Abstract
Background
Systemic lupus erythematosus (SLE) is an autoimmune disease that is a significant source of morbidity and mortality when it manifests in the central nervous system. The early detection and treatment of neuropsychiatric SLE (NPSLE) is very important, but a confirmative diagnostic tool has yet to be developed.
A diversity of neurologic and psychiatric symptoms in systemic lupus erythematosus (SLE) are reported to occur in 14-75% of patients either prior to the diagnosis or during the course of their illness.1 The involvement of the central nervous system (CNS) in SLE is a serious but potentially treatable illness, yet SLE still presents a very difficult diagnostic challenge. Neuropsychiatric involvement in SLE (NPSLE) appears to be caused by complex pathologic processes attributed to autoantibody mediated neuronal or vascular injury, intrathecal production of inflammatory cytokines, and disruption of the blood-brain-barrier (BBB).2 The frequency of NPSLE varies widely, depending on the type of manifestations and the method used for evaluation. The role of the different neuroimaging modalities in the diagnosis and evaluation of disease activities of NPSLE is still very controversial. Magnetic resonance imaging (MRI) remains the gold standard for the non-invasive assessment of NPSLE but there are limitations with its sensitivity and specificity. Single photon emission computed tomography (SPECT) or positron emission tomography (PET) have been found to be useful for early identification of blood-flow or metabolic abnormalities of NPSLE, but they lack spatial resolution. The quantification of change is straightforward.3,4 In this paper we report about brain PET findings in a young woman with NPSLE who first presented with amnesia and anosmia as neurological symptoms.
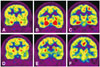
A 20-year-old right-handed female came to the emergency room, complaining about anosmia, a headache and a mild fever. These symptoms had suddenly started 7 days previous and gradually became worse. Three months earlier she had been diagnosed with SLE due to the presence of arthritis in her hands and knees, Raynaud phenomenon, positive antinuclear antibodies (ANA) and positive anti-dsDNA antibodies. There was no history of oral ulcers, malar or discoid rash, photosensitivity, cardio-pulmonary involvement, hair loss, neurologic disorders like seizures, and/or psychosis. She had no family history of either rheumatic or neurologic diseases. She denied smoking and did not consume alcohol. On admission, the patient had taken medications associated with SLE (prednisone 7.5 mg/day, hydroxychloroquine sulfate 300 mg/day). Her vital signs showed a mildly elevated body temperature (37.5℃), but were otherwise within normal limits. The physical examinations were unremarkable. Neurological examinations revealed anosmia, dysgeusia, and impairment of recent memory. She could not detect any scent in either nostril based on an olfactory recognition test, and she also felt that everything tasted sweet even if it was not. Her memory revealed severe impairment based on a three word recall test from the Mini-Mental State Examination (MMSE) (26/30).5 Other neurological examinations showed unremarkable findings. Laboratory findings demonstrated the presence of positive ANA, anti-rheumatoid antibodies, and anti-ribonucleoprotein antibodies (>8.0 AI). Cerebrospinal fluid tests revealed mild pleocystosis with dominance of lymphocytes (white blood cell: 11, red blood cell: 0), but otherwise findings were within normal limits. We diagnosed her with NPSLE, and then we performed a brain MRI with contrast and more detailed neuropsychological tests. The brain MRI revealed unremarkable findings (Fig. 1) but the neuropsychological tests showed a marked decrease in immediate and delayed recall on the Rey-Kim Memory Test, and mild executive dysfunction on the Kims Frontal-Executive Neuropsychological Test. However, language, visuoconstructive and praxis abilities were within normal limits (Table 1).6,7,8 An electroencephalogram showed normal background activities and rhythms. Three days after admission, we performed brain imaging by 18fluorodeoxy glucose-positron emission tomography (FDG-PET). The brain FDG-PET showed marked hypermetabolism bilaterally in both the parahippocampal gyrus and the amygdala (Fig. 2A, B, and C). We administered an augmented dose of prednisone (25 mg/day), cyclophosphamide (100 mg/day), and hydroxychloroquine sulfate for 3 weeks and slowly tapered them off. Her cognitive impairment gradually improved, and her olfactory and gustatory dysfunctions normalized. After three months, we performed a follow-up brain FDG-PET scan and detailed neuropsychological tests. The follow-up FDG-PET scan showed normal glucose consumption bilaterally in both the parahippocampal gyrus and the amygdala (Fig. 2D, E, and F). The follow-up neurolopsychological tests still revealed impairments in immediate and delayed recall for verbal and visual memory, but her results had slightly improved compared to the former neuropsychological results. Moreover, the frontal executive functions normalized (Table 1).
