1. National Institute of Dementia (KR). National Institute of Dementia annual report 2017. Seongnam: National Institute of Dementia;2018.
2. George LK, Gwyther LP. Caregiver well-being: a multidimensional examination of family caregivers of demented adults. Gerontologist. 1986; 26:253–259. PMID:
3721232.
3. Dauphinot V, Delphin-Combe F, Mouchoux C, Dorey A, Bathsavanis A, Makaroff Z, et al. Risk factors of caregiver burden among patients with Alzheimer’s disease or related disorders: a cross-sectional study. J Alzheimers Dis. 2015; 44:907–916. PMID:
25374109.

4. Garand L, Dew MA, Eazor LR, DeKosky ST, Reynolds CF 3rd. Caregiving burden and psychiatric morbidity in spouses of persons with mild cognitive impairment. Int J Geriatr Psychiatry. 2005; 20:512–522. PMID:
15920711.

5. Epstein-Lubow G. Family caregiver health: what to do when a spouse or child needs help. Med Health R I. 2009; 92:106–107. PMID:
19385387.
6. Youn GH. Cultural differences in psychological burden and caregiving obligation of primary caregivers for senile dementia patients. J Korean Geriatr Soc. 1998; 18:75–90.
7. Steinberg M, Tschanz JT, Corcoran C, Steffens DC, Norton MC, Lyketsos CG, et al. The persistence of neuropsychiatric symptoms in dementia: the Cache County Study. Int J Geriatr Psychiatry. 2004; 19:19–26. PMID:
14716695.

8. Aalten P, De Vugt ME, Jaspers N, Jolles J, Verhey FR. The course of neuropsychiatric symptoms in dementia. Part I: findings from the two-year longitudinal Maasbed study. Int J Geriatr Psychiatry. 2005; 20:523–530. PMID:
15920712.
9. Luppa M, Luck T, Brähler E, König HH, Riedel-Heller SG. Prediction of institutionalisation in dementia. A systematic review. Dement Geriatr Cogn Disord. 2008; 26:65–78. PMID:
18617737.
10. Gaugler JE, Kane RL, Kane RA, Newcomer R. Early community-based service utilization and its effects on institutionalization in dementia caregiving. Gerontologist. 2005; 45:177–185. PMID:
15799982.

11. Kwon OY, Ahn HS, Kim HJ, Park KW. Effectiveness of cognitive behavioral therapy for caregivers of people with dementia: a systematic review and meta-analysis. J Clin Neurol. 2017; 13:394–404. PMID:
29057632.

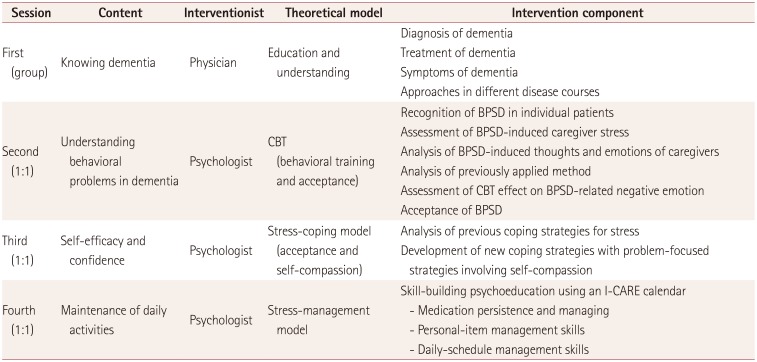
12. Yeom J, Yoo R, Bae S, Kang Y, Kim GH, Na HR, et al. Recent updates of therapeutic intervention programs for caregivers of patient with dementia: proposal of hospital-based individual therapy. Dement Neurocognitive Disord. 2016; 15:29–36.

13. Morris JC. The clinical dementia rating (CDR): current version and scoring rules. Neurology. 1993; 43:2412–2414.
14. Kang Y, Na DL, Hahn S. A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J Korean Neurol Assoc. 1997; 15:300–308.
15. Elliott AF, Burgio LD, Decoster J. Enhancing caregiver health: findings from the resources for enhancing Alzheimer’s caregiver health II intervention. J Am Geriatr Soc. 2010; 58:30–37. PMID:
20122038.

