Abstract
Background
From May to July 2015, the Republic of Korea experienced the largest outbreak of Middle East respiratory syndrome (MERS) outside the Arabian Peninsula. A total of 186 patients, including 36 deaths, had been diagnosed with MERS-coronavirus (MERS-CoV) infection as of September 30th, 2015.
Materials and Methods
We obtained information of patients who were confirmed to have MERS-CoV infection. MERS-CoV infection was diagnosed using real-time reverse-transcriptase polymerase chain reaction assay.
Results
The median age of the patients was 55 years (range, 16 to 86). A total of 55.4% of the patients had one or more coexisting medical conditions. The most common symptom was fever (95.2%). At admission, leukopenia (42.6%), thrombocytopenia (46.6%), and elevation of aspartate aminotransferase (42.7%) were observed. Pneumonia was detected in 68.3% of patients at admission and developed in 80.8% during the disease course. Antiviral agents were used for 74.7% of patients. Mechanical ventilation, extracorporeal membrane oxygenation, and convalescent serum were employed for 24.5%, 7.1%, and 3.8% of patients, respectively. Older age, presence of coexisting medical conditions including diabetes or chronic lung disease, presence of dyspnea, hypotension, and leukocytosis at admission, and the use of mechanical ventilation were revealed to be independent predictors of death.
Conclusion
The clinical features of MERS-CoV infection in the Republic of Korea were similar to those of previous outbreaks in the Middle East. However, the overall mortality rate (20.4%) was lower than that in previous reports. Enhanced surveillance and active management of patients during the outbreak may have resulted in improved outcomes.
Middle East respiratory syndrome (MERS), which is caused by β-coronavirus of the C lineage (MERS-CoV), ranges in severity from asymptomatic to a severe respiratory illness with rapid progression to respiratory failure [1]. Since the identification of the first case in Saudi Arabia, the World Health Organization (WHO) has been notified of 1,638 laboratory-confirmed cases, including 587 fatal cases, in 26 countries as of February 26th, 2016. These confirmed cases have been directly or indirectly linked to countries in the Arabian Peninsula [2]. Although the exact mode of transmission remains unknown, primary MERS-CoV infections are presumably associated with community zoonotic exposure and may be passed to family members via limited secondary transmission [3]. Human-to-human spread of MERS-CoV is assumed to occur through close contact, such as caring for or living with an infected person [45]. As infected people have spread MERS-CoV to others in hospitals, the importance of vigilant surveillance and appropriate infection control measures must be emphasised to prevent transmission in healthcare settings.
From May to July 2015, the Republic of Korea experienced the largest outbreak of MERS outside the Arabian Peninsula. Infection was confirmed in a 68-year-old businessman returning from the Middle East on May 20th, 2015, when he presented with atypical pneumonia and he failed to response to anti-microbial therapy for community-acquired pneumonia. Prior to the confirmation of MERS-CoV infection, he had visited three hospitals where more than 600 people (including other patients, care givers, and healthcare workers) were exposed, resulting in 36 cases of MERS-CoV infection [6]. As of December 5th, 2015, 16,693 individuals had been quarantined and 186 cases of MERS-CoV had been confirmed. All confirmed cases were associated with a total of 17 healthcare facilities, and approximately 80% of the confirmed cases were caused by 'super-spreading' events that originated from five patients in four hospitals [6]. We aimed to determine the clinical features and outcomes of Korean patients confirmed to be infected with MERS-CoV, and the risk factors contributing to mortality.
This retrospective observational study focused on the clinical characteristics of confirmed cases of MERS-CoV infection in the Republic of Korea. Approval for this study and a waiver for requiring informed consent were obtained from institutional review board of Chungnam National University Hospital.
All patients with laboratory-confirmed MERS-CoV infection were identified during the outbreak in the Republic of Korea, during which confirmatory tests were performed only for suspected cases. Suspected cases were defined as follows: 1) fever AND pneumonia or acute respiratory distress syndrome (based on clinical or radiologic evidence) AND either a history of travel from countries in or near the Arabian Peninsula within 14 days before symptom onset OR close contact with a symptomatic traveller who developed fever and acute respiratory illness (not necessarily pneumonia) within 14 days after traveling from countries in the Arabian Peninsula. 2) Fever AND symptoms of respiratory illness (e.g., cough, shortness of breath) and presence in a healthcare facility (as a patient, worker, or visitor) within 14 days before symptom onset in a country in or near the Arabian Peninsula. 3) Fever OR symptoms of respiratory illness (e.g. cough, shortness of breath) AND close contact with a confirmed symptomatic MERS case [7]. Since June 4th, 2015, symptomatic people who visited a healthcare facility within 14 days before the onset of symptoms in two or more confirmed healthcare-associated MERS cases were also included as suspected cases. One of the hospitals that treated patients with laboratory-confirmed MERS-CoV infection also performed active surveillance of asymptomatic health care workers who were involved in the care of confirmed cases.
