1. Mahmud E, Madani MM, Kim NH, et al. Chronic thromboembolic pulmonary hypertension: evolving therapeutic approaches for operable and inoperable disease. J Am Coll Cardiol. 2018; 71:2468–2486. PMID:
29793636.
2. Lang IM, Pesavento R, Bonderman D, Yuan JX. Risk factors and basic mechanisms of chronic thromboembolic pulmonary hypertension: a current understanding. Eur Respir J. 2013; 41:462–468. PMID:
22700839.
3. Ende-Verhaar YM, Cannegieter SC, Vonk Noordegraaf A, et al. Incidence of chronic thromboembolic pulmonary hypertension after acute pulmonary embolism: a contemporary view of the published literature. Eur Respir J. 2017; 49:1601792. PMID:
28232411.
4. Park JS, Ahn J, Choi JH, et al. The predictive value of echocardiography for chronic thromboembolic pulmonary hypertension after acute pulmonary embolism in Korea. Korean J Intern Med. 2017; 32:85–94. PMID:
27044855.
5. Galiè N, Humbert M, Vachiery JL, et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Heart J. 2016; 37:67–119. PMID:
26320113.
6. Feinstein JA, Goldhaber SZ, Lock JE, Ferndandes SM, Landzberg MJ. Balloon pulmonary angioplasty for treatment of chronic thromboembolic pulmonary hypertension. Circulation. 2001; 103:10–13. PMID:
11136677.
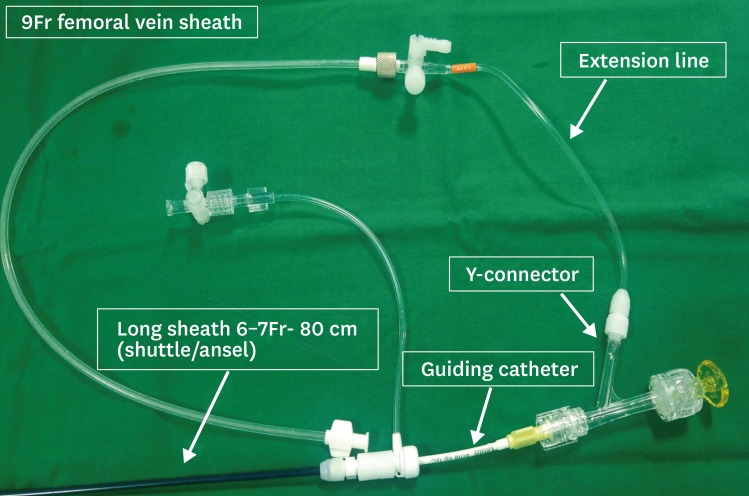
7. Mizoguchi H, Ogawa A, Munemasa M, Mikouchi H, Ito H, Matsubara H. Refined balloon pulmonary angioplasty for inoperable patients with chronic thromboembolic pulmonary hypertension. Circ Cardiovasc Interv. 2012; 5:748–755. PMID:
23192917.
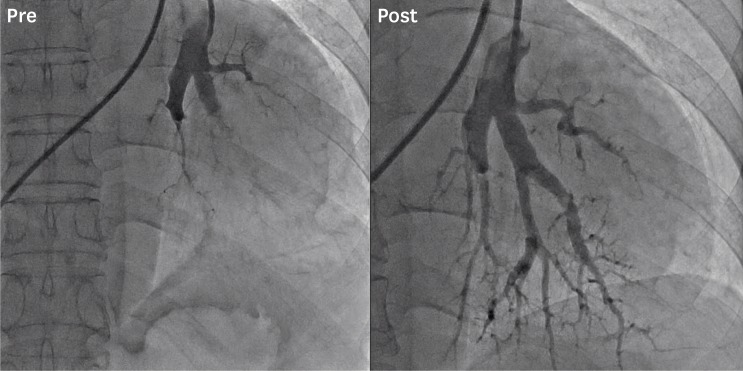
8. Kwon W, Yang JH, Park TK, et al. Impact of balloon pulmonary angioplasty on hemodynamics and clinical outcomes in patients with chronic thromboembolic pulmonary hypertension: the initial Korean experience. J Korean Med Sci. 2018; 33:e24. PMID:
29318791.
9. Oh DK, Song JM, Park DW, et al. The effect of a multidisciplinary team on the implementation rates of major diagnostic and therapeutic procedures of chronic thromboembolic pulmonary hypertension. Heart Lung. 2019; 48:28–33. PMID:
30115494.
10. Fedullo PF, Auger WR, Channick RN, Kerr KM, Rubin LJ. Chronic thromboembolic pulmonary hypertension. Clin Chest Med. 2001; 22:561–581. PMID:
11590849.