Neuropsychiatric manifestations in the form of cognitive dysfunction have been found in up to 80% of patients with SLE. Although patients were influenced by mood disorders, metabolic disturbances, and medications for treatment of cognitive function, previous studied have shown that SLE patients demonstrate cognitive deficits independent of these confounding variables. Notable cognitive impairment occurred in attention, concentration and memory, executive function, and visuospatial skill.9,10 In our reported case, a neuropsychological evaluation indicated marked impairment of verbal and visual memory as well as frontal executive function, but relatively mild impairments in language and visuospatial function. Brain PET findings in the patient demonstrated increased glucose metabolism bilaterally in both the amygdala and the parahippocampal gyrus. Memory has been shown to be the most commonly impaired cognitive process in patients with NPSLE and is consistent with hippocampal changes associated with the disease.11 In SLE, overproduction of autoantibodies against N-methyl-D-aspartic acid receptors (NMDARs) and disruption of BBB caused brain tissue neuronal death or dysfunction. Because the density of NMDARs is highest in the hippocampus and amygdala, these structures may be particularly vulnerable to this process. While NMDARs play an important role in long-term potentiation for learning and memory, excessive production of anti-NMDAR antibodies that are associated with cell death within the limbic system can lead to cognitive dysfunction and mood disorders.12,13 However, NPSLE represents a diagnostic and therapeutic challenge due to the wide range of presentations, lack of diagnostic tests with adequate sensitivity and specificity, and limited controlled evidence for selection of optimal treatments. Brain MRI is the current gold standard in imaging assessment of NPSLE, with T2-weighted image (T2WI) and fluid attenuated inversion recovery (FLAIR) allowing visualization of small high signal lesions in subcortical white matter of the frontoparietal or periventricular regions. In a few cases of NPSLE, brain MRI revealed a reversal of abnormal findings in the bilateral basal ganglia through diffusion weighted imaging, and tiny hyperintensities in frontal periventricular white matter through T2WI and FLAIR images.14,15 However, in many patients with obvious CNS manifestations, MRI may not show abnormalities, especially for affective disorders, confusional states, and headache. More than 40% of SLE patients with various neuropsychiatric manifestations have MRI scans that show no findings. Another drawback of MRI is the difficulty in differentiating lesions of active CNS manifestations from old lesions.16,17,18 More recently, brain SPECT and 18FDG-PET have been explored as functional imaging tools and both appear to be more sensitive in detecting subtle brain changes in NPSLE. The most prevalent finding of brain 18FDG-PET in NPSLE is hypometabolism in the parieto-occipital or frontal lobe. Brain PET scans can identify fluctuations in regional cerebral metabolism even in the absence of MRI lesions. In a recent study, brain 18FDG-PET revealed significant glucose metabolic abnormalities in up to 75% patients of NPSLE. However, neuropsychiatric symptoms were not topographically correlated with the brain 18FDG-PET findings.4,19,20 In our reported case, the brain MRI also showed unremarkable findings, but a PET scan revealed exceptional hypermetabolism in both the amygdala and parahippocampal regions. We suggest that these findings reflect active inflammation by anti-NMDARs and are related to her neuropsychiatric manifestations. Moreover, we recognized that her cognitive dysfunctions improved and a follow-up PET scan showed near normalization after three months.
We report a unique case of NPSLE with in which reversal in functional damage was observed in images retrieved through brain FDG-PET, but where MRI scans did not reveal any abnormal findings. Knowledge of the advantages and limitations of each anatomical and functional imaging technique are fundamental to correctly identifying a patient with NPSLE. Therefore, further research and more follow-up data may help to determine the best methods to diagnose NPSLE and to understand the underlying pathogenic mechanisms.
Figures and Tables
 | Fig. 1Brain magnetic resonance imaging on admission. T2-weighted image (A and B), fluid-attenuated inversion recovery (C and D) image showed unremarkable findings. |
References
1. Borowoy AM, Pope JE, Silverman E, Fortin PR, Pineau C, Smith CD, et al. Neuropsychiatric lupus: the prevalence and autoantibody associations depend on the definition: results from the 1000 faces of lupus cohort. Semin Arthritis Rheum. 2012; 42:179–185.

