1. Song JH, Huh K, Chung DR. Community-acquired pneumonia in the Asia-Pacific region. Semin Respir Crit Care Med. 2016; 37:839–854. PMID:
27960208.

2. Yoon HK. Changes in the epidemiology and burden of community-acquired pneumonia in Korea. Korean J Intern Med. 2014; 29:735–737. PMID:
25378971.

3. Yoo KH, Yoo CG, Kim SK, Jung JY, Lee MG, Uh ST, Shim TS, Jeon K, Shim JJ, Lee HB, Chung CR, Kang KW, Jung KS. Economic burden and epidemiology of pneumonia in Korean adults aged over 50 years. J Korean Med Sci. 2013; 28:888–895. PMID:
23772154.

4. Seong GM, Kim M, Lee J, Lee JH, Jeong SY, Choi Y, Kim WJ. Healthcare-associated pneumonia among hospitalized patients: Is it different from community acquired pneumonia? Tuberc Respir Dis (Seoul). 2014; 76:66–74. PMID:
24624215.

5. Kim JE, Kim UJ, Kim HK, Cho SK, An JH, Kang SJ, Park KH, Jung SI, Jang HC. Predictors of viral pneumonia in patients with community-acquired pneumonia. PLoS One. 2014; 9:e114710. PMID:
25531901.

6. Kang YS, Ryoo SR, Byun SJ, Jeong YJ, Oh JY, Yoon YS. Antimicrobial resistance and clinical outcomes in nursing home-acquired pneumonia, compared to community-acquired pneumonia. Yonsei Med J. 2017; 58:180–186. PMID:
27873512.

7. Jeong BH, Koh WJ, Yoo H, Um SW, Suh GY, Chung MP, Kim H, Kwon OJ, Jeon K. Performances of prognostic scoring systems in patients with healthcare-associated pneumonia. Clin Infect Dis. 2013; 56:625–632. PMID:
23155150.

8. Jeon EJ, Cho SG, Shin JW, Kim JY, Park IW, Choi BW, Choi JC. The difference in clinical presentations between healthcare-associated and community-acquired pneumonia in university-affiliated hospital in Korea. Yonsei Med J. 2011; 52:282–287. PMID:
21319347.

9. Choi MJ, Song JY, Cheong HJ, Jeon JH, Kang SH, Jung EJ, Noh JY, Kim WJ. Clinical usefulness of pneumococcal urinary antigen test, stratified by disease severity and serotypes. J Infect Chemother. 2015; 21:672–679. PMID:
26212866.

10. Chong YP, Jung KS, Lee KH, Kim MN, Moon SM, Park S, Hur J, Kim DM, Jeon MH, Woo JH. The bacterial etiology of community-acquired pneumonia in Korea: A nationwide prospective multicenter study. Infect Chemother. 2010; 42:397–403.

11. Song JH, Oh WS, Kang CI, Chung DR, Peck KR, Ko KS, Yeom JS, Kim CK, Kim SW, Chang HH, Kim YS, Jung SI, Tong Z, Wang Q, Huang SG, Liu JW, Lalitha MK, Tan BH, Van PH, Carlos CC, So T; Asian Network for Surveillance of Resistant Pathogens Study Group. Epidemiology and clinical outcomes of community-acquired pneumonia in adult patients in Asian countries: a prospective study by the Asian network for surveillance of resistant pathogens. Int J Antimicrob Agents. 2008; 31:107–114. PMID:
18162378.

12. Sohn JW, Park SC, Choi YH, Woo HJ, Cho YK, Lee JS, Sim HS, Kim MJ. Atypical pathogens as etiologic agents in hospitalized patients with community-acquired pneumonia in Korea: a prospective multi-center study. J Korean Med Sci. 2006; 21:602–607. PMID:
16891800.
13. Lee SJ, Lee MG, Jeon MJ, Jung KS, Lee HK, Kishimoto T. Atypical pathogens in adult patients admitted with community-acquired pneumonia in Korea. Jpn J Infect Dis. 2002; 55:157–159. PMID:
12501255.
14. Choi SH, Hong SB, Ko GB, Lee Y, Park HJ, Park SY, Moon SM, Cho OH, Park KH, Chong YP, Kim SH, Huh JW, Sung H, Do KH, Lee SO, Kim MN, Jeong JY, Lim CM, Kim YS, Woo JH, Koh Y. Viral infection in patients with severe pneumonia requiring intensive care unit admission. Am J Respir Crit Care Med. 2012; 186:325–332. PMID:
22700859.

15. Mandell LA, Wunderink RG, Anzueto A, Bartlett JG, Campbell GD, Dean NC, Dowell SF, File TM Jr, Musher DM, Niederman MS, Torres A, Whitney CG; Infectious Diseases Society of America. American Thoracic Society. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007; 44(Suppl 2):S27–S72. PMID:
17278083.
16. Lim WS, Baudouin SV, George RC, Hill AT, Jamieson C, Le Jeune I, Macfarlane JT, Read RC, Roberts HJ, Levy ML, Wani M, Woodhead MA; Pneumonia Guidelines Committee of the BTS Standards of Care Committee. BTS guidelines for the management of community acquired pneumonia in adults: update 2009. Thorax. 2009; 64(Suppl 3):iii1–iii55. PMID:
19783532.

17. Song JH, Jung SI, Ki HK, Shin MH, Ko KS, Son JS, Chang HH, Kim SW, Lee H, Kim YS, Oh WS, Peck KR, Chongthaleong A, Lalitha MK, Perera J, Yee TT, Jamal F, Kamarulzaman A, Carlos CC, So T; Asian Network for Surveillance of Resistant Pathogens Study Group. Clinical outcomes of pneumococcal pneumonia caused by antibiotic-resistant strains in Asian countries: a study by the Asian Network for Surveillance of Resistant Pathogens. Clin Infect Dis. 2004; 38:1570–1578. PMID:
15156445.

18. Kim SH, Song JH, Chung DR, Thamlikitkul V, Yang Y, Wang H, Lu M, So TM, Hsueh PR, Yasin RM, Carlos CC, Pham HV, Lalitha MK, Shimono N, Perera J, Shibl AM, Baek JY, Kang CI, Ko KS, Peck KR; ANSORP Study Group. Changing trends in antimicrobial resistance and serotypes of Streptococcus pneumoniae isolates in Asian countries: an Asian Network for Surveillance of Resistant Pathogens (ANSORP) study. Antimicrob Agents Chemother. 2012; 56:1418–1426. PMID:
22232285.

19. Kim SH, Song SA, Yi J, Song D, Chang CL, Park DC, Urm SH, Kim HR, Shin JH. Distribution and antimicrobial resistance of Streptococcus pneumoniae at four university hospitals in Busan and Gyeongnam. Ann Clin Microbiol. 2016; 19:48–53.
20. Kim T, Park SJ, Chong YP, Park KH, Lee YM, Hong HL, Kim HS, Kim ES, Lee S, Choi DR, Kim SH, Jeong JY, Lee SO, Choi SH, Woo JH, Kim YS. Fluoroquinolone resistance of
Streptococcus pneumoniae isolates causing invasive disease: special focus on zabofloxacin. Diagn Microbiol Infect Dis. 2016; 86:181–183. PMID:
27498611.
21. Lee S, Bae S, Lee KJ, Yu JY, Kang Y. Changes in serotype prevalence and antimicrobial resistance among invasive Streptococcus pneumoniae isolates in Korea, 1996-2008. J Med Microbiol. 2013; 62:1204–1210. PMID:
23657529.

22. Torumkuney D, Chaiwarith R, Reechaipichitkul W, Malatham K, Chareonphaibul V, Rodrigues C, Chitins DS, Dias M, Anandan S, Kanakapura S, Park YJ, Lee K, Lee H, Kim JY, Lee Y, Lee HK, Kim JH, Tan TY, Heng YX, Mukherjee P, Morrissey I. Results from the survey of antibiotic resistance (SOAR) 2012-14 in Thailand, India, South Korea and Singapore. J Antimicrob Chemother. 2016; 71(Suppl 1):i3–i19. PMID:
27048580.