In the Republic of Korea, real-time reverse-transcriptase polymerase chain reaction (rRT-PCR) assays for MERS-CoV diagnosis were performed exclusively at the Korea Centre for Disease Control and Prevention until May 28th, 2015. Medical centres and referral laboratories began to perform rRT-PCR assays for MERS-CoV on May 28th and June 3rd, 2015, respectively. These rRT-PCR tests targeted the upstream E protein (UP-E) and open reading frame 1a (ORF1a) gene segments of MERS-CoV, as described in the WHO guideline [8].
Patient information was collected using a standardized case-report form. Gathering of data was carried out by the physicians who cared for the MERS-CoV-infected patients of each hospital. A major means of obtaining data was review of medical records, including doctors' and nurses' charts, imaging findings, and laboratory results. In those cases where additional information was necessary, re-interview of patients was performed.
Collected data were categorized with respect to demographics, clinical symptoms, comorbidities, laboratory test results, image findings, treatments performed, and clinical outcomes. We carried out descriptive analysis for the aforementioned categories. Continuous variables were reported as means with standard deviations or medians with ranges. For categorical variables, the proportion of patients was calculated for each variable. Comparison analysis for subgroups was performed using Student's t-test, Pearson's chi-square test, or Fisher's exact test, and Cox-regression analysis was also performed to evaluate risk factors of mortality. For Cox-regression analysis, the data was censored at September 30, 2015 for survivors. A P-value <0.05 was considered to indicate statistical significance. All analysis was performed with SPSS software (SPSS version 20.0, SPSS Inc., Chicago, IL, USA) for Windows.
We described the clinical characteristics of all 186 patients with confirmed MERS-CoV infection during the outbreak in the Republic of Korea (Table 1). We also compared the clinical characteristics of survivors and deceased at 28 days after symptom onset.
The median age of the patients was 55 years (range, 16 to 86); 52.2% of the patients were 55 years of age or older (Table 1). Male patients predominated in number over female patients, with a sex ratio of approximately 3:2. A total of 55.4% of the patients had one or more coexisting medical conditions. Hypertension, diabetes, and solid organ malignancy were the most common coexisting medical conditions. Thirty-nine patients (21.0%) were health care workers (HCWs), of which nurses were most common (38.6%), and two cases (1.1%) were asymptomatic. Compared with survivors, the deceased were older and more frequently had coexisting medical conditions.
One hundred fifty-two patients (81.7%) manifested fever and more than 50% had developed cough at admission. Almost all of the patients (95.2%) developed fever during the course of disease. Unstable vital signs were observed in approximately a quarter of the patients at admission. Compared with survivors, the deceased more frequently presented with dyspnea, hypotension, and tachypnea at admission.
At admission, 178 (95.7%) of the 186 patients underwent one or more blood tests (Table 1) including complete blood counts in 178 (95.7%), C-reactive protein (CRP) analyses in 168 (90.3%), and liver function and blood chemistry tests in 173 (93.0%). Among those, 42.1% had leukopenia (<4,000 cells/mm3), 4.5% had leukocytosis (>10,000/mm3), 32.0% had anemia (<12 g/dL), and 46.6% had thrombocytopenia (<150,000/mm3). CRP was elevated (>3 mg/dL) in 47.6% of patients. Elevated level of aspartate aminotransferase (AST) was observed in 43.4%, whereas elevated creatinine was observed in only 5.8% of patients. Among the 103 patients who underwent urinalysis, 44(42.7%) had proteinuria and 36(35.0%) had hematuria. Forty-four patients underwent arterial blood gas analysis, which revealed a decreased PaO2:FiO2 ratio (<300) in 56.8% of patients. Compared with survivors, the deceased more frequently exhibited leucocytosis, hypoalbuminaemia, elevated serum creatinine and CRP levels, and hypoxemia (PaO2:FiO2 ratio <300).
Abnormalities in chest radiography were detected in 123 of 180 patients (68.3%) who underwent chest radiography at admission and 147 (80.8%) of the 182 patients manifested pneumonia during the course of disease. The most common features of the abnormalities were ground glass opacity or consolidation. The abnormal findings appeared in peripheral (78.9%), focal (57.1%), and unilateral (62.6%) patterns. Multifocal or bilateral location was relatively rare (42.9% and 37.4%, respectively). The abnormalities had disappeared in only one-third of patients by the time of discharge.
