 Hz, H-8), 5.36 (1H, dd, J = 3.0, 13.0 Hz, H-2), 3.88 (3H, s, OCH3), 3.17 (1H, dd, J = 13.0, 17.0 Hz, H-3α), 2.73 (1H, dd, J = 3.0, 17.0 Hz, H-3β); 13C NMR (CD3OD, 125 MHz): δ 197.8 (C-4), 167.2 (C-5), 165.5 (C-7), 164.9 (C-9), 149.2 (C-3′), 148.2 (C-4′), 131.8 (C-1′), 120.6 (C-2′), 116.2 (C-5′), 111.3 (C-6′), 103.4 (C-10), 97.2 (C-6), 96.3 (C-8), 80.5 (C-2), 56.5 (OCH3), 44.2 (C-3).
Colorless gum, UV (MeOH) λmax nm: 296, 360; IR (KBr) νmax cm−1: 3327, 1646; ESIMS m/z: 287.0 [M+H]+; 1H NMR (CD3OD, 400 MHz): δ 7.8 (1H, d, J = 15.5 Hz, H-α), 7.70 (1H, d, J = 15.5 Hz, H-β), 7.52 (2H, d, J = 8.6 Hz, H-2, 6), 6.84 (2H, d, J = 8.6 Hz, H-3, 5), 6.01 (1H, d, J = 2.2 Hz, H-3′), 5.93 (1H, d, J = 2.2 Hz, H-5′), 3.93 (3H, s, OCH3); 13C NMR (CD3OD, 100 MHz): δ 194.0 (C=O), 168.6 (C-2′), 166.4 (C-4′), 164.7 (C-6′), 161.2 (C-4), 143.8 (C-β), 131.3 (C-3, 5), 130.3 (C-1), 128.4 (C-α), 116.8 (C-2, 6), 106.6 (C-1′), 97.1 (C-3′), 92.4 (C-5′), 56.4 (OCH3).
Pale brown oil; UV (MeOH) λmax nm; 228, 231; IR (KBr) νmax cm−1: 3396, 2868, 2728, 1683, 1634, 1428, 1379, 1181, 1062, 1026, 963, 903, 740; ESIMS m/z: 167.08 [M+H]+; 1H NMR (CDCl3, 400 MHz): δ 9.41 (1H, s, H-10), 6.66 (1H, ddd, J = 5.6, 2.8, 1.2 Hz, H-3), 4.07 (1H, dt, J = 9.2, 5.0 Hz, H-5), 2.98 (H, m, H-1), 2.48 (1H, dddt, J = 18.8, 5.6, 5.0, 1.4 Hz, H-4β), 2.42 (1H, m, H-6), 2.38 (1H, ddt, J = 18.8, 9.2, 2.8 Hz, H-4α), 2.02 (1H, m, H-9β), 1.73 (1H, m, H-7α), 1.54 (1H, m, H-7β), 1.50 (2H, m, H-8). 1.40 (1H, m, H-9α); 13C NMR (CD3OD, 125 MHz): δ 194.5 (C-10), 148.1 (C-3), 144.7 (C-2), 67.4 (C-5), 42.8 (C-6), 37.1 (C-1), 31.9 (C-9), 30.3 (C-4), 24.5 (C-7), 24.2 (C-8).
Pale yellow amorphous solid, [α]D25: −12.1 (c=1.10, CHCl3); UV (MeOH) λmax nm; 233, 304, 349; IR (KBr) νmax cm−1: 3500, 1630, 1450, 1380, 1100, 900; ESIMS m/z: 405.08 [M+H]+; 1H NMR (CDCl3, 400 MHz): δ 14.5 (1H, s, 2′-OH), 7.90 (1H, d, J = 15.6 Hz, H-α), 7.79 (1H, d, J = 15.6 Hz, H-β), 7.61 (2H, m, H-2, 6), 7.41 (1H, m, H-4), 7.38 (2H, m, H-3, 5), 6.74 (1H, d, J = 10.0 Hz, H-1″), 5.91 (1H, s, H-5′), 5.42 (1H, d, J = 10.0 Hz, H-2″), 5.12 (1H, m, H-7″), 3.92 (3H, s, 6′-OCH3), 2.13 (2H, m, H-6″), 1.81 (2H, m, H-4″), 1.66 (3H, s, CH3-9″), 1.57 (3H, s, CH3-10″), 1.42 (3H, s, CH3- 5″); 13C NMR (CDCl3, 100 MHz): δ 192.6 (C=O), 162.6 (C-6′), 162.5 (C-2′), 160.8 (C-4′), 142.1 (C-β), 135.6 (C-1), 131.9 (C-8″), 130.0 (C-4), 128.9 (C-2, 6), 128.3 (C-3, 5), 127.6 (C-α), 124.2 (C-2″), 123.8 (C-7″), 116.5 (C-1″), 105.9 (C-1′), 102.8 (C-3′), 91.3 (C-5′), 80.8 (C-3″), 55.9 (6′-OCH3), 41.7 (C-4″), 27.2 (C-5″), 25.7 (C-9″), 22.7 (C-6″), 17.7 (C-10″).