23. Bae S, Lee J, Lee J, Kim E, Lee S, Yu J, Kang Y. Antimicrobial resistance in
Haemophilus influenzae respiratory tract isolates in Korea: results of a nationwide acute respiratory infections surveillance. Antimicrob Agents Chemother. 2010; 54:65–71. PMID:
19884366.
24. Kim IS, Ki CS, Kim S, Oh WS, Peck KR, Song JH, Lee K, Lee NY. Diversity of ampicillin resistance genes and antimicrobial susceptibility patterns in
Haemophilus influenzae strains isolated in Korea. Antimicrob Agents Chemother. 2007; 51:453–460. PMID:
17116681.
25. Hong KB, Choi EH, Lee HJ, Lee SY, Cho EY, Choi JH, Kang HM, Lee J, Ahn YM, Kang YH, Lee JH. Macrolide resistance of
Mycoplasma pneumoniae, South Korea, 2000-2011. Emerg Infect Dis. 2013; 19:1281–1284. PMID:
23876792.
26. Uh Y, Hong JH, Oh KJ, Cho HM, Park SD, Kim J, Yoon KJ. Macrolide resistance of
Mycoplasma pneumoniae and its detection rate by real-time PCR in primary and tertiary care hospitals. Ann Lab Med. 2013; 33:410–414. PMID:
24205489.
27. Sohn KM, Chung DR, Baek JY, Kim SH, Joo EJ, Ha YE, Ko KS, Kang CI, Peck KR, Song JH. Post-influenza pneumonia caused by the USA300 community-associated methicillin-resistant
Staphylococcus aureus in Korea. J Korean Med Sci. 2012; 27:313–316. PMID:
22379344.
28. Leem AY, Jung WJ, Kang YA, Park SC, Kim YJ, Hwang ED, Kim EY, Jung KS, Park MS, Kim SY, Kim YS, Kim SK, Chang J, Jung JY. Comparison of methicillin-resistant
Staphylococcus aureus community-acquired and healthcare-associated pneumonia. Yonsei Med J. 2014; 55:967–974. PMID:
24954325.
29. Hwang JW, Joo EJ, Ha JM, Lee W, Kim E, Yune S, Chung DR, Jeon K. Community-acquired necrotizing pneumonia caused by ST72-SCCmec type IV-methicillin-resistant
Staphylococcus aureus in Korea. Tuberc Respir Dis (Seoul). 2013; 75:75–78. PMID:
24023561.
30. Ruuskanen O, Lahti E, Jennings LC, Murdoch DR. Viral pneumonia. Lancet. 2011; 377:1264–1275. PMID:
21435708.

31. Lieberman D, Lieberman D, Shimoni A, Keren-Naus A, Steinberg R, Shemer-Avni Y. Identification of respiratory viruses in adults: nasopharyngeal versus oropharyngeal sampling. J Clin Microbiol. 2009; 47:3439–3443. PMID:
19726607.

32. Loens K, Van Heirstraeten L, Malhotra-Kumar S, Goossens H, Ieven M. Optimal sampling sites and methods for detection of pathogens possibly causing community-acquired lower respiratory tract infections. J Clin Microbiol. 2009; 47:21–31. PMID:
19020070.

33. Prina E, Ranzani OT, Torres A. Community-acquired pneumonia. Lancet. 2015; 386:1097–1108. PMID:
26277247.

34. Musher DM, Roig IL, Cazares G, Stager CE, Logan N, Safar H. Can an etiologic agent be identified in adults who are hospitalized for community-acquired pneumonia: results of a one-year study. J Infect. 2013; 67:11–18. PMID:
23523447.

35. Johansson N, Kalin M, Tiveljung-Lindell A, Giske CG, Hedlund J. Etiology of community-acquired pneumonia: increased microbiological yield with new diagnostic methods. Clin Infect Dis. 2010; 50:202–209. PMID:
20014950.

36. Johnstone J, Majumdar SR, Fox JD, Marrie TJ. Viral infection in adults hospitalized with community-acquired pneumonia: prevalence, pathogens, and presentation. Chest. 2008; 134:1141–1148. PMID:
18689592.
37. Falsey AR, Becker KL, Swinburne AJ, Nylen ES, Formica MA, Hennessey PA, Criddle MM, Peterson DR, Baran A, Walsh EE. Bacterial complications of respiratory tract viral illness: a comprehensive evaluation. J Infect Dis. 2013; 208:432–441. PMID:
23661797.

38. Jain S, Self WH, Wunderink RG, Fakhran S, Balk R, Bramley AM, Reed C, Grijalva CG, Anderson EJ, Courtney DM, Chappell JD, Qi C, Hart EM, Carroll F, Trabue C, Donnelly HK, Williams DJ, Zhu Y, Arnold SR, Ampofo K, Waterer GW, Levine M, Lindstrom S, Winchell JM, Katz JM, Erdman D, Schneider E, Hicks LA, McCullers JA, Pavia AT, Edwards KM, Finelli L; CDC EPIC Study Team. Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med. 2015; 373:415–427. PMID:
26172429.

39. Karhu J, Ala-Kokko TI, Vuorinen T, Ohtonen P, Syrjälä H. Lower respiratory tract virus findings in mechanically ventilated patients with severe community-acquired pneumonia. Clin Infect Dis. 2014; 59:62–70. PMID:
24729498.

40. Musher DM, Thorner AR. Community-acquired pneumonia. N Engl J Med. 2014; 371:1619–1628. PMID:
25337751.

41. Avni T, Bieber A, Green H, Steinmetz T, Leibovici L, Paul M.Diagnostic accuracy of PCR alone and compared to urinary antigen testing for detection of Legionella spp.: a systematic review. Clin Microbiol. 2016; 54:401–411.
42. Maze MJ, Slow S, Cumins AM, Boon K, Goulter P, Podmore RG, Anderson TP, Barratt K, Young SA, Pithie AD, Epton MJ, Werno AM, Chambers ST, Murdoch DR. Enhanced detection of Legionnaires' disease by PCR testing of induced sputum and throat swabs. Eur Respir J. 2014; 43:644–646. PMID:
24311761.

43. Cho MC, Kim H, An D, Lee M, Noh SA, Kim MN, Chong YP, Woo JH. Comparison of sputum and nasopharyngeal swab specimens for molecular diagnosis of
Mycoplasma pneumoniae, Chlamydophila pneumoniae, and
Legionella pneumophila. Ann Lab Med. 2012; 32:133–138. PMID:
22389880.
44. Vikerfors T, Brodin G, Grandien M, Hirschberg L, Krook A, Pettersson CA. Detection of specific IgM antibodies for the diagnosis of
Mycoplasma pneumoniae infections: a clinical evaluation. Scand J Infect Dis. 1988; 20:601–610. PMID:
3146809.
45. Nilsson A, Björkman P, Persson K. Polymerase chain reaction is superior to serology for the diagnosis of acute
Mycoplasma pneumoniae infection and reveals a high rate of persistent infection. BMC Microbiol. 2008; 8:93. PMID:
18547431.
46. Uldum SA, Jensen JS, Søndergård-Andersen J, Lind K. Enzyme immunoassay for detection of immunoglobulin M (IgM) and IgG antibodies to
Mycoplasma pneumoniae. J Clin Microbiol. 1992; 30:1198–1204. PMID:
1583120.
47. Parrott GL, Kinjo T, Fujita J. A compendium for
Mycoplasma pneumoniae. Front Microbiol. 2016; 7:513. PMID:
27148202.
48. Loens K, Beck T, Ursi D, Overdijk M, Sillekens P, Goossens H, Ieven M. Evaluation of different nucleic acid amplification techniques for the detection of
M. pneumoniae, C. pneumoniae and
Legionella spp. in respiratory specimens from patients with community-acquired pneumonia. J Microbiol Methods. 2008; 73:257–262. PMID:
18378345.
49. Herrera M, Aguilar YA, Rueda ZV, Muskus C, Vélez LA. Comparison of serological methods with PCR-based methods for the diagnosis of community-acquired pneumonia caused by atypical bacteria. J Negat Results Biomed. 2016; 15:3. PMID:
26932735.

50. Villegas E, Sorlózano A, Gutiérrez J.Serological diagnosis of Chlamydia pneumoniae infection: limitations and perspectives. J Med Microbiol. 2010; 59:1267–1274. PMID:
20724512.

51. Benitez AJ, Thurman KA, Diaz MH, Conklin L, Kendig NE, Winchell JM. Comparison of real-time PCR and a microimmunofluorescence serological assay for detection of Chlamydophila pneumoniae infection in an outbreak investigation. J Clin Microbiol. 2012; 50:151–153. PMID:
22031704.

52. Waterer GW. Diagnosing viral and atypical pathogens in the setting of community-acquired pneumonia. Clin Chest Med. 2017; 38:21–28. PMID:
28159158.

53. Syrjst H, Broas M, Suramo I, Ojala A, Lähde S. High-resolution computed tomography for the diagnosis of community-acquired pneumonia. Clin Infect Dis. 1998; 27:358–363. PMID:
9709887.

54. Heffner JE, Klein JS, Hampson C. Diagnostic utility and clinical application of imaging for pleural space infections. Chest. 2010; 137:467–479. PMID:
20133295.

55. Arenas-Jiménez J, Alonso-Charterina S, Sánchez-Payá J, Fernández-Latorre F, Gil-Sánchez S, Lloret-Llorens M.Evaluation of CT findings for diagnosis of pleural effusions. Eur Radiol. 2000; 10:681–690. PMID:
10795555.
56. Ketai L, Jordan K, Busby KH. Imaging infection. Clin Chest Med. 2015; 36:197–217. PMID:
26024600.

57. Raoof S, Amchentsev A, Vlahos I, Goud A, Naidich DP. Pictorial essay: multinodular disease: a high-resolution CT scan diagnostic algorithm. Chest. 2006; 129:805–815. PMID:
16537886.
58. Revel MP, Triki R, Chatellier G, Couchon S, Haddad N, Hernigou A, Danel C, Frija G. Is it possible to recognize pulmonary infarction on multisection CT images? Radiology. 2007; 244:875–882. PMID:
17709834.

