This article has been
cited by other articles in ScienceCentral.
Abstract
Transcatheter aortic valve replacement (TAVR) or transcatheter aortic valve implantation (TAVI) for severe aortic stenosis (AS) is a minimally invasive interventional procedure that repairs a valve without removing the old, damaged valve. Instead, a replacement valve is wedged into the location of the native aortic valve. During TAVR, contrast is used for conventional aortic root angiography, positioning of the TAVR valve device, and assessing the peripheral vasculature. Therefore, contrast-induced acute kidney injury (AKI) is a major concern when performing TAVR and is associated with increased mortality in patients with impaired renal function. Although the exact mechanism of post-TAVR AKI is unknown and appears multifactorial, contrast medium has been reported as a major contributing factor. We report a case of zero-contrast TAVR for severe AS in a patient with chronic kidney disease (CKD). The procedure was successfully performed with only fluoroscopic and transesophageal echocardiography (TEE) guidance.
Keywords: Transcatheter aortic valve replacement, Acute kidney injury, Chronic kidney diseases, Aortic stenosis
INTRODUCTION
Transcatheter aortic valve replacement (TAVR) or transcatheter aortic valve implantation (TAVI) for severe aortic stenosis (AS) is the minimally invasive interventional procedure that repair valve without removing the old, damaged valve. Instead, it wedges a replacement into the place of native aortic valve.
1
During TAVR, contrast is used for conventional aortic root angiography, positioning of the TAVR valve device and assessing the peripheral vasculature. Therefore, contrast induced acute kidney injury (AKI) is one of the major concerns of TAVR and is associated with increased mortality in patients with impaired renal function. Although the exact mechanism of post-TAVR AKI is unknown and seems to be multifactorial, however, contrast medium itself has been reported as a major contributing factor.
2
We report a case of zero-contrast usage for TAVR for severe AS in patient with chronic kidney disease (CKD). This procedure was performed successfully in this case with only fluoroscopic and transesophageal echocardiography (TEE) guidance.
CASE REPORT
An 88-year-old female was admitted due to aggravating dyspnea (New York Heart Association functional class III). She had a history of hypertension, diabetes, CKD and persistent atrial fibrillation. Coronary angiography showed no significant stenotic lesion. Echocardiography showed normal left ventricular ejection fraction but severe AS was noted (
Fig. 1). Aortic valve area was 0.96 cm
2 on the planimetry method. Peak velocity across aortic valve was 4.2 m/s and mean pressure gradient were 30 mmHg. Society of Thoracic Surgeons score was 8%.
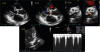 | Fig. 1
Transthoracic echocardiogram showed severe AS (AVA: 0.96 cm2 by continuity equation) with peak/mean pressure gradient (64/33 mmHg).
AS; aortic stenosis, AVA; aortic valve area, BP; blood pressure.

|
She was in CKD stage IV, blood urea nitrogen (BUN) 40.1 mg/dL, creatinine 2.9 mg/dL, epidermal growth factor receptor (eGFR) 14 mL/min/1.73 m
2. Because of CKD, computed tomography (CT) angiography was not done. And she was sensitive to volume overload, so we did not do hydration. From the 3D echocardiographic data (
Fig. 2), the mean annulus diameter was 24.6 mm and the perimeter was 71.2 mm. Distance from annulus to left main and right coronary artery ostium was 16.3 and 15.8 mm, respectively. Because of stage IV CKD, TAVR was planned without using contrast agent.
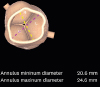 | Fig. 2 Annulus dimension and perimeter derived diameter by 3-dimentional echocardiogram.
|
Under general anesthesia, temporary pacemaker was inserted through right femoral vein. 7 Fr sheath and 6 Fr pig-tail catheter were inserted through right femoral artery under sono-guided puncture technique. 8 Fr sheath was inserted through left femoral artery and replaced with 18 Fr Sentrant sheath. Straight coil wire under back-up with an AL 1 diagnostic catheter was crossed the stenotic aortic valve. Then, straight coil wire was changed to the round-shaped Amplatz stiff wire. Under TEE guidance, a 29-mm Evolut R prosthesis was placed at the optimal position and was deployed successfully (
Fig. 3). After valve implantation, follow-up TEE showed mild para-valvular leak and mild to moderate AR. AR index was 24. All procedure was finished.
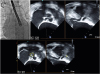 | Fig. 3
Location of catheter was identified by using biplane TEE (dotted circle indicated tip of catheter and asterisk indicated catheter).
TEE; transesophageal echocardiography, PAT; patient, T; temperature.