11. Ogawa A, Kitani M, Mizoguchi H, et al. Pulmonary microvascular remodeling after balloon pulmonary angioplasty in a patient with chronic thromboembolic pulmonary hypertension. Intern Med. 2014; 53:729–733. PMID:
24694486.
12. Fedullo PF, Auger WR, Kerr KM, Rubin LJ. Chronic thromboembolic pulmonary hypertension. N Engl J Med. 2001; 345:1465–1472. PMID:
11794196.
13. Galiè N, Hoeper MM, Humbert M, et al. Guidelines for the diagnosis and treatment of pulmonary hypertension: the Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS), endorsed by the International Society of Heart and Lung Transplantation (ISHLT). Eur Heart J. 2009; 30:2493–2537. PMID:
19713419.
14. Ogawa A, Satoh T, Fukuda T, et al. Balloon pulmonary angioplasty for chronic thromboembolic pulmonary hypertension: results of a multicenter registry. Circ Cardiovasc Qual Outcomes. 2017; 10:e004029. PMID:
29101270.
15. Kim NH, Delcroix M, Jais X, et al. Chronic thromboembolic pulmonary hypertension. Eur Respir J. 2018; [Epub ahead of print].
16. Ogawa A, Matsubara H. After the dawn - balloon pulmonary angioplasty for patients with chronic thromboembolic pulmonary hypertension. Circ J. 2018; 82:1222–1230. PMID:
29669971.
17. Pepke-Zaba J, Ghofrani HA, Hoeper MM. Medical management of chronic thromboembolic pulmonary hypertension. Eur Respir Rev. 2017; 26:160107. PMID:
28356404.
18. Ghofrani HA, D'Armini AM, Grimminger F, et al. Riociguat for the treatment of chronic thromboembolic pulmonary hypertension. N Engl J Med. 2013; 369:319–329. PMID:
23883377.
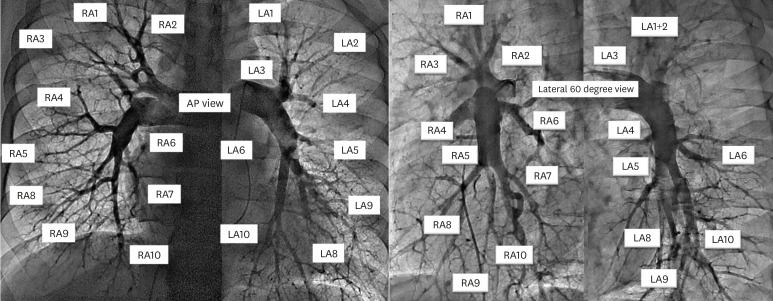
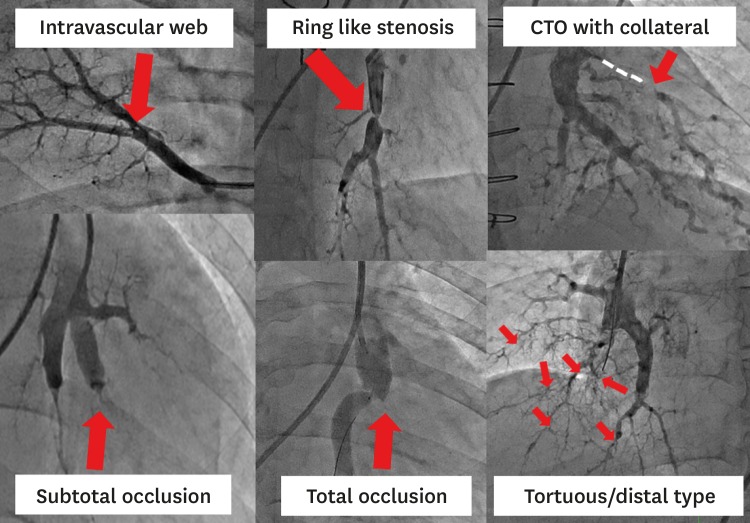
19. Kawakami T, Ogawa A, Miyaji K, et al. Novel angiographic classification of each vascular lesion in chronic thromboembolic pulmonary hypertension based on selective angiogram and results of balloon pulmonary angioplasty. Circ Cardiovasc Interv. 2016; 9:e003318. PMID:
27729418.
20. Kataoka M, Inami T, Hayashida K, et al. Percutaneous transluminal pulmonary angioplasty for the treatment of chronic thromboembolic pulmonary hypertension. Circ Cardiovasc Interv. 2012; 5:756–762. PMID:
23132237.
21. Sugimura K, Fukumoto Y, Satoh K, et al. Percutaneous transluminal pulmonary angioplasty markedly improves pulmonary hemodynamics and long-term prognosis in patients with chronic thromboembolic pulmonary hypertension. Circ J. 2012; 76:485–488. PMID:
22185711.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download