59. Shah RM, Friedman AC. CT angiogram sign: incidence and significance in lobar consolidations evaluated by contrast-enhanced CT. AJR Am J Roentgenol. 1998; 170:719–721. PMID:
9490960.

60. Okada F, Ando Y, Tanoue S, Ishii R, Matsushita S, Ono A, Maeda T, Mori H. Radiological findings in acute
Haemophilus influenzae pulmonary infection. Br J Radiol. 2012; 85:121–126. PMID:
21224303.
61. Miller WT Jr, Mickus TJ, Barbosa E Jr, Mullin C, Van Deerlin VM, Shiley KT. CT of viral lower respiratory tract infections in adults: comparison among viral organisms and between viral and bacterial infections. Am J Roentgenol. 2011; 197:1088–1095. PMID:
22021500.

62. Nambu A, Saito A, Araki T, Ozawa K, Hiejima Y, Akao M, Ohki Z, Yamaguchi H.
Chlamydia pneumoniae: comparison with findings of
Mycoplasma pneumoniae and
Streptococcus pneumoniae at thin-section CT. Radiology. 2006; 238:330–338. PMID:
16304082.
63. Ors F, Deniz O, Bozlar U, Gumus S, Tasar M, Tozkoparan E, Tayfun C, Bilgic H, Grant BJ. High-resolution CT findings in patients with pulmonary tuberculosis: correlation with the degree of smear positivity. J Thorac Imaging. 2007; 22:154–159. PMID:
17527119.
64. Koh WJ, Lee KS, Kwon OJ, Jeong YJ, Kwak SH, Kim TS. Bilateral bronchiectasis and bronchiolitis at thin-section CT: diagnostic implications in nontuberculous mycobacterial pulmonary infection. Radiology. 2005; 235:282–288. PMID:
15703315.

65. Brenner DJ, Hall EJ. Computed tomography--an increasing source of radiation exposure. N Engl J Med. 2007; 357:2277–2284. PMID:
18046031.
66. Volpicelli G, Elbarbary M, Blaivas M, Lichtenstein DA, Mathis G, Kirkpatrick AW, Melniker L, Gargani L, Noble VE, Via G, Dean A, Tsung JW, Soldati G, Copetti R, Bouhemad B, Reissig A, Agricola E, Rouby JJ, Arbelot C, Liteplo A, Sargsyan A, Silva F, Hoppmann R, Breitkreutz R, Seibel A, Neri L, Storti E, Petrovic T; International Liaison Committee on Lung Ultrasound (ILC-LUS) for International Consensus Conference on Lung Ultrasound (ICC-LUS). International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med. 2012; 38:577–591. PMID:
22392031.

67. Chavez MA, Shams N, Ellington LE, Naithani N, Gilman RH, Steinhoff MC, Santosham M, Black RE, Price C, Gross M, Checkley W. Lung ultrasound for the diagnosis of pneumonia in adults: a systematic review and meta-analysis. Respir Res. 2014; 15:50. PMID:
24758612.

68. Reissig A, Copetti R, Mathis G, Mempel C, Schuler A, Zechner P, Aliberti S, Neumann R, Kroegel C, Hoyer H. Lung ultrasound in the diagnosis and follow-up of community-acquired pneumonia: a prospective, multicenter, diagnostic accuracy study. Chest. 2012; 142:965–972. PMID:
22700780.
69. Kearney SE, Davies CW, Davies RJ, Gleeson FV. Computed tomography and ultrasound in parapneumonic effusions and empyema. Clin Radiol. 2000; 55:542–547. PMID:
10924379.

70. Ramirez P, Torres A. Should ultrasound be included in the initial assessment of respiratory patients? Lancet Respir Med. 2014; 2:599–600. PMID:
24998675.

71. Sanyal S, Smith PR, Saha AC, Gupta S, Berkowitz L, Homel P. Initial microbiologic studies did not affect outcome in adults hospitalized with community-acquired pneumonia. Am J Respir Crit Care Med. 1999; 160:346–348. PMID:
10390423.

72. Woodhead MA, Macfarlane JT, McCracken JS, Rose DH, Finch RG. Prospective study of the aetiology and outcome of pneumonia in the community. Lancet. 1987; 1:671–674. PMID:
2882091.

73. Menéndez R, Córdoba J, de La Cuadra P, Cremades MJ, López-Hontagas JL, Salavert M, Gobernado M. Value of the polymerase chain reaction assay in noninvasive respiratory samples for diagnosis of community-acquired pneumonia. Am J Respir Crit Care Med. 1999; 159:1868–1873. PMID:
10351932.

74. Waterer GW, Wunderink RG. The influence of the severity of community-acquired pneumonia on the usefulness of blood cultures. Respir Med. 2001; 95:78–82. PMID:
11207022.

75. Campbell SG, Marrie TJ, Anstey R, Dickinson G, Ackroyd-Stolarz S. The contribution of blood cultures to the clinical management of adult patients admitted to the hospital with community-acquired pneumonia: a prospective observational study. Chest. 2003; 123:1142–1150. PMID:
12684305.
76. Jefferson H, Dalton HP, Escobar MR, Allison MJ. Transportation delay and the microbiological quality of clinical specimens. Am J Clin Pathol. 1975; 64:689–693. PMID:
1190128.

77. Kim S, Sung H, Kim DJ, Kim MN. Clinical relevance of positive NOW(TM) legionella urinary antigen test in a tertiary-care hospital in Korea. Korean J Lab Med. 2006; 26:93–97. PMID:
18156707.
78. Rosón B, Fernández-Sabé N, Carratalà J, Verdaguer R, Dorca J, Manresa F, Gudiol F. Contribution of a urinary antigen assay (Binax NOW) to the early diagnosis of pneumococcal pneumonia. Clin Infect Dis. 2004; 38:222–226. PMID:
14699454.

79. Yzerman EP, den Boer JW, Lettinga KD, Schellekens J, Dankert J, Peeters M. Sensitivity of three urinary antigen tests associated with clinical severity in a large outbreak of Legionnaires' disease in The Netherlands. J Clin Microbiol. 2002; 40:3232–3236. PMID:
12202558.

80. Arancibia F, Ewig S, Martinez JA, Ruiz M, Bauer T, Marcos MA, Mensa J, Torres A. Antimicrobial treatment failures in patients with community-acquired pneumonia: causes and prognostic implications. Am J Respir Crit Care Med. 2000; 162:154–160. PMID:
10903235.
81. van der Eerden MM, Vlaspolder F, de Graaff CS, Groot T, Jansen HM, Boersma WG. Value of intensive diagnostic microbiological investigation in low- and high-risk patients with community-acquired pneumonia. Eur J Clin Microbiol Infect Dis. 2005; 24:241–249. PMID:
15902529.

82. Domínguez J, Galí N, Blanco S, Pedroso P, Prat C, Matas L, Ausina V.Detection of
Streptococcus pneumoniae antigen by a rapid immunochromatographic assay in urine samples. Chest. 2001; 119:243–249. PMID:
11157611.
83. Gutiérrez F, Masiá M, Rodríguez JC, Ayelo A, Soldán B, Cebrián L, Mirete C, Royo G, Hidalgo AM. Evaluation of the immunochromatographic Binax NOW assay for detection of
Streptococcus pneumoniae urinary antigen in a prospective study of community-acquired pneumonia in Spain. Clin Infect Dis. 2003; 36:286–292. PMID:
12539069.
84. Murdoch DR, Laing RT, Mills GD, Karalus NC, Town GI, Mirrett S, Reller LB. Evaluation of a rapid immunochromatographic test for detection of
Streptococcus pneumoniae antigen in urine samples from adults with community-acquired pneumonia. J Clin Microbiol. 2001; 39:3495–3498. PMID:
11574562.
85. Murdoch DR, Laing RT, Cook JM. The NOW S. pneumoniae urinary antigen test positivity rate 6 weeks after pneumonia onset and among patients with COPD. Clin Infect Dis. 2003; 37:153–154. PMID:
12830428.

86. Navarro D, García-Maset L, Gimeno C, Escribano A, García-de-Lomas J; Spanish Pneumococcal Infection Study Network. Performance of the Binax NOW
Streptococcus pneumoniae urinary antigen assay for diagnosis of pneumonia in children with underlying pulmonary diseases in the absence of acute pneumococcal infection. J Clin Microbiol. 2004; 42:4853–4855. PMID:
15472361.
87. Ortega L, Sierra M, Domínguez J, Martínez J, Matas L, Bastart F, Galí N, Ausina V. Utility of a pneumonia severity index in the optimization of the diagnostic and therapeutic effort for community-acquired pneumonia. Scand J Infect Dis. 2005; 37:657–663. PMID:
16126566.

88. Smith MD, Derrington P, Evans R, Creek M, Morris R, Dance DA, Cartwright K. Rapid diagnosis of bacteremic pneumococcal infections in adults by using the Binax NOW
Streptococcus pneumoniae urinary antigen test: a prospective, controlled clinical evaluation. J Clin Microbiol. 2003; 41:2810–2813. PMID:
12843005.
89. Porcel JM, Ruiz-González A, Falguera M, Nogués A, Galindo C, Carratalá J, Esquerda A. Contribution of a pleural antigen assay (Binax NOW) to the diagnosis of pneumococcal pneumonia. Chest. 2007; 131:1442–1447. PMID:
17317736.