Antiviral therapy was administered to 139 patients (74.7%; Table 2). The median time from the onset of illness to the initiation of antiviral therapy was 6 days (range, 1–20 days), although 14.0% of patients received antiviral therapy within 48 hours of symptom onset. The most commonly prescribed antiviral regimen was a combination of interferon (IFN), ribavirin, and lopinavir/ritonavir. One hundred thirty-eight patients (75.0%) received antibiotic therapy. In addition, 45 patients (24.5%) were treated with mechanical ventilation, 15 (8.2%) with haemodialysis, and 13 (7.1%) with extracorporeal membrane oxygenation (ECMO). Seven (3.8%) patients were treated with convalescent serum.
Acute respiratory distress syndrome, acute kidney injury, and shock occurred in 20.7%, 14.0%, and 13.4% of patients, respectively. Among the 186 patients, a total of 38 (20.4%) died in hospitals. From the day of symptom onset, seven patients (3.8%) died within 7 days and 33 (17.7%) died within 28 days. The median interval from symptom onset to death was 14 days (range, 1–174 days). Two patients (1.1%) died before confirmation of MERS.
Among cured patients, the median interval from symptom onset to discharge was 21 days (range, 7–187 days), the median duration of fever was 8 days (range, 0–54 days), and the median time to a negative conversion of virus as determined via rRT-PCR analysis of sputum was 17 days (range, 4–45 days).
In univariate analysis, the deceased were older than the survivors (median age, 69 vs. 51 years), and included a smaller portion of HCWs than did the survivors (2.8 vs. 25.3%). The deceased had fever, dyspnea, or decreased consciousness more frequently at admission. They had coexisting medical conditions more frequently, especially diabetes, chronic heart disease, chronic lung disease, and solid organ malignancy. Hypotension and tachypnea were observed more frequently in the deceased. Laboratory abnormalities, namely leukocytosis, thrombocytopenia, hypoalbuminemia, elevated serum creatinine, and elevated CRP, were reported more frequently in the deceased at admission and during the course of disease. However, leukopenia was reported more frequently in survivors.
In multivariate Cox-regression analysis, age ≥55 years, occurrence of dyspnea during the disease course, presence of coexisting medical conditions including diabetes or chronic lung disease, systolic blood pressure <90 mm Hg at admission, leukocytosis at admission, and the use of mechanical ventilation were found to be independent predictors of death (Table 3).
The use of mechanical ventilation was assumed to be an independent indicator of severe disease. Among the 45 patients with mechanical ventilation, 28 (55.6%) died. The deceased among patients with mechanical ventilation were older than the survivors, and the proportion of HCWs was smaller than among the survivors (Table 4). They also had underlying diseases more frequently, especially chronic lung disease and solid organ malignancy. Elevation of serum creatinine was observed more frequently in the deceased. In contrast, the occurrence of diarrhea and the elevation of ALT were observed more frequently in survivors. ECMO or convalescent serum was employed more frequently among survivors.
Since its initial identification in Saudi Arabia in 2012, 1,638 cases of laboratory-confirmed MERS-CoV infection have been reported as of February 26th, 2016. In that time interval, another multi-facility outbreak in an endemic area has occurred since the first healthcare facility-related outbreak was reported by Assiri, et al [9]. Here, we describe the clinical findings of 186 MERS patients in the Republic of Korea from May through September 2015. To the best of our knowledge, this is the largest report of an outbreak outside of the Middle East. In this study of the MERS outbreak in the Republic of Korea, the reported data suggest that MERS-CoV is most likely to be transmitted as a healthcare-associated infection. No community-associated infections lacking any history of contact with MERS-CoV were found in this outbreak. Even though the overall transmissibility of MERS-CoV was relatively low, there were several so-called 'super-spreading' events posing a serious threat to public health. Rapid transmission and high attack rates in dialysis units were noted, in a previous report, in Saudi Arabia [5]. However, no additional cases in dialysis units were reported in this Korean outbreak, even though many patients undergoing hemodialysis were exposed to MERS in dialysis units. This apparent heterogeneity in transmission, with many infected patients not transmitting disease at all and several patients transmitting disease to others, may be characteristic of MERS-CoV infections, similar to SARS [451011].
