Abstract
Background
As rapidly progressive dementia (RPD), general paresis and Creutzfeldt-Jakob disease (CJD) may have overlapping clinical presentation due to a wide variety of clinical manifestations.
Case Report
A 57-year-old man presented with rapid progressive cognitive decline, behavioral change, ataxic gait, tremor and pyramidal signs for 3 months. In addition to these multiple systemic involvements, positive result for the cerebrospinal fluid (CSF) 14-3-3 protein tentatively diagnosed him as probable CJD. However, due to increased serum rapid plasma reagin, venereal disease research laboratory, and fluorescent treponemal antibody-absorption reactivity in CSF, the final diagnosis was changed to general paresis.
Conclusions
A patient with RPD needs to be carefully considered for differential diagnosis, among a long list of diseases. It is important to rule out CJD, which is the most frequent in RPD and is a fatal disease with no cure. Diagnostic criteria or marker of CJD, such as 14-3-3 protein, may be inconclusive, and a typical pattern in diffusion-weighted imaging is important to rule out other reversible diseases.
Neurosyphilis develops in patients with syphilis due to the invasion of the central nerve system by Treponema pallidum. Due to its varied clinical presentations, the diagnosis is occasionally difficult. Early form includes asymptomatic presentation and meningitis, while the late form can be meningovascular, paretic and tabetic neurosyphilis.1 General paresis or paretic neurosyphilis usually develops more than 15 years after infection, with associated clinical manifestations such as behavioural changes, cognitive impairment, psychiatric features as well as dysarthria, myoclonus, intention tremor, hyperreflexia, seizure and Argyll Robertson pupils.2 Creutzfeldt-Jakob disease (CJD) is a fatal prion disease characterized by rapidly progressive dementia (RPD), myoclonus, pyramidal and extrapyramidal signs, visual field defect, and cerebellar symptoms.3 Due to these extensively varied clinical manifestations, general paresis and CJD may have overlapping clinical symptoms. We herein report a patient with RPD, whose initial clinical presentation was memory loss, and was misdiagnosed as CJD.
A 57-year-old man transferred to the neurocognitive behavior center of Seoul National University Bundang Hospital in 2012, from other university hospital. He presented with a three-month history of rapidly progressive memory loss and behavioral changes. Although previously a healthy man, he was unable to recollect the place for the tools, and his processing speed had decreased substantially while working as a carpenter, due to which his boss suggested he should quit his job. He experienced tremors in the upper and lower extremities, especially when targeting objects such as tools or shoes; he sometimes also experienced jerky movement. Previously of a meek and mild disposition, he had recent episodes of short-temper with emotional incontinence.
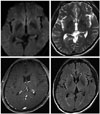
Neurological examination showed increased deep tendon reflex (DTR) at biceps, knees and ankles, with Hoffmann sign and ankle clonus at the four extremities. Dysmetria at finger to nose and heel to shin test, dysdiadochokinesia at rapid alternating movement, and bilateral sway at tandem gait, were noticed. Postural tremor at the upper extremities, and myoclonic jerky movement at the right big toe were also observed. Magnetic resonance imaging (MRI) and electoroencephalography (EEG) were unremarkable (Fig. 1). The Korean version of Mini Mental Status Examination score was 29/30 (normal), whereas the comprehensive neuropsychological test showed decreased memory at immediate, delayed recall as well as recognition, and scores on stroop test fell below normal limits (Table 1). Before being transferred to our clinic, he underwent cerebrospinal fluid (CSF) tapping with mild pleocytosis (100 leukocyte/mm3, mainly lymphocyte) and hyperproteinorrachia (134 md/dL), as well as tested 14-3-3 positivity on Western blot. After confirmation of CSF study, he was transferred to our hospital as a suspected CJD, due to the subacute progressive cognitive impairment, tremor, myoclonus, increased DTR, pathologic reflex, cerebellar ataxia and 14-3-3 protein positivity in CSF.