Yellowish oil, [α]D25: −6.9 (c=0.1, MeOH); UV (MeOH) λmax nm; 233, 304, 349; IR (KBr) νmax cm−1: 3497, 1625, 1430, 1380, 1100; ESIMS m/z: 421.17 [M+H]+; HRESIMS m/z: 421.2009 [M+H]+ (cacld for C26H29O5, 421.2010); 1H NMR (CDCl3, 400 MHz) and 13C NMR (CDCl3, 100 MHz), see Table 1.
Colorless oil, [α]D25: +8.3 (c=0.12, MeOH); UV (MeOH) λmax nm; 224, 279; IR (KBr) νmax cm−1: 3355, 1705, 1614, 1515, 1365, 1242, 830; ESIMS m/z: 325.14 [M+H]+; 1H NMR (CD3OD, 500 MHz): δ 7.41 (1H, dd, J = 15.4, 10.6 Hz, H-5), 7.13 (1H, d, J = 2.0 Hz, H-2″), 7.04 (2H, d, J = 8.6 Hz, H-2′, 6′), 7.01 (1H, dd, J = 8.4, 2.0 Hz, H-6″), 6.98 (1H, d, J = 15.4 Hz, H-6), 6.88 (1H, dd, J = 15.4, 10.6 Hz, H-4′), 6.78 (1H, d, J = 8.2 Hz, H-5″), 6.69 (2H, d, J = 8.6 Hz, H-3″, 5″), 6.28 (1H, d, J = 15.4 Hz, H-4), 3.89 (3H, s, 3″-OCH3), 2.91 (2H, m, H-1), 2.84 (2H, m, H-2); 13C NMR (CD3OD, 125 MHz): δ 202.7 (C-3), 156.6 (C-4′), 149.5 (C-4″), 149.3 (C-3″), 146.2 (C-5), 143.9 (C-7), 133.3 (C-1′), 130.4 (C-2′, 6′), 129.8 (C-1″), 129.0 (C-4), 125.2 (C-6), 123.0 (C-6″), 116.5 (C-5″), 116.2 (C-3′, 5′), 111.0 (C-2″), 56.4 (3″-OCH3), 43.2 (C-2), 30.9 (C-1).
RAW 264.7 cells were seeded into 96-well culture plates at 2 × 106 cells/mL, and stimulated with 1 µg/mL of LPS in the presence or absence of compounds. After incubation at 37℃ for 24 h, nitrite concentration in culture medium was measured with the Griess reagent [containing equal volumes of 2% (w/v) sulfanilamide in 5% (w/v) phosphoric acid and 0.2% (w/v) of N-(1-naphthyl) ethylenediamine solution] as an indicator of NO production. The absorbance was measured at 550 nm against a calibration curve with sodium nitrite standards. Cell viability of the remaining cells was determined by MTT (Sigma Chemical Co., St. Louis, MO)-based colorimetric assay.
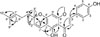
In the continuing search for the plant-derived inhibitors of nitric oxide (NO) production, repeated column chromatographic separation of CH2Cl2 and EtOAc-soluble fractions resulted in the isolation of nine compounds (1 – 9) (Fig. 1). Their structures were identified as alpinetin (1),6 naringenin-5-O-methyl ether (2),7 naringenin (3),8 hesperetin (4),9 2′,4′,6′-trihydroxy-4-methoxy chalcone (5),10 tsaokoin (6),11 boesenbergin B (7),12 4-hydroxyboesenbergin B (8),13 and tsaokoarylone (9).14 Among them, compound 8 was previously reported but only as a synthetic product. The full NMR assignments of 8 were determined by analysis of 2D NMR such as HMQC and HMBC spectroscopic data.