The clinical features of patients with MERS-CoV infection in the Republic of Korea were generally similar to those reported in Saudi Arabia [4510]. Asymptomatic patients were rare in this outbreak, presumably because confirmatory MERS-CoV tests were performed only for symptomatic patients, although active surveillance of asymptomatic HCWs involved in patient care was conducted at one hospital. In a previous outbreak in Jeddah, 25.1% of laboratory-confirmed cases were reported to have been asymptomatic; however, a telephone survey revealed that about 80% of those who were reached by telephone had experienced at least one symptom [4]. The proportions of patients with underlying medical conditions such as diabetes (18.8%), chronic lung disease (10.2%), or chronic kidney disease (4.8%) were smaller in this study, which might explain why cases with complicating respiratory or renal failure were relatively uncommon in the outbreak in the Republic of Korea, compared to those reported in Saudi Arabia. The incidence of pneumonia (80.8%) in this outbreak is similar to that reported in Saudi Arabia, although the proportion of patients requiring mechanical ventilation was smaller (24.2% in the Republic of Korea vs. 56-78% in Saudi Arabia) [59]. In addition to the reduced presence of comorbidities, many patients in this study were diagnosed relatively early because rRT-PCR tests for MERS-CoV were actively performed for those with an epidemiological linkage as soon as fever or respiratory symptoms occurred. This early diagnosis might have led to early treatment initiation and thus prevented disease progression to a severe status.
The case fatality rate observed in this study was lower than that reported in other studies of previous MERS outbreaks (20.4% vs. 36.5–65%) [4510]. This low case fatality rate could be attributed to the application of aggressive treatment measures, including antiviral agents, mechanical ventilation, or ECMO. Antiviral treatments were administered to 74.5% of the patients, although clinical decisions for their use were made by attending physicians. During the MERS-CoV epidemic in the Republic of Korea, the Korean Society of Infectious Diseases and the Korean Society for Chemotherapy collaborated to generate and distribute recommendations for the use of antiviral treatments based on existing available data from experiences with SARS and MERS [1213]. We were not able to assess the clinical impact of antiviral therapy on outcomes, as most patients with severe pneumonia received combination antiviral therapy along with comprehensive supportive care. The therapeutic efficacy of combination antiviral therapy should be evaluated in further studies.
Our study has several limitations. First, clinical data were retrospectively collected through electronic medical records and chart review, although cases were enrolled prospectively by means of active surveillance of outbreaks. The present study was observational, and thus unknown risk factors and bias might have been unequally distributed between the two groups in the survival analysis. The possibility of limitations that preclude accurate comparisons should be kept in mind given the observational nature of this study. Clinical judgments regarding management of patients were made by attending physicians, not by researchers. Second, we have presented 4 months of clinical data after control of the outbreak, and thus the data might represent acute complications of MERS-CoV-infected patients. Only 36.2% of patients showed resolution of abnormal findings on chest radiologic evaluation, and there are concerns regarding chronic complications such as pulmonary fibrosis. Further follow-up prospective cohort studies are warranted in the future. Third, although this study was performed with a nationwide database, a relatively small number of patients with mechanical ventilation were included, with limited statistical power. The impact of ECMO and convalescent serum therapy on survival may not be fully adjusted for in the survival analysis. Finally, this study was conducted mainly at large referral centers in Korea, and our findings may not be generalizable to other institutions outside of Korea.
In conclusion, a MERS-CoV outbreak in the Republic of Korea was initiated by a patient recently returned from Saudi Arabia, and transmitted as a healthcare-associated infection. Although clinical features did not differ significantly from those of previous outbreaks in the Middle East, the overall mortality rate was 20.4%, which was lower than that reported in other countries. The early detection of cases with MERS-CoV infection and active management of patients during this outbreak may have improved patient outcomes. Our data suggest that MERS-CoV infections pose an important public health threat in regions outside the Middle East, and that more information on the emergence and dissemination of this virus is necessary in order to prevent its further spread.
Figures and Tables
Table 1
Clinical features of 186 patients with confirmed Middle East respiratory syndrome-coronavirus infection and comparison of those between survivors and deceased at 28 days after symptom onset
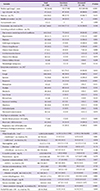
Table 2
Treatment modalities administered to patients with confirmed Middle East respiratory syndrome-coronavirus infection