Initial assessment included infectious disease, CJD, paraneoplastic syndrome and toxic-metabolic disease. Further laboratory studies were performed after transfer. Tumor markers, thyroid function test, vitamin B12, anti-nuclear antigen, and anti-HIV test were all negative, but serum RPR level was increased up to 33.2 (normal range as 0−1 RU). This was followed by venereal disease research laboratory positivity (1:64) and fluorescent treponemal antibody-absorption reactivity in CSF. Additional history was taken, and his wife revealed that the patient had an ulcerative lesion with discharge on his penis about 15 years ago, but he was reluctant to get regular treatment from a hospital. Thereafter, the diagnosis was changed to general paresis by neurosyphilis, and the patient was treated with aqueous Penicillin G 24 million units a day intravenously for 2 weeks. Two weeks after treatment, neurological examination revealed partial recovery of gait and limb ataxia, whereas cognitive dysfunction persisted in the memory domain, in addition to visuospatial and frontal executive domains, according to the follow-up study after 1 year (Table 1).
RPD with behavioral changes is a common presentation of general paresis, and includes disturbances such as neuropsychiatric symptoms, memory impairment, hallucination, delusion and delirium. Less frequent clinical manifestations include stroke, seizures, cranial nerve and brain stem dysfunction.4 Our patient showed behavior change, pyramidal signs, cerebellar signs and subacute dementia, which were consistent with previous studies.56 He was initially diagnosed as CJD since his clinical symptoms and positive CSF 14-3-3 protein were compatible with probable sporadic CJD (sCJD) according to diagnostic criteria from national CJD surveillance unit.7
In neurosyphilis, CSF examination reveals lymphocytic and monocytic pleocytosis and hyperproteinorrachia,1 and such a pattern was observed in our patient. In addition, the 14-3-3 protein was also positive, due to which this patient was misdiagnosed as sCJD. CSF 14-3-3 protein reveals a low specificity at 68%,8 because it can show false positive results in other diseases such as hypoxic brain damage, paraneoplastic syndrome, metabolic syndrome, herpes simplex encephalitis or brain metastasis.9 Hence, some laboratories use other CSF markers such as S100b protein or tau protein in combination with CSF 14-3-3 protein, as markers of injured brain tissue other than CJD.10 Among the CSF 14-3-3 protein positive referred cases to the Korea Centers For Disease Control and Prevention, the ratio of confirmed CJD is about a fourth, with remaining cases having a final diagnosis of other diseases, including toxic-metabolic disease, epileptic disorder, tumor or infectious disease.11
To exclude other diseases that clinically mimic neurosyphilis, such as sCJD, autoimmune/inflammatory, malignancy or toxic-metabolic diseases, brain MRI is helpful, although there is no specific pattern.4 According to a previous study, diffuse or focal atrophy as well as white matter changes and infarcts are described.12 Since our patient revealed no significant abnormality in brain MRI, we conclude that the disease course might not be enough to induce cortical atrophy. Brain MRI findings of sCJD has higher sensitivity and specificity (96% and 93%, respectively), and diffusion-weighted imaging (DWI) may reflect disease stage-dependent difference even with negative EEG and CSF 14-3-3 protein.1314 Therefore, MRI features are incorporated in the diagnostic criteria of sCJD,715 and a negative finding of DWI in our patient was considered before making diagnosis of probable sCJD.
The cognitive impairment pattern of neurosyphilis as general paresis was reported as being similar to mild Alzheimer's disease (AD),16 and our patient performed poorly at immediate, delayed recall, and recognition of verbal learning test, thus encoding type memory impairment. Considering the fact that bilateral mesiotemporal hyperintensity on T2-weighted image on MRI or AD-like atrophy can occasionally be observed in patients with neurosyphilis,1617 it is possible that the pathophysiological changes of medial temporal lobe resulted in AD-like cognitive impairment.
The prevalence of syphilis has decreased since the 1940s due to penicillin, and is maintained as low as <1% in Korea.18 As this illness has become rare, opportunities for experiencing general paresis are few. If tissue damage has already occurred, recovery is often incomplete, and patients with general paresis may have different outcome.19 Since appropriate early treatment may prevent progression and allow better recovery, precise and early diagnosis is much desired for neurosyphilis, and we hope that our experience emphasizes the importance of differential points of neuroshypilis, from other diseases with rapid progressive dementia.
Figures and Tables
References
1. Ghanem KG. REVIEW: neurosyphilis: a historical perspective and review. CNS Neurosci Ther. 2010; 16:e157–e168.