90. Oka H, Ueda A, Watanuki Y, Tsukiji J, Kuroda H, Akashi S, Hirai Y, Fuyuki T, Kaneko T, Ishigatsubo Y. The efficacy of high-dose penicillin for community-acquired pneumonia diagnosed by pneumococcal urine antigen test. J Infect Chemother. 2009; 15:108–112. PMID:
19396521.

91. Birtles RJ, Harrison TG, Samuel D, Taylor AG. Evaluation of urinary antigen ELISA for diagnosing Legionella pneumophila serogroup 1 infection. J Clin Pathol. 1990; 43:685–690. PMID:
2401738.

92. Helbig JH, Uldum SA, Lück PC, Harrison TG. Detection of
Legionella pneumophila antigen in urine samples by the BinaxNOW immunochromatographic assay and comparison with both Binax
Legionella urinary enzyme immunoassay (EIA) and Biotest
Legionella urin antigen EIA. J Med Microbiol. 2001; 50:509–516. PMID:
11393288.
93. Alvarez J, Domínguez A, Sabrià M, Ruiz L, Torner N, Cayla J, Barrabeig I, Sala MR, Godoy P, Camps N, Minguell S. Impact of the
Legionella urinary antigen test on epidemiological trends in community outbreaks of legionellosis in Catalonia, Spain, 1990-2004. Int J Infect Dis. 2009; 13:e365–70. PMID:
19356959.
94. Lim WS, Macfarlane JT, Boswell TC, Harrison TG, Rose D, Leinonen M, Saikku P. Study of community acquired pneumonia aetiology (SCAPA) in adults admitted to hospital: implications for management guidelines. Thorax. 2001; 56:296–301. PMID:
11254821.

95. Lee JS, Kritchevsky SB, Harris TB, Tylavsky F, Rubin SM, Newman AB. Short-term weight changes in community-dwelling older adults: the health, aging, and body composition weight change substudy. Am J Clin Nutr. 2005; 82:644–650. PMID:
16155279.

96. Afshar N, Tabas J, Afshar K, Silbergleit R. Blood cultures for community-acquired pneumonia: are they worthy of two quality measures? A systematic review. J Hosp Med. 2009; 4:112–123. PMID:
19219920.

97. Metersky ML, Ma A, Bratzler DW, Houck PM. Predicting bacteremia in patients with community-acquired pneumonia. Am J Respir Crit Care Med. 2004; 169:342–347. PMID:
14630621.

98. Labarere J, Stone RA, Obrosky DS, Yealy DM, Meehan TP, Fine JM, Graff LG, Fine MJ. Comparison of outcomes for low-risk outpatients and inpatients with pneumonia: a propensity-adjusted analysis. Chest. 2007; 131:480–488. PMID:
17296651.
99. Alikhan R, Cohen AT, Combe S, Samama MM, Desjardins L, Eldor A, Janbon C, Leizorovicz A, Olsson CG, Turpie AG. MEDENOX Study. Risk factors for venous thromboembolism in hospitalized patients with acute medical illness: analysis of the MEDENOX Study. Arch Intern Med. 2004; 164:963–968. PMID:
15136304.
100. Minogue MF, Coley CM, Fine MJ, Marrie TJ, Kapoor WN, Singer DE. Patients hospitalized after initial outpatient treatment for community-acquired pneumonia. Ann Emerg Med. 1998; 31:376–380. PMID:
9506497.

101. Neill AM, Martin IR, Weir R, Anderson R, Chereshsky A, Epton MJ, Jackson R, Schousboe M, Frampton C, Hutton S, Chambers ST, Town GI. Community acquired pneumonia: aetiology and usefulness of severity criteria on admission. Thorax. 1996; 51:1010–1016. PMID:
8977602.

102. McMahon LF Jr, Wolfe RA, Tedeschi PJ. Variation in hospital admissions among small areas. A comparison of Maine and Michigan. Med Care. 1989; 27:623–631. PMID:
2498586.

103. Labarere J, Stone RA, Scott Obrosky D, Yealy DM, Meehan TP, Auble TE, Fine JM, Graff LG, Fine MJ. Factors associated with the hospitalization of low-risk patients with community-acquired pneumonia in a cluster-randomized trial. J Gen Intern Med. 2006; 21:745–752. PMID:
16808776.

104. Song JH, Jung KS, Kang MW, Kim DJ, Pai H, Suh GY, Shim TS, Ahn JH, Ahn CM, Woo JH, Lee NY, Lee DG, Lee MS, Lee SM, Lee YS, Lee H, Chung DR. A Joint Committee for CAP Treatment Guideline. Treatment guidelines for community-acquired pneumonia in Korea: An evidence-based approach to appropriate antimicrobial therapy. Tuberc Respir Dis. 2009; 67:281–301.
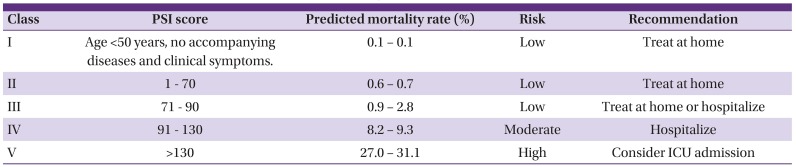
105. Fine MJ, Auble TE, Yealy DM, Hanusa BH, Weissfeld LA, Singer DE, Coley CM, Marrie TJ, Kapoor WN. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med. 1997; 336:243–250. PMID:
8995086.

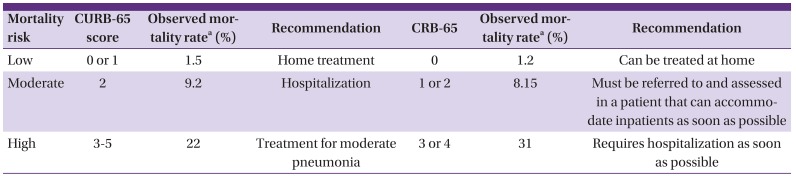
106. Lim WS, van der Eerden MM, Laing R, Boersma WG, Karalus N, Town GI, Lewis SA, Macfarlane JT. Defining community acquired pneumonia severity on presentation to hospital: an international derivation and validation study. Thorax. 2003; 58:377–382. PMID:
12728155.

107. Aujesky D, Auble TE, Yealy DM, Stone RA, Obrosky DS, Meehan TP, Graff LG, Fine JM, Fine MJ. Prospective comparison of three validated prediction rules for prognosis in community-acquired pneumonia. Am J Med. 2005; 118:384–392. PMID:
15808136.

108. Bauer TT, Ewig S, Marre R, Suttorp N, Welte T; CAPNETZ Study Group. CRB-65 predicts death from community-acquired pneumonia. J Intern Med. 2006; 260:93–101. PMID:
16789984.

109. Capelastegui A, España PP, Quintana JM, Areitio I, Gorordo I, Egurrola M, Bilbao A. Validation of a predictive rule for the management of community-acquired pneumonia. Eur Respir J. 2006; 27:151–157. PMID:
16387948.
110. España PP, Capelastegui A, Gorordo I, Esteban C, Oribe M, Ortega M, Bilbao A, Quintana JM. Development and validation of a clinical prediction rule for severe community-acquired pneumonia. Am J Respir Crit Care Med. 2006; 174:1249–1256. PMID:
16973986.

111. Barlow G, Nathwani D, Davey P.The CURB65 pneumonia severity score outperforms generic sepsis and early warning scores in predicting mortality in community-acquired pneumonia. Thorax. 2007; 62:253–259. PMID:
16928720.

112. Bont JE, Hak E, Hoes AW, Schipper M, Schellevis FG, Verheij TJ. A prediction rule for elderly primary-care patients with lower respiratory tract infections. Eur Respir J. 2007; 29:969–975. PMID:
17215313.

113. Man SY, Lee N, Ip M, Antonio GE, Chau SS, Mak P, Graham CA, Zhang M, Lui G, Chan PK, Ahuja AT, Hui DS, Sung JJ, Rainer TH. Prospective comparison of three predictive rules for assessing severity of community-acquired pneumonia in Hong Kong. Thorax. 2007; 62:348–353. PMID:
17121867.

114. Yealy DM, Auble TE, Stone RA, Lave JR, Meehan TP, Graff LG, Fine JM, Obrosky DS, Mor MK, Whittle J, Fine MJ. Effect of increasing the intensity of implementing pneumonia guidelines: a randomized, controlled trial. Ann Intern Med. 2005; 143:881–894. PMID:
16365469.
115. Atlas SJ, Benzer TI, Borowsky LH, Chang Y, Burnham DC, Metlay JP, Halm EA, Singer DE. Safely increasing the proportion of patients with community-acquired pneumonia treated as outpatients: an interventional trial. Arch Intern Med. 1998; 158:1350–1356. PMID:
9645830.
116. Marrie TJ, Lau CY, Wheeler SL, Wong CJ, Vandervoort MK, Feagan BG. A controlled trial of a critical pathway for treatment of community-acquired pneumonia. CAPITAL study investigators. Community-acquired pneumonia intervention trial assessing levofloxacin. JAMA. 2000; 283:749–755. PMID:
10683053.
117. Suchyta MR, Dean NC, Narus S, Hadlock CJ. Effects of a practice guideline for community-acquired pneumonia in an outpatient setting. Am J Med. 2001; 110:306–309. PMID:
11239849.