Table 3
Risk factrs for death frm Middle East respiratory syndromea

Table 4
Comparison of survivors and deceased among severe patients requiring mechanical ventilation

Acknowledgements
This work was supported by a grant (2015P2600100) from the Research of Korea Centers for Disease Control and Prevention (KCDC). We express the deepest thanks to physicians, field epidemiologists, and the officials of KCDC, who did not spare the dedicated efforts for the control of the MERS outbreak in Korea.
References
1. Zaki AM, van Boheemen S, Bestebroer TM, Osterhaus AD, Fouchier RA. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012; 367:1814–1820.

2. Cotten M, Watson SJ, Zumla AI, Makhdoom HQ, Palser AL, Ong SH, Al Rabeeah AA, Alhakeem RF, Assiri A, Al-Tawfiq JA, Albarrak A, Barry M, Shibl A, Alrabiah FA, Hajjar S, Balkhy HH, Flemban H, Rambaut A, Kellam P, Memish ZA. Spread, circulation, and evolution of the Middle East respiratory syndrome coronavirus. MBio. 2014; 5:pii:e01062-13.

3. Memish ZA, Zumla AI, Al-Hakeem RF, Al-Rabeeah AA, Stephens GM. Family cluster of Middle East respiratory syndrome coronavirus infections. N Engl J Med. 2013; 368:2487–2494.

4. Oboho IK, Tomczyk SM, Al-Asmari AM, Banjar AA, Al-Mugti H, Aloraini MS, Alkhaldi KZ, Almohammadi EL, Alraddadi BM, Gerber SI, Swerdlow DL, Watson JT, Madani TA. 2014 MERS-CoV outbreak in Jeddah--a link to health care facilities. N Engl J Med. 2015; 372:846–854.

5. Assiri A1, McGeer A, Perl TM, Price CS, Al Rabeeah AA, Cummings DA, Alabdullatif ZN, Assad M, Almulhim A, Makhdoom H, Madani H, Alhakeem R, Al-Tawfiq JA, Cotten M, Watson SJ, Kellam P, Zumla AI, Memish ZA. Hospital outbreak of Middle East respiratory syndrome coronavirus. N Engl J Med. 2013; 369:407–416.

6. Korea Centers for Disease Control and Prevention (KCDC). Middle East Respiratory Syndrome coronavirus outbreak in the Republic of Korea, 2015. Osong Public Health Res Perspect. 2015; 6:269–278.
7. Korea Centers for Disease Control and Prevention (KCDC). Response guidelines for Middle East respiratory syndrome (the 3rd revision). Accessed 22 June 2016. Available at: http://www.cdc.go.kr/CDC/info.
8. World Health Organization (WHO). Laboratory testing for Middle East Respiratory Syndrome Coronavirus - Interim recommendations (revised) September 2014. Accessed 22 June 2016. Available at: http://www.who.int/csr/disease/coronavirus infections/en/.
9. Assiri A, Abedi GR, Bin Saeed AA, Abdalla MA, al-Masry M, Choudhry AJ, Lu X, Erdman DD, Tatti K, Binder AM, Rudd J, Tokars J, Miao C, Alarbash H, Nooh R, Pallansch M, Gerber SI, Watson JT. Multifacility Outbreak of Middle East Respiratory Syndrome in Taif, Saudi Arabia. Emerg Infect Dis. 2016; 22:32–40.

10. Assiri A, Al-Tawfiq JA, Al-Rabeeah AA, Al-Rabiah FA, Al-Hajjar S, Al-Barrak A, Flemban H, Al-Nassir WN, Balkhy HH, Al-Hakeem RF, Makhdoom HQ, Zumla AI, Memish ZA. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: a descriptive study. Lancet Infect Dis. 2013; 13:752–761.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download