INTRODUCTION
Gastrointestinal (GI) duplication cysts represent a myriad of congenital anomalies characterized by an epithelial lining from, and an attachment to, some part of the alimentary tract. Gastric duplication cysts (GDCs) constitute 4% of these anomalies. Mostly diagnosed in children, the presentation of GDC varies from an asymptomatic abdominal mass to fulminant or massive GI bleeding. GDCs are usually diagnosed using computerized tomography and treated with surgical resection.
CASE REPORT
Our patient was a 10-month-old infant admitted because of unexplained massive GI bleeding. The child was born to consanguineous healthy parents. His perinatal history was uneventful. He had no medical problems and did not take any medications. Five days prior to admission, the patient developed severe hematemesis and melena. On admission, the physical exam revealed a pale, dehydrated child, with mild growth retardation. Abdominal ultrasonography revealed no abnormalities. Esophagogastroduodenoscopy (EGD) to the proximal jejunum was negative. Contrast-enhanced computed tomography (CT) was ordered, but was delayed for technical difficulties. Two days later, the patient experienced another episode of massive melena and hematochezia. An urgent EGD revealed two large ulcers on the greater curvature of the stomach, in a place that appeared completely normal on the preceding examination (
Fig. 1B). Biopsies of the gastric ulcers were non-diagnostic. An ileocolonoscopy was normal. A repeat EGD three days later revealed a large orifice in place of the aforementioned ulcers, with evident gastric mucosa appearing behind the opening (
Fig. 1C). The orifice was intubated, disclosing another cavity with multiple ulcerations and a patent opening of 1 cm proximal to the pylorus (
Supplementary Video 1;
Fig. 1D). The multiple ulcerations of the GDC mucosa provided an explanation for the patient's bleeding episodes. CT revealed doubling of the esophageal lumen, an expanded doubled stomach, and stricture of the pyloric canal (
Fig. 2). The cyst was resected surgically, and pathological evaluation indicated a tubular-shaped cyst lined with ulcerated pyloric mucosa without any signs of neoplasia (
Fig. 3). The patient recovered well after surgery with no recurrence of symptoms on follow-up. One of the patient's parents provided written consent for the inclusion of the patient's data in this report, acknowledging that the patient is fully anonymized and cannot be identified from this paper.
Fig. 1
Development of the ulcers and the consequent orifice, along with the cyst connection to the pylorus. (A) Intact gastric mucosa. (B) The black arrows point to the ulcers that measure 2×2 cm and 5×5 cm, while the white arrow points to the gastric cardia. (C) The orifice of the cyst. (D) The white arrow points to the pyloric sphincter, while the black arrows point to the cyst opening to the pylorus.
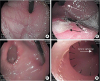
Fig. 2
Computerized tomography images. Both thin and thick white arrows point to different ends of the gastric duplication cyst.
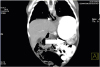
Fig. 3
(A) The gross appearance of the resected cyst. (B, C) Light microscopy of the gastric duplication cyst exhibiting gastric pyloric mucosa (B, H&E stain ×100; C, H&E stain ×600).
H&E: hematoxylin and eosin.

DISCUSSION
Identified in one of 4,500 live births, GI duplication cysts are peculiar congenital malformations. Although they can present anywhere along the GI tract, they are common in the ileum, esophagus, and colon, while rare in the pharynx and tongue. GDCs constitute 4% to 8% of these anomalies and thus, have an approximate frequency of 1/100,000 live births [
12]. Many hypotheses have been proposed to explain the embryonic origin of these lesions [
3]. Most cysts are composed of smooth muscle and alimentary tract mucosa [
3]. GDCs are often found at the greater or lesser curvature, can be spherical or tubular, and can sometimes communicate with an adjacent GI structure. In the vast majority of cases, GDCs are spherical non-communicating cysts. Rarely, GDCs may communicate with the gastric cavity [
14], the chest wall, colon, spleen [
5], pancreatic duct, and intrahepatic bile duct [
6].
Most GDCs are diagnosed in the first years of life. Rarely, they may be diagnosed in adults, either as an incidental finding or due to a complication [
7]. GDCs commonly present as an abdominal mass or with vomiting due to gastric outlet obstruction. Rarely, a GDC may present with a gastric ulcer, hemorrhage, failure to thrive, perforation, abdominal pain, pancreatitis, hemoptysis [
7], or hemobilia [
6]. GDC-associated massive GI hemorrhage has been rarely reported in the literature. These reports mention ulcers, perforation, and splenic capsular erosion as possible causes of the bleeding [
35].
GDCs are associated with different types of ulcers. These could be gastric, duodenal, or colonic [
1]. Gastric ulcers could affect the mucosa of the stomach or the gastric lining of the cyst [
4]. These ulcers can cause GI bleeding, abdominal pain, perforation, and penetration, as reported in our case [
47]. Hypergastrinemia has been reported as a peculiar associated finding [
1]. It can be an important ulcer-precipitating factor in GDCs. Of note, the serum gastrin level was normal in our case.
In 1937, Ladd introduced the term “duplication of the alimentary tract”, and set the diagnostic criteria for GI duplications. The criteria included the presence of a well-developed smooth muscle layer, the presence of an epithelial alimentary tract lining, and an attachment to part of the alimentary tract [
8]. Duplication cysts are usually described by the adjacent alimentary tract, rather than by the mucosal lining [
3]. The cyst in our case fulfilled these criteria. Moreover, since it was adjacent to the stomach, it was identified as a GDC. GDCs are typically diagnosed by ultrasonography, which usually shows a bowel wall signature. Other diagnostic modalities include contrast CT/magnetic resonance imaging, endoscopic ultrasound, and 99m technetium pertechnetate scintigraphy that can be used effectively to detect cysts with a gastric mucosa [
23]. Endoscopic studies are not usually used to diagnose GDCs. However, the delay in obtaining the CT contributed to the endoscopic diagnosis with documented progression of a complicated GDC, further highlighting the peculiarity of this case.
GDCs have been cited as premalignant lesions and long-term complications include malignant transformation, which has been commonly described in the literature [
910]. All this mandate surgical removal, regardless of the age of the patient at presentation or the symptoms.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download