118. España PP, Capelastegui A, Quintana JM, Soto A, Gorordo I, García-Urbaneja M, Bilbao A. A prediction rule to identify allocation of inpatient care in community-acquired pneumonia. Eur Respir J. 2003; 21:695–701. PMID:
12762359.

119. Ananda-Rajah MR, Charles PG, Melvani S, Burrell LL, Johnson PD, Grayson ML. Comparing the pneumonia severity index with CURB-65 in patients admitted with community acquired pneumonia. Scand J Infect Dis. 2008; 40:293–300. PMID:
17918017.

120. Abisheganaden J, Ding YY, Chong WF, Heng BH, Lim TK. Predicting mortality among older adults hospitalized for community-acquired pneumonia: an enhanced confusion, urea, respiratory rate and blood pressure score compared with pneumonia severity index. Respirology. 2012; 17:969–975. PMID:
22574694.

121. Kim HI, Kim SW, Chang HH, Cha SI, Lee JH, Ki HK, Cheong HS, Yoo KH, Ryu SY, Kwon KT, Lee BK, Choo EJ, Kim DJ, Kang CI, Chung DR, Peck KR, Song JH, Suh GY, Shim TS, Kim YK, Kim HY, Moon CS, Lee HK, Park SY, Oh JY, Jung SI, Park KH, Yun NR, Yoon SH, Sohn KM, Kim YS, Jung KS. Mortality of community-acquired pneumonia in Korea: assessed with the pneumonia severity index and the CURB-65 score. J Korean Med Sci. 2013; 28:1276–1282. PMID:
24015030.

122. Lee JC, Hwang HJ, Park YH, Joe JH, Chung JH, Kim SH. Comparison of severity predictive rules for hospitalised nursing home-acquired pneumonia in Korea: a retrospective observational study. Prim Care Respir J. 2013; 22:149–154. PMID:
23494188.

123. Niederman MS, Mandell LA, Anzueto A, Bass JB, Broughton WA, Campbell GD, Dean N, File T, Fine MJ, Gross PA, Martinez F, Marrie TJ, Plouffe JF, Ramirez J, Sarosi GA, Torres A, Wilson R, Yu VL; American Thoracic Society. Guidelines for the management of adults with community-acquired pneumonia. Diagnosis, assessment of severity, antimicrobial therapy, and prevention. Am J Respir Crit Care Med. 2001; 163:1730–1754. PMID:
11401897.
124. Chalmers JD, Taylor JK, Mandal P, Choudhury G, Singanayagam A, Akram AR, Hill AT. Validation of the Infectious Diseases Society of America/American Thoratic Society minor criteria for intensive care unit admission in community-acquired pneumonia patients without major criteria or contraindications to intensive care unit care. Clin Infect Dis. 2011; 53:503–511. PMID:
21865188.

125. Marti C, Garin N, Grosgurin O, Poncet A, Combescure C, Carballo S, Perrier A. Prediction of severe community-acquired pneumonia: a systematic review and meta-analysis. Crit Care. 2012; 16:R141. PMID:
22839689.

126. Salih W, Schembri S, Chalmers JD. Simplification of the IDSA/ATS criteria for severe CAP using meta-analysis and observational data. Eur Respir J. 2014; 43:842–851. PMID:
24114960.

127. Charles PG, Wolfe R, Whitby M, Fine MJ, Fuller AJ, Stirling R, Wright AA, Ramirez JA, Christiansen KJ, Waterer GW, Pierce RJ, Armstrong JG, Korman TM, Holmes P, Obrosky DS, Peyrani P, Johnson B, Hooy M; Australian Community-Acquired Pneumonia Study Collaboration. Grayson ML. SMART-COP: a tool for predicting the need for intensive respiratory or vasopressor support in community-acquired pneumonia. Clin Infect Dis. 2008; 47:375–384. PMID:
18558884.
128. Yandiola PPE, Capelastegui A, Quintana J, Diez R, Gorordo I, Bilbao A, Zalacain R, Menendez R, Torres A. Prospective comparison of severity scores for predicting clinically relevant outcomes for patients hospitalized with community-acquired pneumonia. Chest. 2009; 135:1572–1579. PMID:
19141524.

129. Arnold FW, Summersgill JT, Lajoie AS, Peyrani P, Marrie TJ, Rossi P, Blasi F, Fernandez P, File TM Jr, Rello J, Menendez R, Marzoratti L, Luna CM, Ramirez JA; Community-Acquired Pneumonia Organization (CAPO) Investigators. A worldwide perspective of atypical pathogens in community-acquired pneumonia. Am J Respir Crit Care Med. 2007; 175:1086–1093. PMID:
17332485.

130. Mills GD, Oehley MR, Arrol B. Effectiveness of beta lactam antibiotics compared with antibiotics active against atypical pathogens in non-severe community acquired pneumonia: meta-analysis. BMJ. 2005; 330:456. PMID:
15684024.
131. Eliakim-Raz N, Robenshtok E, Shefet D, Gafter-Gvili A, Vidal L, Paul M, Leibovici L. Empiric antibiotic coverage of atypical pathogens for community-acquired pneumonia in hospitalized adults. Cochrane Database Syst Rev. 2012; (9):CD004418. PMID:
22972070.

132. Postma DF, van Werkhoven CH, van Elden LJ, Thijsen SF, Hoepelman AI, Kluytmans JA, Boersma WG, Compaijen CJ, van der Wall E, Prins JM, Oosterheert JJ, Bonten MJ; CAP-START Study Group. Antibiotic treatment strategies for community-acquired pneumonia in adults. N Engl J Med. 2015; 372:1312–1323. PMID:
25830421.

133. Lee MY, Ko KS, Oh WS, Park S, Lee JY, Baek JY, Suh JY, Peck KR, Lee NY, Song JH. In vitro activity of cefditoren: antimicrobial efficacy against major respiratory pathogens from Asian countries. Int J Antimicrob Agents. 2006; 28:14–18. PMID:
16777383.

134. Yu VL, Chiou CC, Feldman C, Ortqvist A, Rello J, Morris AJ, Baddour LM, Luna CM, Snydman DR, Ip M, Ko WC, Chedid MB, Andremont A, Klugman KP; International Pneumococcal Study Group. An international prospective study of pneumococcal bacteremia: correlation with in vitro resistance, antibiotics administered, and clinical outcome. Clin Infect Dis. 2003; 37:230–237. PMID:
12856216.

135. Noreddin AM, Marras TK, Sanders K, Chan CK, Hoban DJ, Zhanel GG. Pharmacodynamic target attainment analysis against
Streptococcus pneumoniae using levofloxacin 500 mg, 750 mg and 1000 mg once daily in plasma (P) and epithelial lining fluid (ELF) of hospitalized patients with community acquired pneumonia (CAP). Int J Antimicrob Agents. 2004; 24:479–484. PMID:
15519481.
136. File TM Jr, Milkovich G, Tennenberg AM, Xiang JX, Khashab MM, Zadeikis N. Clinical implications of 750 mg, 5-day levofloxacin for the treatment of community-acquired pneumonia. Curr Med Res Opin. 2004; 20:1473–1481. PMID:
15383197.
137. File TM Jr, Mandell LA, Tillotson G, Kostov K, Georgiev O. Gemifloxacin once daily for 5 days versus 7 days for the treatment of community-acquired pneumonia: a randomized, multicentre, double-blind study. J Antimicrob Chemother. 2007; 60:112–120. PMID:
17537866.

138. Gleason PP, Meehan TP, Fine JM, Galusha DH, Fine MJ. Associations between initial antimicrobial therapy and medical outcomes for hospitalized elderly patients with pneumonia. Arch Intern Med. 1999; 159:2562–2572. PMID:
10573046.

139. Houck PM, MacLehose RF, Niederman MS, Lowery JK. Empiric antibiotic therapy and mortality among medicare pneumonia inpatients in 10 western states: 1993, 1995, and 1997. Chest. 2001; 119:1420–1426. PMID:
11348948.
140. Dudas V, Hopefl A, Jacobs R, Guqlielmo BJ. Antimicrobial selection for hospitalized patients with presumed community-acquired pneumonia: a survey of nonteaching US community hospitals. Ann Pharmacother. 2000; 34:446–452. PMID:
10772428.