2. Bradley WG. Neurology in clinical practice: principles of diagnosis and management. 4th ed. Philadelphia: Taylor & Francis;2004.

3. Brown P, Gibbs CJ Jr, Rodgers-Johnson P, Asher DM, Sulima MP, Bacote A, et al. Human spongiform encephalopathy: the National Institutes of Health series of 300 cases of experimentally transmitted disease. Ann Neurol. 1994; 35:513–529.

4. Stefani A, Riello M, Rossini F, Mariotto S, Fenzi F, Gambina G, et al. Neurosyphilis manifesting with rapidly progressive dementia: report of three cases. Neurol Sci. 2013; 34:2027–2030.

5. Jantzen SU, Ferrea S, Langebner T, Gaebel W, Griese M, Arendt G, et al. Late-stage neurosyphilis presenting with severe neuropsychiatric deficits: diagnosis, therapy, and course of three patients. J Neurol. 2012; 259:720–728.

6. Zheng D, Zhou D, Zhao Z, Liu Z, Xiao S, Xing Y, et al. The clinical presentation and imaging manifestation of psychosis and dementia in general paresis: a retrospective study of 116 cases. J Neuropsychiatry Clin Neurosci. 2011; 23:300–307.

7. The National CJD Research & Surveillance Unit (NCJDRSU, formerly NCJDSU). National Creutzfeldt-Jakob disease Surveillance Diagnositic Criteria. cited 2016 Nov 15. Available from: http://www.cjd.ed.ac.uk/documents/criteria.pdf.

8. Zerr I, Pocchiari M, Collins S, Brandel JP, de Pedro Cuesta J, Knight RS, et al. Analysis of EEG and CSF 14-3-3 proteins as aids to the diagnosis of Creutzfeldt-Jakob disease. Neurology. 2000; 55:811–815.

9. Sanchez-Juan P, Green A, Ladogana A, Cuadrado-Corrales N, Saanchez-Valle R, Mitrovaa E, et al. CSF tests in the differential diagnosis of Creutzfeldt-Jakob disease. Neurology. 2006; 67:637–643.

10. Satoh J, Kurohara K, Yukitake M, Kuroda Y. The 14-3-3 protein detectable in the cerebrospinal fluid of patients with prion-unrelated neurological diseases is expressed constitutively in neurons and glial cells in culture. Eur Neurol. 1999; 41:216–225.

11. Jang JW, Park YH, Lim JS, Park SC, Cheong HK, Kim JE, et al. Neurologists' awareness and preparedness on prion diseases in Korea. Dement Neurocognitive Disord. 2013; 12:9–20.

12. Nagappa M, Sinha S, Taly AB, Rao SL, Nagarathna S, Bindu PS, et al. Neurosyphilis: MRI features and their phenotypic correlation in a cohort of 35 patients from a tertiary care university hospital. Neuroradiology. 2013; 55:379–388.

13. Park SY, Wang MJ, Jang JW, Park YH, Lim JS, Youn YC, et al. The clinical stages of sporadic Creutzfeldt-Jakob disease with Met/Met genotype in Korean patients. Eur Neurol. 2016; 75:213–222.

14. Vitali P, Maccagnano E, Caverzasi E, Henry RG, Haman A, Torres-Chae C, et al. Diffusion-weighted MRI hyperintensity patterns differentiate CJD from other rapid dementias. Neurology. 2011; 76:1711–1719.

15. Zerr I, Kallenberg K, Summers DM, Romero C, Taratuto A, Heinemann U, et al. Updated clinical diagnostic criteria for sporadic Creutzfeldt-Jakob disease. Brain. 2009; 132(Pt 10):2659–2668.

16. Wang J, Guo Q, Zhou P, Zhang J, Zhao Q, Hong Z. Cognitive impairment in mild general paresis of the insane: AD-like pattern. Dement Geriatr Cogn Disord. 2011; 31:284–290.

17. Hama K, Ishiguchi H, Tuji T, Miwa H, Kondo T. Neurosyphilis with mesiotemporal magnetic resonance imaging abnormalities. Intern Med. 2008; 47:1813–1817.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download