|
After procedure, her renal function had not been aggravated until she was discharged (directly after: BUN, 46.1 mg/dL; creatinine, 3.33 mg/dL; eGFR, 11.7 mL/min/1.73 m2 and when she was discharged: BUN, 46.1 mg/dL; creatinine, 3.33 mg/dL; eGFR, 13 mL/min/1.73 m2).
DISCUSSION
We have reported a case of zero-contrast TAVR for severe AS in patient with CKD. This case is different from previous TAVR cases in terms of usage of contrast, 3D-echo in pre-evaluation, TEE during intervention, peripheral US in vascular access. TAVR may be an option for people who are considered at intermediate or high risk of complications from surgical aortic valve replacement. AKI remains a major concern in the field of TAVR and is associated with increased mortality, up to twofold to eightfold.
3
AKI in TAVR patients is most likely a combination of prerenal azotemia and direct nephrotoxic influences leading to renal ischemia and acute tubular necrosis (ATN). Causes of prerenal azotemia include (among others) hypovolemia, hemorrhage, impaired cardiac output or renal vasoconstriction caused by vasoconstrictive medication. The normal response of the kidney to prerenal conditions is to concentrate the urine and reabsorb sodium in order to maintain or increase intravascular volume and normalize renal perfusion. Therapy to restore renal perfusion promptly improves renal function. However, prolonged or profound prerenal azotemia can result in ischemic damage leading to ischemic AKI, particularly in combination with the presence of exogenous toxic compounds (e.g., aminoglycosides, contrast media).
So, AKI after TAVR can be considered as the common final path resulting from prerenal azotemia due to pre-, intra- and post-operative factors and additional nephrotoxic influences resulting in ATN.
456
We particularly performed a femoral puncture by vascular ultrasound (
Fig. 4). Furthermore, we used a 3D-echo in pre-evaluation and TEE during TAVR differently from the other cases (
Fig. 5).
7
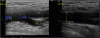 | Fig. 4
Vascular ultrasound showed acceptable access site of left external iliac artery.
CFA; common femoral artery, SFA; superficial femoral artery, DFA; deep femoral artery.

|
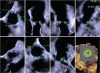 | Fig. 5
3D-echo in pre-evaluation and TEE during TAVR.
TEE; transesophageal echocardiography, TAVR; transcatheter aortic valve replacement, Comm; commissure.

|
Kidney function is a powerful long-term predictor of mortality in patients undergoing TAVR. Patients with severe impaired renal function had the highest mortality after TAVR. Thus, the beneficial effects of TAVR in patients with severe impaired renal function seem to be limited and should be implemented in a decision-making process before TAVR.
8910
All the while, it has been important to do pre-procedural assessments using multimodality imaging including CT angiography and conventional angiography. So, CT angiography and conventional angiography has been thought essential. But, we could overcome this aspect by using 3D-echo in pre-evaluation.
And we have to pay the particular attention to do pre-procedural assessments by using 3D-echo to avoid the increment of complication or failure.
Furthermore, we used TEE during the procedure, TEE-TAVR was associated with similar and midterm results as angiography guiding TAVR and significantly reduced contrast media use during the procedures.
11
In conclusion, this is the case report of zero contrast with TEE guidance. The zero contrast TAVR is more effective method in CKD patients in avoidance of AKI.








 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download