141. Brown RB, Iannini P, Gross P, Kunkel M. Impact of initial antibiotic choice on clinical outcomes in community-acquired pneumonia: analysis of a hospital claims-made database. Chest. 2003; 123:1503–1511. PMID:
12740267.
142. Garin N, Genné D, Carballo S, Chuard C, Eich G, Hugli O, Lamy O, Nendaz M, Petignat PA, Perneger T, Rutschmann O, Seravalli L, Harbarth S, Perrier A. β-lactam monotherapy vs β-lactam-macrolide combination treatment in moderately severe community-acquired pneumonia: a randomized noninferiority trial. JAMA Intern Med. 2014; 174:1894–1901. PMID:
25286173.
143. Woodhead M, Blasi F, Ewig S, Garau J, Huchon G, Ieven M, Ortqvist A, Schaberg T, Torres A, van der Heijden G, Read R, Verheij TJ; Joint Taskforce of the European Respiratory Society and European Society for Clinical Microbiology and Infectious Diseases. Guidelines for the management of adult lower respiratory tract infections--full version. Clin Microbiol Infect. 2011; 17(Suppl 6):E1–59.
144. Paul M, Nielsen AD, Gafter-Gvili A, Tacconelli E, Andreassen S, Almanasreh N, Goldberg E, Cauda R, Frank U, Leibovici L. The need for macrolides in hospitalised community-acquired pneumonia: propensity analysis. Eur Respir J. 2007; 30:525–531. PMID:
17537772.

145. Ray WA, Murray KT, Hall K, Arboqast PG, Stein CM. Azithromycin and the risk of cardiovascular death. N Engl J Med. 2012; 366:1881–1890. PMID:
22591294.

146. Trac MH, McArthur E, Jandoc R, Dixon SN, Nash DM, Hackam DG, Garg AX. Macrolide antibiotics and the risk of ventricular arrhythmia in older adults. CMAJ. 2016; 188:E120–9. PMID:
26903359.

147. Chang KC, Leung CC, Yew WW, Lau TY, Leung WM, Tam CM, Lam HC, Tse PS, Wong MY, Lee SN, Wat KI, Ma YH. Newer fluoroquinolones for treating respiratory infection: do they mask tuberculosis? Eur Respir J. 2010; 35:606–613. PMID:
19717477.

148. Chen TC, Lu PL, Lin CY, Lin WR, Chen YH. Fluoroquinolones are associated with delayed treatment and resistance in tuberculosis: a systematic review and meta-analysis. Int J Infect Dis. 2011; 15:e211–6. PMID:
21195001.

149. Wang JY, Hsueh PR, Jan IS, Lee LN, Liaw YS, Yang PC, Luh KT. Empirical treatment with a fluoroquinolone delays the treatment for tuberculosis and is associated with a poor prognosis in endemic areas. Thorax. 2006; 61:903–908. PMID:
16809417.

150. Torres A, Garau J, Arvis P, Carlet J, Choudhri S, Kureishi A, Le Berre MA, Lode H, Winter J, Read RC. MOTIV (MOxifloxacin Treatment IV) Study Group. Moxifloxacin monotherapy is effective in hospitalized patients with community-acquired pneumonia: the MOTIV study--a randomized clinical trial. Clin Infect Dis. 2008; 46:1499–1509. PMID:
18419482.
151. Bhavnani SM, Ambrose PG. Cost-effectiveness of oral gemifloxacin versus intravenous ceftriaxone followed by oral cefuroxime with/without a macrolide for the treatment of hospitalized patients with community-acquired pneumonia. Diagn Microbiol Infect Dis. 2008; 60:59–64. PMID:
17889491.

152. Woodhead M, Blasi F, Ewig S, Huchon G, Ieven M, Ortqvist A, Schaberg T, Torres A, van der Heijden G, Verheij TJ; European Respiratory Society. European Society of Clinical Microbiology and Infectious Diseases. Guidelines for the management of adult lower respiratory tract infections. Eur respir J. 2005; 26:1138–1180. PMID:
16319346.
153. Rizzato G, Montemurro L, Fraioli P, Montanari G, Fanti D, Pozzoli R, Magliano E. Efficacy of a three day course of azithromycin in moderately severe community-acquired pneumonia. Eur respir J. 1995; 8:398–402. PMID:
7789484.

154. Dunbar LM, Wunderink RG, Habib MP, Smith LG, Tennenberg AM, Khashab MM, Wiesinger BA, Xiang JX, Zadeikis N, Kahn JB. High-dose, short-course levofloxacin for community-acquired pneumonia: a new treatment paradigm. Clin Infect Dis. 2003; 37:752–760. PMID:
12955634.

155. Schönwald S, Skerk V, Petricevic I, Car V, Majerus-Misic L, Gunjaca M. Comparison of three-day and five-day courses of azithromycin in the treatment of atypical pneumonia. Eur J Clin Microbiol Infect Dis. 1991; 10:877–880. PMID:
1662637.

156. Ramirez JA. Switch therapy in community-acquired pneumonia. Diagn Microbiol Infect Dis. 1995; 22:219–223. PMID:
7587043.
157. Ramirez JA, Vargas S, Ritter GW, Brier ME, Wright A, Smith S, Newman D, Burke J, Mushtaq M, Huang A. Early switch from intravenous to oral antibiotics and early hospital discharge: a prospective observational study of 200 consecutive patients with community-acquired pneumonia. Arch Intern Med. 1999; 159:2449–2454. PMID:
10665893.
158. Ramirez JA, Bordon J. Early switch from intravenous to oral antibiotics in hospitalized patients with bacteremic community-acquired
Streptococcus pneumoniae pneumonia. Arch Intern Med. 2001; 161:848–850. PMID:
11268227.
159. Castro-Guardiola A, Viejo-Rodríguez AL, Soler-Simon S, Armengou-Arxé A, Bisbe-Company V, Peñarroja-Matutano G, Bisbe-Company J, García-Bragado F. Efficacy and safety of oral and early-switch therapy for community-acquired pneumonia: a randomized controlled trial. Am J Med. 2001; 111:367–374. PMID:
11583639.

160. Nathan RV, Rhew DC, Murray C, Bratzler DW, Houck PM, Weingarten SR. In-hospital observation after antibiotic switch in pneumonia: a national evaluation. Am J Med. 2006; 119:512.e1–512.e7. PMID:
16750965.

161. Rhew DC, Riedinger MS, Sandhu M, Bowers C, Greengold N, Weingarten SR. A prospective, multicenter study of a pneumonia practice guideline. Chest. 1998; 114:115–119. PMID:
9674457.

162. Halm EA, Fine MJ, Kapoor WN, Singer DE, Marrie TJ, Siu AL. Instability on hospital discharge and the risk of adverse outcomes in patients with pneumonia. Arch Intern Medicine. 2002; 162:1278–1284.

163. Capelastegui A, España PP, Bilbao A, Martinez-Vazquez M, Gorordo I, Oribe M, Urrutia I, Quintana JM. Pneumonia: criteria for patient instability on hospital discharge. Chest. 2008; 134:595–600. PMID:
18490403.
164. Halm EA, Fine MJ, Marrie TJ, Coley CM, Kapoor WN, Obrosky DS, Singer DE. Time to clinical stability in patients hospitalized with community-acquired pneumonia: implications for practice guidelines. JAMA. 1998; 279:1452–1457. PMID:
9600479.
165. O'Driscoll BR, Howard LS, Davison AG. BTS guideline for emergency oxygen use in adult patients. Thorax. 2008; 63(Suppl 6):vi1–68. PMID:
18838559.
166. Siempos II, Vardakas KZ, Kopterides P, Falagas ME. Adjunctive therapies for community-acquired pneumonia: a systematic review. J Antimicrob Chemother. 2008; 62:661–668. PMID:
18641037.

167. Mismetti P, Laporte-Simitsidis S, Tardy B, Cucherat M, Buchmüller A, Juillard-Delsart D, Decousus H. Prevention of venous thromboembolism in internal medicine with unfractionated or low-molecular-weight heparins: a meta-analysis of randomised clinical trials. Thromb Haemost. 2000; 83:14–19. PMID:
10669147.

168. Dean NC, Silver MP, Bateman KA, James B, Hadlock CJ, Hale D. Decreased mortality after implementation of a treatment guideline for community-acquired pneumonia. Am J Med. 2001; 110:451–457. PMID:
11331056.

169. Mundy LM, Leet TL, Darst K, Schnitzler MA, Dunagan WC. Early mobilization of patients hospitalized with community-acquired pneumonia. Chest. 2003; 124:883–889. PMID:
12970012.

170. File TM. Community-acquired pneumonia. Lancet. 2003; 362:1991–2001. PMID:
14683661.

171. Torres A, Serra-Batlles J, Ferrer A, Jiménez P, Celis R, Cobo E, Rodriguez-Roisin R.Severe community-acquired pneumonia. Epidemiology and prognostic factors. Am Rev Respir Dis. 1991; 144:312–318. PMID:
1859053.

172. Briones ML, Blanquer J, Ferrando D, Blasco ML, Gimeno C, Marín J. Assessment of analysis of urinary pneumococcal antigen by immunochromatography for etiologic diagnosis of community-acquired pneumonia in adults. Clin Vaccine Immunol. 2006; 13:1092–1097. PMID:
17028212.

173. Leroy O, Saux P, Bédos JP, Caulin E. Comparison of levofloxacin and cefotaxime combined with ofloxacin for ICU patients with community-acquired pneumonia who do not require vasopressors. Chest. 2005; 128:172–183. PMID:
16002932.