Compound 8 was obtained as yellowish oil. The molecular formula of 8 was determined to be C26H28O5 by the 13C NMR data and an HRESIMS ion peak at m/z 421.2009 [M+H]+ (calcd 421.2010), requiring thirteen indices of hydrogen deficiency. The 1H NMR spectrum of 8 indicated the presence of a pair of characteristic trans olefinic protons at the C-α and C-β positions of a chalcone skeleton [δH 7.79 (1H, d, J = 15.7 Hz) and 7.76 (1H, d, J = 15.7 Hz)], and revealed presence of p-disubstituted benzene ring [δH 7.51 (2H, d, J = 8.6 Hz) and 6.87 (2H, d, J = 8.6 Hz)], an aromatic singlet proton [δH 5.91 (1H, s)], a hydrogen-bonded hydroxy group [δH 14.70 (s)], and a methoxy group [δH 3.91 (3H, s)]. The remaining signals of the 1H NMR spectrum of 8 exhibited the presence of 2-methyl-2-(4-methylpent-3-enyl) 2H-pyran ring [δH 6.74 (1H, d, J = 10.0 Hz), 5.42 (1H, d, J = 10.0 Hz), 5.11 (1H, m), 2.13 (2H, dt, J = 7.0, 7.5 Hz), 1.81 (1H, m), 1.69 (1H, m), and 1.66, 1.58, 1.42 (each 3H, s)]. The 13C-NMR data showed 26 carbon signals comprising a carbonyl carbon, twelve aromatic carbons, six olefinic carbons, a quaternary carbon, two methylene carbons, a methoxy carbon, and three methyl carbons (Table 1). The location of the methoxy group at C-6′ position was confirmed by the HMBC correlation from the methoxy singlet at δH 3.91 (3H, s) to C-6′ at δC 162.6 (Fig. 2). In addition, the HMBC correlations from δH 6.74 (H-1″) to δC 162.5 (C-2′), 160.8 (C-4′), and 80.7 (C-3″), as well as the correlations from δH 5.42 (H-2″) to δC 102.8 (C-3′), 80.7 (C-3″), 41.7 (C-4″), and 27.2 (C-5″) indicated that 2-methyl-2-(4-methylpent-3-enyl) 2H-pyran ring was attached at C-3′ and C-4′ (Fig. 2). The data of 8 are similar to those of 7, boesenbergin B, except for the presence of hydroxyl group at C-4. Thus, the structure of 8 was defined as (2E)-1-(5-hydroxy-7-methoxy-2-methyl-2-(4-methylpent-3-enyl)-2H-chromen-6-yl)-3-(4-hydroxyphenyl)prop-2-en-1-one, and named 4-hydroxyboesenbergin B.
NO plays an important role in the inflammatory process and therefore, inhibitors of NO production may be potential anti-inflammatory agents.15 All isolates were tested for their inhibitory effects on LPS-induced NO production in RAW 264.7 cells with aminoguanidine as the positive control (IC50 value: 21.4 µM) (Table 2). Compounds 5, 7, and 8, which possesses a chalcone skeleton, showed significant inhibitory effects on the NO production with IC50 values ranging from 10.9 to 22.5 µM. Tsaokoarylone (9), a diarylheptanoid derivative, also showed considerable inhibitory activity with IC50 value of 11.6 µM. On the other hand, simple flavanones (1 – 4) and a bicyclic nonane (6) showed inactive inhibitory effects (IC50 values >50 µM). None of the compounds showed obvious cytotoxicity at concentration of 50 µM in the MTT assay (data not shown). These results indicate that three chalcones and a diarylheptanoid from A. tsao-ko are potential natural products for developing anti-inflammatory agents.
Figures and Tables
Acknowledgments
This work was financially supported by the Research Year of Chungbuk National University in 2016.
References
1. Tang W, Eisenbrand B. Handbook of Chinese medicinal plants: Chemistry, pharmacology, toxicology. Weinheim: Wiley-VCH;2011. p. 106.
4. Starkenmann C, Mayenzet F, Brauchli R, Wunsche L, Vial C. J Agric Food Chem. 2007; 55:10902–10907.

5. Hong SS, Lee JH, Choi YH, Jeong W, Ahn EK, Lym SH, Oh JS. Tetrahedron Lett. 2015; 56:6681–6684.
6. Itokawa H, Morita M, Mihashi S. Phytochemistry. 1981; 20:2503–2506.
10. McCormick S, Robson K, Bohm B. Phytochemistry. 1985; 24:1614–1616.
13.
L Aigner
E Oberbauer-Hofmann
S Couillard-Despres
FJ Rivera
H Riepl
C Urmann
M Biendl
. US patents. 9527860B2. 2016.
14. Moon SS, Cho SC, Lee JY. Bull Korean Chem Soc. 2005; 26:447–450.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download