2. Fanouriakis A, Boumpas DT, Bertsias GK. Pathogenesis and treatment of CNS lupus. Curr Opin Rheumatol. 2013; 25:577–583.

3. Govoni M, Castellino G, Padovan M, Borrelli M, Trotta F. Recent advances and future perspective in neuroimaging in neuropsychiatric systemic lupus erythematosus. Lupus. 2004; 13:149–158.

4. Peterson PL, Axford JS, Isenberg D. Imaging in CNS lupus. Best Pract Res Clin Rheumatol. 2005; 19:727–739.

5. Folstein MF, Folstein SE, McHugh PR. "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975; 12:189–198.

6. Kim HG. Assessment of memory disorders using Rey-Kim Memory Test. Korean J Rehabil Psychol. 2001; 8:29–48.

7. Kim HG. Clinical evaluation of the frontal lobe syndrome using Kims frontal-executive neuropsychological test. Korean J Rehabil Psychol. 2001; 8:173–190.

8. Kim H, Na DL. Normative data on the Korean version of the Boston Naming Test. J Clin Exp Neuropsychol. 1999; 21:127–133.

9. Glanz BI, Schur PH, Lew RA, Khoshbin S. Lateralized cognitive dysfunction in patients with systemic lupus erythematosus. Lupus. 2005; 14:896–902.

10. Emori A, Matsushima E, Aihara O, Ohta K, Koike R, Miyasaka N, et al. Cognitive dysfunction in systemic lupus erythematosus. Psychiatry Clin Neurosci. 2005; 59:584–589.

11. Kozora E, Arciniegas DB, Filley CM, West SG, Brown M, Miller D, et al. Cognitive and neurologic status in patients with systemic lupus erythematosus without major neuropsychiatric syndromes. Arthritis Rheum. 2008; 59:1639–1646.

12. DeGiorgio LA, Konstantinov KN, Lee SC, Hardin JA, Volpe BT, Diamond B. A subset of lupus anti-DNA antibodies cross-reacts with the NR2 glutamate receptor in systemic lupus erythematosus. Nat Med. 2001; 7:1189–1193.

13. Watson P, Storbeck J, Mattis P, Mackay M. Cognitive and emotional abnormalities in systemic lupus erythematosus: evidence for amygdala dysfunction. Neuropsychol Rev. 2012; 22:252–270.
14. Sato S, Nakajima J, Shimura M, Kawashima H, Yoshio T, Hara Y. Reversible basal ganglia lesions in neuropsychiatric lupus: a report of three pediatric cases. Int J Rheum Dis. 2014; 17:274–279.
15. Poil AR, Yousef Khan F, Lutf A, Hammoudeh M. Chorea as the first and only manifestation of systemic lupus erythematosus. Case Rep Rheumatol. 2012; 2012:907402.

16. Sibbitt WL Jr, Sibbitt RR, Brooks WM. Neuroimaging in neuropsychiatric systemic lupus erythematosus. Arthritis Rheum. 1999; 42:2026–2038.

17. Abreu MR, Jakosky A, Folgerini M, Brenol JC, Xavier RM, Kapczinsky F. Neuropsychiatric systemic lupus erythematosus: correlation of brain MR imaging, CT, and SPECT. Clin Imaging. 2005; 29:215–221.

18. Luyendijk J, Steens SC, Ouwendijk WJ, Steup-Beekman GM, Bollen EL, van der Grond J, et al. Neuropsychiatric systemic lupus erythematosus: lessons learned from magnetic resonance imaging. Arthritis Rheum. 2011; 63:722–732.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download