174. Raz-Pasteur A, Shasha D, Paul M. Fluoroquinolones or macrolides alone versus combined with beta-lactams for adults with community-acquired pneumonia: systematic review and meta-analysis. Int J Antimicrob Agents. 2015; 46:242–248. PMID:
26092096.
175. Lodise TP, Kwa A, Cosler L, Gupta R, Smith RP. Comparison of beta-lactam and macrolide combination therapy versus fluoroquinolone monotherapy in hospitalized Veterans Affairs patients with community-acquired pneumonia. Antimicrob Agents Chemother. 2007; 51:3977–3982. PMID:
17709460.
176. Baddour LM, Yu VL, Klugman KP, Feldman C, Ortqvist A, Rello J, Morris AJ, Luna CM, Snydman DR, Ko WC, Chedid MB, Hui DS, Andremont A, Chiou CC; International Pneumococcal Study Group. Combination antibiotic therapy lowers mortality among severely ill patients with pneumococcal bacteremia. Am J Respir Crit Care Med. 2004; 170:440–444. PMID:
15184200.

177. Martínez JA, Horcajada JP, Almela M, Marco F, Soriano A, García E, Marco MA, Torres A, Mensa J. Addition of a macrolide to a beta-lactam-based empirical antibiotic regimen is associated with lower in-hospital mortality for patients with bacteremic pneumococcal pneumonia. Clin Infect Dis. 2003; 36:389–395. PMID:
12567294.
178. Waterer GW, Somes GW, Wunderink RG. Monotherapy may be suboptimal for severe bacteremic pneumococcal pneumonia. Arch Intern Med. 2001; 161:1837–1842. PMID:
11493124.

179. Weiss K, Low DE, Cortes L, Beaupre A, Gauthier R, Gregoire P, Legare M, Nepveu F, Thibert D, Tremblay C, Tremblay J. Clinical characteristics at initial presentation and impact of dual therapy on the outcome of bacteremic
Streptococcus pneumoniae pneumonia in adults. Can Respir J. 2004; 11:589–593. PMID:
15611810.
180. Arancibia F, Bauer TT, Ewig S, Mensa J, Gonzalez J, Niederman MS, Torres A. Community-acquired pneumonia due to gram-negative bacteria and Pseudomonas aeruginosa: incidence, risk, and prognosis. Arch Intern Med. 2002; 162:1849–1858. PMID:
12196083.
181. Kang CI, Song JH, Oh WS, Ko KS, Chung DR, Peck KR; Asian Network for Surveillance of Resistant Pathogens (ANSORP) Study Group. Clinical outcomes and risk factors of community-acquired pneumonia caused by gram-negative bacilli. Eur J Clin Microbiol Infect Dis. 2008; 27:657–661. PMID:
18317821.

182. Kollef MH, Sherman G, Ward S, Fraser VJ. Inadequate antimicrobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest. 1999; 115:462–474. PMID:
10027448.
183. Murray RJ, Robinson JO, White JN, Hughes F, Coombs GW, Pearson JC, Tan HL, Chidlow G, Williams S, Christiansen KJ, Smith DW. Community-acquired pneumonia due to pandemic A(H1N1)2009 influenza virus and methicillin resistant
Staphylococcus aureus co-infection. PLoS One. 2010; 5:e8705. PMID:
20090931.
184. Bernardo K, Pakulat N, Fleer S, Schnaith A, Utermöhlen O, Krut O, Müller S, Krönke M. Subinhibitory concentrations of linezolid reduce
Staphylococcus aureus virulence factor expression. Antimicrob Agents Chemother. 2004; 48:546–555. PMID:
14742208.
185. Micek ST, Dunne M, Kollef MH. Pleuropulmonary complications of Panton-Valentine leukocidin-positive community-acquired methicillin-resistant
Staphylococcus aureus: importance of treatment with antimicrobials inhibiting exotoxin production. Chest. 2005; 128:2732–2738. PMID:
16236949.
186. Carpenter CF, Chambers HF. Daptomycin: another novel agent for treating infections due to drug-resistant gram-positive pathogens. Clin Infect Dis. 2004; 38:994–1000. PMID:
15034832.

187. Falagas ME, Karageorgopoulos DE, Dimopoulos G. Clinical significance of the pharmacokinetic and pharmacodynamic characteristics of tigecycline. Curr Drug Metab. 2009; 10:13–21. PMID:
19149509.

188. Ruiz M, Ewig S, Torres A, Arancibia F, Marco F, Mensa J, Sanchez M, Martinez JA. Severe community-acquired pneumonia. Risk factors and follow-up epidemiology. Am J Respir Crit Care Med. 1999; 160:923–929. PMID:
10471620.
189. Snijders D, Daniels JM, de Graaff CS, van der Werf TS, Boersma WG. Efficacy of corticosteroids in community-acquired pneumonia: a randomized double-blinded clinical trial. Am J Respir Crit Care Med. 2010; 181:975–982. PMID:
20133929.
190. Meijvis SC, Hardeman H, Remmelts HH, Heijligenberg R, Rijkers GT, van Velzen-Blad H, Voorn GP, van de Garde EM, Endeman H, Grutters JC, Bos WJ, Biesma DH. Dexamethasone and length of hospital stay in patients with community-acquired pneumonia: a randomised, double-blind, placebo-controlled trial. Lancet. 2011; 377:2023–2030. PMID:
21636122.

191. Torres A, Sibila O, Ferrer M, Polverino E, Menendez R, Mensa J, Gabarrús A, Sellarés J, Restrepo MI, Anzueto A, Niederman MS, Agustí C. Effect of corticosteroids on treatment failure among hospitalized patients with severe community-acquired pneumonia and high inflammatory response: a randomized clinical trial. JAMA. 2015; 313:677–686. PMID:
25688779.

192. Blum CA, Nigro N, Briel M, Schuetz P, Ullmer E, Suter-Widmer I, Winzeler B, Bingisser R, Elsaesser H, Drozdov D, Arici B, Urwyler SA, Refardt J, Tarr P, Wirz S, Thomann R, Baumgartner C, Duplain H, Burki D, Zimmerli W, Rodondi N, Mueller B, Christ-Crain M. Adjunct prednisone therapy for patients with community-acquired pneumonia: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2015; 385:1511–1518. PMID:
25608756.

193. Confalonieri M, Urbino R, Potena A, Piattella M, Parigi P, Puccio G, Della Porta R, Giorgio C, Blasi F, Umberger R, Meduri GU. Hydrocortisone infusion for severe community-acquired pneumonia: a preliminary randomized study. Am J Respir Crit Care Med. 2005; 171:242–248. PMID:
15557131.
194. Marik P, Kraus P, Sribante J, Havlik I, Lipman J, Johnson DW. Hydrocortisone and tumor necrosis factor in severe community-acquired pneumonia. A randomized controlled study. Chest. 1993; 104:389–392. PMID:
8339624.
195. Montón C, Ewig S, Torres A, El-Ebiary M, Filella X, Rañó A, Xaubet A. Role of glucocorticoids on inflammatory response in nonimmunosuppressed patients with pneumonia: a pilot study. Eur Respir J. 1999; 14:218–220. PMID:
10489855.

196. Annane D, Sébille V, Charpentier C, Bollaert PE, François B, Korach JM, Capellier G, Cohen Y, Azoulay E, Troché G, Chaumet-Riffaud P, Bellissant E. Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA. 2002; 288:862–871. PMID:
12186604.

197. Annane D, Sébille V, Bellissant E. Ger-Inf-05 Study G. Effect of low doses of corticosteroids in septic shock patients with or without early acute respiratory distress syndrome. Crit Care Med. 2006; 34:22–30. PMID:
16374152.
198. Bruns AH, Oosterheert JJ, Prokop M, Lammers JW, Hak E, Hoepelman AI. Patterns of resolution of chest radiograph abnormalities in adults hospitalized with severe community-acquired pneumonia. Clin Infec Dis. 2007; 45:983–991. PMID:
17879912.

199. Mittl RL Jr, Schwab RJ, Duchin JS, Goin JE, Albeida SM, Miller WT. Radiographic resolution of community-acquired pneumonia. Am J Respir Crit Care Med. 1994; 149:630–635. PMID:
8118630.

200. El Solh AA, Aquilina AT, Gunen H, Ramadan F. Radiographic resolution of community-acquired bacterial pneumonia in the elderly. J Am Geriatr Soc. 2004; 52:224–229. PMID:
14728631.

201. Tang KL, Eurich DT, Minhas-Sandhu JK, Marrie TJ, Majumdar SR. Incidence, correlates, and chest radiographic yield of new lung cancer diagnosis in 3398 patients with pneumonia. Arch Intern Med. 2011; 171:1193–1198. PMID:
21518934.

202. Holmberg H, Kragsbjerg P. Association of pneumonia and lung cancer: the value of convalescent chest radiography and follow-up. Scand J Infect Dis. 1993; 25:93–100. PMID:
8460356.

203. Menéndez R, Cavalcanti M, Reyes S, Mensa J, Martinez R, Marcos MA, Filella X, Niederman M, Torres A. Markers of treatment failure in hospitalised community acquired pneumonia. Thorax. 2008; 63:447–452. PMID:
18245147.