16. Lee HS, Kim DK, Ko HJ, Ku HM, Kwon EJ, Kim JH. Measurement of stress in the caregivers of dementia patients: reliability and validity of the revised-memory and behavior problem checklist and the burden interview. Korean J Clin Psychol. 2004; 23:1029–1050.
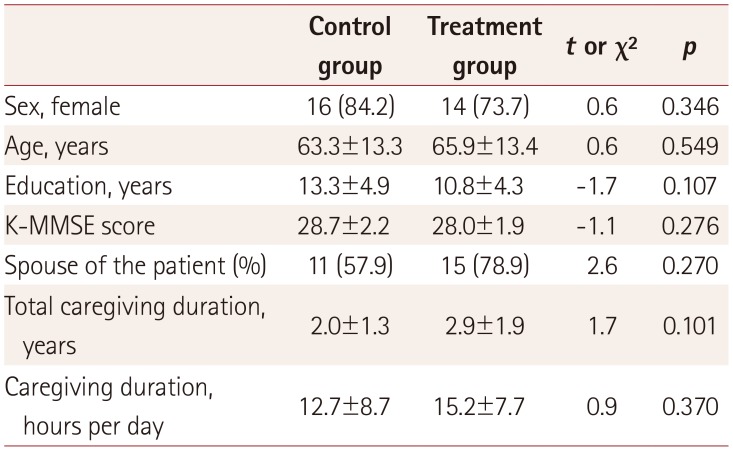
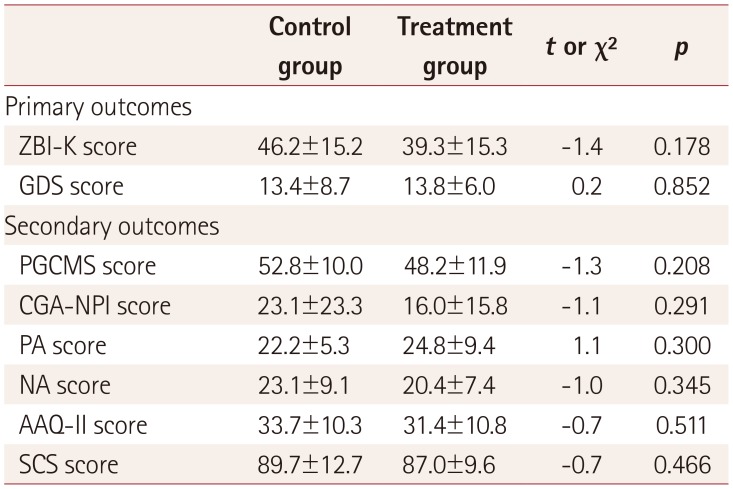
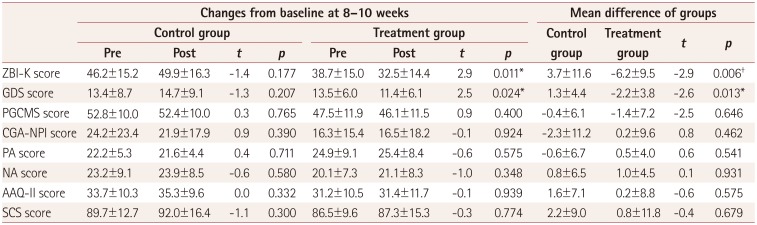
17. Park M, Park MH, Kim HH. Effects of a multimodal psychoeducational program on burden, depression, and problem coping behaviors in family caregivers of patients with dementia. J Korean Gerontol Nurs. 2015; 17:10–19.

18. Cho MJ, Bae JN, Suh GH, Hahm BJ, Kim JK, Lee DW, et al. Validation of geriatric depression scale, Korean version(GDS) in the assessment of DSM-III-R major depression. J Korean Neuropsychiatr Assoc. 1999; 38:48–63.
19. Ryu K, Kim JH, Kang YW, Rie JI. A validation study of the Korean version of Philadelphia Geriatric Center for Morale Scale. J Korean Geriatr Soc. 2012; 32:207–222.
20. Kang SJ, Choi SH, Lee BH, Jeong Y, Hahm DS, Han IW, et al. Caregiver-administered neuropsychiatric inventory (CGA-NPI). J Geriatr Psychiatry Neurol. 2004; 17:32–35. PMID:
15018695.

21. Lee HH, Kim EJ, Lee MK. A validation study of Korea positive and negative affect schedule: the PANAS scales. Korean J Clin Psychol. 2003; 22:935–946.
22. Heo JH, Choi MS, Jin HJ. Study on the reliability and validity of Korean translated Acceptance-Action Questionnaire-II. Korean J Couns Psychother. 2009; 21:861–878.
23. Kim KE, Yi GD, Cho YR, Chai SH, Lee WK. The validation study of the Korean version of the Self-Compassion Scale. Korean J Health Psychol. 2008; 13:1023–1044.
24. Choi JY, Jeong H, Park JY, Kim TH, Lee DY, Lee DW, et al. Factors associated with the attitudes toward dementia in community caregivers: results from the nationwide survey on dementia care in Korea. J Korean Geriatr Psychiatry. 2015; 19:24–31.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download