Dear Editor,
Subacute combined degeneration (SCD) is a potentially reversible and treatable myelopathy that primarily affects the dorsolateral columns of the spinal cord. It is the well-documented complication of cobalamin (vitamin B12) deficiency, which is often observed in patients who are suffering from pernicious anemia, various conditions leading to malnutrition, tropical sprue, or HIV infection.1 Nitrous oxide (N2O) exerts neurotoxic effects by interfering with the bioavailability of cobalamin, but N2O as an etiology of the condition is not well known and often underestimated. Herein we report a case series of two patients diagnosed with SCD caused by massive N2O inhalation.
Patient A was a 22-year-old woman who presented with progressive paresthesia in her legs and hands, and unsteady gait that had first appeared about 2 months previously. She was currently taking medication for depression. She had been diagnosed and treated for a pulmonary embolism 1 month previously. In addition, she was injected intramuscularly with single high-dose supplementation of cobalamin for incidentally found cobalamin deficiency. Further questioning revealed a habit of inhaling ‘Happy Balloons’ more than 100 times for the past 2 months. A neurological examination revealed decreased tactile and vibratory sensations of the legs, dysmetria in heel-to-shin tests on both sides, a positive Romberg's sign, and bilateral weakness of both lower extremities (each with Medical Research Council grade III).
Patient B was a 33-year-old man who presented with progressive symmetrical numbness in his legs, imbalance, and difficulty walking. He had a history of a diagnosis of reflux esophagitis with seropositive for Helicobacter pylori (H. pylori), but this was never treated. Further questioning revealed a daily habit of inhaling up to 5 L of ‘laughing gas’ for the past 6 months. A neurological examination revealed weakness in both upper and lower extremities, decreased vibration and proprioception sensations, bilateral hyporeflexia, sensory ataxia, and a positive Romberg's sign.
Both patients A and B had no cognitive impairment or psychiatric problems, with the exception of patient A having suffered from depression several years previously. The initial laboratory tests revealed that both patients A and B had elevated mean corpuscular volumes and low cobalamin levels. The folate level was normal in both patients, who were negative for antibodies to human immunodeficiency virus and neurosyphilis. Whole-spine MRI revealed increased T2-weighted signals in the dorsal columns with a characteristic ‘inverted V sign’ (Fig. 1A and C). A nerve conduction study showed axonal motor polyneuropathy (Fig. 1B and D), and somatosensory evoked potentials suggested a lesion in somatosensory pathways between the upper cervical cord and somatosensory cortex.
Both patients were diagnosed with SCD of the spinal cord induced by N2O consumption, and they were treated with daily intramuscular cobalamin (1 mg) injections for 2 weeks followed by oral medication. This cobalamin treatment combined with the removal of the offending agent resulted in both patients showing a gradual neurological improvement with rehabilitation.
Cobalamin is an essential cofactor for cellular reactions, with it supporting methionine synthetase and further participating in the synthesis of myelin sheath.2 Low cobalamin levels may result in various neurological manifestations such as peripheral neuropathy, myelopathy with dorsal column dysfunction, and encephalopathy with cognitive decline.3 There have been many reports of early diagnosis and rapid treatment being crucial to a favorable outcome.
N2O was originally used as an inhalational anesthetic agent. However, its recreational consumption is increasing due to its easy accessibility and low cost. The consumption of a large amount of N2O accelerates the irreversible oxidation of the cobalt ion of cobalamin, which results in the inactivation of the cofactor required for myelin formation.123 Any patients with preexisting conditions for SCD—anorexia nervosa and H. pylori in our two cases—or a chronically low cobalamin level may be more vulnerable to the adverse effects of N2O.4 It is paramount that clinicians inquire about and perform screening for N2O abuse if there is no clear etiology of SCD, especially in young patients without predisposing conditions.
Figures and Tables
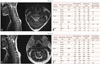 | Fig. 1Findings of MRI and nerve conduction studies in both patients. Sagittal and axial T2-weighted images exhibited hyperintensities in the cervical cord region with an ‘inverted V sign’ (arrows) typical of subacute combined degeneration (A and C). Nerve conduction studies revealed axonal motor neuropathy (B and D). *Abnormal values. ADM: abductor digiti minimi muscle, AH: abductor hallucis brevis muscle, APB: abductor pollicis brevis muscle, EDB: extensor digitorum brevis muscle, FH: fibular head, LT: left, ND: not done, NR: no response, PF: popliteal fossa, RT: right, TA: tibialis anterior muscle. |
References
1. Green R, Kinsella LJ. Current concepts in the diagnosis of cobalamin deficiency. Neurology. 1995; 45:1435–1440.


2. Jordan JT, Weiser J, Van Ness PC. Unrecognized cobalamin deficiency, nitrous oxide, and reversible subacute combined degeneration. Neurol Clin Pract. 2014; 4:358–361.







 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download