204. Chalmers JD, Singanayagam A, Hill AT. C-reactive protein is an independent predictor of severity in community-acquired pneumonia. Am J Med. 2008; 121:219–225. PMID:
18328306.

205. Menéndez R, Martinez R, Reyes S, Mensa J, Polverino E, Filella X, Esquinas C, Martinez A, Ramirez P, Torres A.Stability in community-acquired pneumonia: one step forward with markers? Thorax. 2009; 64:987–992. PMID:
19762338.

206. Coelho LM, Salluh JI, Soares M, Bozza FA, Verdeal JC, Castro-Faria-Neto HC, Lapa e Silva JR, Bozza PT, Póvoa P. Patterns of c-reactive protein RATIO response in severe community-acquired pneumonia: a cohort study. Crit Care. 2012; 16:R53. PMID:
22449513.

207. Hohenthal U, Hurme S, Helenius H, Heiro M, Meurman O, Nikoskelainen J, Kotilainen P.Utility of C-reactive protein in assessing the disease severity and complications of community-acquired pneumonia. Clin Microbiol Infect. 2009; 15:1026–1032. PMID:
19548923.

208. Boussekey N, Leroy O, Alfandari S, Devos P, Georges H, Guery B. Procalcitonin kinetics in the prognosis of severe community-acquired pneumonia. Intensive Care Med. 2006; 32:469–472. PMID:
16477418.

209. Lacoma A, Rodríguez N, Prat C, Ruiz-Manzano J, Andreo F, Ramírez A, Bas A, Pérez M, Ausina V, Domínguez J. Usefulness of consecutive biomarkers measurement in the management of community-acquired pneumonia. Eur J Clin Microbiol Infect Dis. 2012; 31:825–833. PMID:
21870054.

210. Schuetz P, Wirz Y, Sager R, Christ-Crain M, Stolz D, Tamm M, Bouadma L, Luyt CE, Wolff M, Chastre J, Tubach F, Kristoffersen KB, Burkhardt O, Welte T, Schroeder S, Nobre V, Wei L, Bucher HC, Bhatnagar N, Annane D, Reinhart K, Branche A, Damas P, Nijsten M, de Lange DW, Deliberato RO, Lima SS, Maravić-Stojković V, Verduri A, Cao B, Shehabi Y, Beishuizen A, Jensen JS, Corti C, Van Oers JA, Falsey AR, de Jong E, Oliveira CF, Beghe B, Briel M, Mueller B. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev. 2017; CD007498. PMID:
29025194.

211. Schuetz P, Briel M, Mueller B. Clinical outcomes associated with procalcitonin algorithms to guide antibiotic therapy in respiratory tract infections. JAMA. 2013; 309:717–718. PMID:
23423417.

212. de Jong E, van Oers JA, Beishuizen A, Vos P, Vermeijden WJ, Haas LE, Loef BG, Dormans T, van Melsen GC, Kluiters YC, Kemperman H, van den Elsen MJ, Schouten JA, Streefkerk JO, Krabbe HG, Kieft H, Kluge GH, van Dam VC, van Pelt J, Bormans L, Otten MB, Reidinga AC, Endeman H, Twisk JW, van de Garde EMW, de Smet AMGA, Kesecioglu J, Girbes AR, Nijsten MW, de Lange DW. Efficacy and safety of procalcitonin guidance in reducing the duration of antibiotic treatment in critically ill patients: a randomised, controlled, open-label trial. Lancet Infect Dis. 2016; 16:819–827. PMID:
26947523.

213. Butler JC, Breiman RF, Campbell JF, Lipman HB, Broome CV, Facklam RR. Pneumococcal polysaccharide vaccine efficacy: an evaluation of current recommendations. JAMA. 1993; 270:1826–1831. PMID:
8411526.

214. Shapiro ED, Berg AT, Austrian R, Schroeder D, Parcells V, Margolis A, Adair RK, Clemens JD. The protective efficacy of polyvalent pneumococcal polysaccharide vaccine. N Engl J Med. 1991; 325:1453–1460. PMID:
1944423.

215. Moberley S, Holden J, Tatham DP, Andrews RM. Vaccines for preventing pneumococcal infection in adults. Cochrane Database Syst Rev. 2008; CD000422. PMID:
18253977.

216. Maruyama T, Taguchi O, Niederman MS, Morser J, Kobayashi H, Kobayashi T, D'Alessandro-Gabazza C, Nakayama S, Nishikubo K, Noguchi T, Takei Y, Gabazza EC. Efficacy of 23-valent pneumococcal vaccine in preventing pneumonia and improving survival in nursing home residents: double blind, randomised and placebo controlled trial. BMJ. 2010; 340:c1004. PMID:
20211953.

217. Christenson B, Hedlund J, Lundbergh P, Ortqvist A. Additive preventive effect of influenza and pneumococcal vaccines in elderly persons. Eur Respir J. 2004; 23:363–368. PMID:
15065822.

218. Vila-Córcoles A, Ochoa-Gondar O, Hospital I, Ansa X, Vilanova A, Rodríguez T, Llor C; EVAN Study Group. Protective effects of the 23-valent pneumococcal polysaccharide vaccine in the elderly population: the EVAN-65 study. Clin Infect Dis. 2006; 43:860–868. PMID:
16941367.

219. Christenson B, Pauksen K, Sylvan SP. Effect of influenza and pneumococcal vaccines in elderly persons in years of low influenza activity. Virol J. 2008; 5:52. PMID:
18442371.

220. Fisman DN, Abrutyn E, Spaude KA, Kim A, Kirchner C, Daley J. Prior pneumococcal vaccination is associated with reduced death, complications, and length of stay among hospitalized adults with community-acquired pneumonia. Clin Infect Dis. 2006; 42:1093–1101. PMID:
16575726.

221. Mykietiuk A, Carratalà J, Domínguez A, Manzur A, Fernández-Sabé N, Dorca J, Tubau F, Manresa F, Gudiol F. Effect of prior pneumococcal vaccination on clinical outcome of hospitalized adults with community-acquired pneumococcal pneumonia. Eur J Clin Microbiol Infect Dis. 2006; 25:457–462. PMID:
16773389.

222. Jackson LA, Neuzil KM, Yu O, Benson P, Barlow WE, Adams AL, Hanson CA, Mahoney LD, Shay DK, Thompson WW. Vaccine Safety Datalink. Effectiveness of pneumococcal polysaccharide vaccine in older adults. N Engl J Med. 2003; 348:1747–1755. PMID:
12724480.
223. Skull SA, Andrews RM, Byrnes GB, Kelly HA, Nolan TM, Brown GV, Campbell DA. Prevention of community-acquired pneumonia among a cohort of hospitalized elderly: benefit due to influenza and pneumococcal vaccination not demonstrated. Vaccine. 2007; 25:4631–4640. PMID:
17478015.

224. Jackson LA, Neuzil KM, Whitney CG, Starkovich P, Dunstan M, Yu O, Nelson JC, Feikin DR, Shay DK, Baggs J, Carste B, Nahm MH, Carlone G. Safety of varying dosages of 7-valent pneumococcal protein conjugate vaccine in seniors previously vaccinated with 23-valent pneumococcal polysaccharide vaccine. Vaccine. 2005; 23:3697–3703. PMID:
15882530.

225. Törling J, Hedlund J, Konradsen HB, Ortqvist A. Revaccination with the 23-valent pneumococcal polysaccharide vaccine in middle-aged and elderly persons previously treated for pneumonia. Vaccine. 2003; 22:96–103. PMID:
14604576.

226. Waites KB, Canupp KC, Chen YY, DeVivo MJ, Nahm MH. Revaccination of adults with spinal cord injury using the 23-valent pneumococcal polysaccharide vaccine. J Spinal Cord Med. 2008; 31:53–59. PMID:
18533412.

227. Nuorti JP, Butler JC, Farley MM, Harrison LH, McGeer A, Kolczak MS, Breiman RF. Cigarette smoking and invasive pneumococcal disease. Active Bacterial Core Surveillance Team. N Engl J Med. 2000; 342:681–689. PMID:
10706897.
228. Almirall J, Bolíbar I, Serra-Prat M, Roig J, Hospital I, Carandell E, Agustí M, Ayuso P, Estela A, Torres A; Community-Acquired Pneumonia in Catalan Countries (PACAP) Study Group. New evidence of risk factors for community-acquired pneumonia: a population-based study. Eur Respira J. 2008; 31:1274–1284.

229. Baik I, Curhan GC, Rimm EB, Bendich A, Willett WC, Fawzi WW. A prospective study of age and lifestyle factors in relation to community-acquired pneumonia in US men and women. Arch Intern Med. 2000; 160:3082–3088. PMID:
11074737.

230. Marston BJ, Lipman HB, Breiman RF. Surveillance for Legionnaires' disease: risk factors for morbidity and mortality. Arch Intern Med. 1994; 154:2417–2422. PMID:
7979837.

231. West R, McNeill A, Raw M. Smoking cessation guidelines for health professionals: an update. Thorax. 2000; 55:987–999. PMID:
11083883.









 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download